- CDSCO Manufacturing License For Ventilators are regulated as Class C (high-risk) notified medical devices under India's Medical Devices Rules, 2017 — a valid CDSCO manufacturing license is legally mandatory before any domestic manufacturing or sale begins.
- Domestic manufacturers must apply via Form MD-7 or Form MD-9 on the Sugam portal, supported by BIS IS 13450 (Part 2/Sec 12): 2023 and IS 7655 compliance test reports from NABL-accredited laboratories.
- The full approval cycle — from online application to Form MD-9 license grant — typically takes 45 to 90 working days for a well-prepared, GMP-ready applicant; incomplete dossiers or inspection non-conformances can stretch timelines to 4–6 months.
- 2026 regulatory update: CDSCO has tightened post-market surveillance (PMSR) reporting timelines for life-support devices, increased frequency of unannounced GMP inspections, and is actively implementing UDI (Unique Device Identification) alignment for Class C and D devices including ventilators.
Introduction
If you are planning to manufacture ventilators in India, you are stepping into one of the most heavily regulated product categories in the entire medical device sector — and that is entirely justified. Ventilators sustain human life. When they fail or are poorly manufactured, patients die. The Central Drugs Standard Control Organization (CDSCO), operating under the Ministry of Health and Family Welfare, exists precisely to prevent that.
This guide is your complete, honest, and practically grounded reference for the CDSCO manufacturing license for ventilators as of 2026. Whether you are a startup building your first ICU-grade ventilator, an established medical equipment company expanding your product range, or an engineer trying to understand the regulatory pathway, this page covers everything you need — from legal framework and BIS standards to the exact documents, fees, process steps, and post-license compliance obligations.
We have also incorporated the BIS IS 13450 (Part 2/Sec 12): 2023 standard — a critical update that many applicants are still unaware of — alongside the more widely known IS 7655. Getting this right from the beginning saves months of rework.
What Is a Ventilator Under Indian Medical Device Law?
Under Schedule I of the Medical Devices Rules, 2017 (MDR 2017), ventilators are a notified medical device and are classified as Class C — the high-risk tier, just below Class D. This classification is not cosmetic. It determines which licensing authority is competent (CDSCO, not the State Drug Authority), the applicable fee structure, the depth of technical documentation required, and the frequency of post-market oversight.


The regulatory definition of 'ventilator' in India is broad. If your device delivers, controls, or supports mechanical ventilation — even as a secondary function — CDSCO may treat it as a ventilator. The categories that fall within this classification include:
- Invasive mechanical ventilators (ICU-grade, requiring intubation)
- Non-invasive ventilators — BiPAP and CPAP devices used in respiratory therapy
- Transport and ambulance ventilators
- High-frequency oscillatory ventilators (HFOV)
- Neonatal and paediatric ventilators
- Home-care ventilators for chronic respiratory patients
- Anaesthesia workstations with integrated ventilation function
Important: Confirm your device's regulatory classification with CDSCO before starting your application. A device that ventilates — even partially — may be treated as a Class C ventilator, with all associated compliance obligations.
Why Is a CDSCO License Mandatory for Ventilator Manufacturing?
The legal mandate is clear and unambiguous. Under Rule 9 of the Medical Devices Rules, 2017, read with Section 18 of the Drugs and Cosmetics Act, 1940 (as extended to medical devices), no person shall manufacture any notified medical device in India without a valid license from the Central Licensing Authority (CLA). For Class C devices including ventilators, the CLA is CDSCO, New Delhi.
Manufacturing or selling a ventilator without a valid CDSCO license is not a technical oversight that can be regularized later — it is a criminal offence. Consequences include:
- Immediate seizure and destruction of all unlicensed goods
- Cancellation of all existing CDSCO licenses held by the entity
- Substantial monetary penalties under the Drugs and Cosmetics Act
- Criminal prosecution of company directors and responsible persons
- Permanent debarment from government procurement including CMSS and GeM
Regulatory Framework Governing Ventilator Manufacturing in India
| Regulation / Standard | Relevance to Ventilator Manufacturers |
| Drugs and Cosmetics Act, 1940 | Primary statute; criminal and civil liability for violations |
| Medical Devices Rules, 2017 (MDR 2017) | Classification, application process, GMP, PMSR obligations |
| Schedule I (MDR 2017) | Lists ventilators as notified medical devices requiring CDSCO license |
| Schedule III / V (MDR 2017) | GMP standards applicable to Class C device manufacturers |
| IS 13450 (Part 2/Sec 12): 2023 | BIS Indian Standard for lung ventilators — critical 2023 update covering performance and safety requirements aligned to ISO 80601-2-12 |
| IS 7655 | BIS Indian Standard for lung ventilators for medical use (earlier standard, still referenced) |
| IEC 60601-1 | International general safety standard for electrical medical devices |
| ISO 80601-2-12 | International standard for critical care ventilators; referenced by CDSCO |
| IEC 60601-1-2 | Electromagnetic compatibility (EMC) requirements for medical devices |
| ISO 14971 | Risk management for medical devices — mandatory for Class C dossier |
| ISO 13485:2016 | Quality Management System standard effectively required by CDSCO review |
| CDSCO Guidance Documents | SOPs and checklists published on cdsco.gov.in; must be followed |
| DPCO / NPPA | Price control applicable if ventilators are placed under price regulation |
BIS Standards for Ventilators — IS 13450 (Part 2/Sec 12): 2023 and IS 7655 Explained
This is the section most applicants get wrong, and it is the single biggest cause of CDSCO technical queries and application delays in 2025 and 2026. Understanding both the old and the updated BIS standards is essential.


IS 13450 (Part 2/Sec 12): 2023 — The Updated Standard You Must Know
IS 13450 (Part 2/Sec 12): 2023 is the Bureau of Indian Standards (BIS) standard specifically for lung ventilators for medical use. This standard is aligned with ISO 80601-2-12 (Requirements for critical care ventilators) and represents a significant upgrade from earlier ventilator standards. Published in 2023, this is the standard that CDSCO reviewers are now expecting your test reports to reference when you submit a new application or renewal in 2026.
IS 13450 is a multi-part umbrella standard covering medical electrical equipment. Part 2 deals with particular requirements for specific types of medical equipment, and Section 12 covers lung ventilators. The 2023 edition incorporates updated requirements around:
- Alarm system performance and priority classification for critical care ventilators
- Tidal volume accuracy and respiratory rate accuracy under varying patient lung conditions
- PEEP (Positive End-Expiratory Pressure) delivery accuracy
- Software and firmware validation requirements for electronically controlled ventilators
- Electromagnetic compatibility (EMC) performance in hospital environments
- Biocompatibility of patient-contact components (patient circuits, masks)
- Usability and human factors engineering documentation requirements
📌 Action Point: If your test reports reference only IS 7655 or IEC 60601-1 without IS 13450 (Part 2/Sec 12): 2023 alignment, CDSCO is likely to raise a query or reject your dossier at the technical review stage in 2026. Get your test lab to confirm which standards your product has been tested against.
IS 7655 — The Earlier BIS Standard, Still Referenced
IS 7655 is the Bureau of Indian Standards specification for lung ventilators for medical use, published before the IS 13450 series was expanded. It aligns with IEC 60601-1 (general electrical safety) and has historically been the primary BIS reference for ventilators. While IS 13450 (Part 2/Sec 12): 2023 is now the more specific and updated standard, IS 7655 compliance evidence is still referenced in many CDSCO checklists and is expected as part of your technical dossier for electrical safety compliance.


Is BIS Product Registration Separately Mandatory?
As of 2026, BIS product registration under IS 13450 or IS 7655 is not an independent mandatory requirement as a pre-condition for CDSCO licensing — but compliance with the standards (demonstrated by accredited test reports) is effectively mandatory within the CDSCO process. The distinction matters:
- CDSCO licensing requires: Test reports showing compliance with IS 13450 (Part 2/Sec 12): 2023 and IS 7655, submitted as part of your technical dossier.
- BIS product registration (voluntary): A separate BIS registration marking your product compliant with the standard. Not required for CDSCO licensing, but strongly recommended if you intend to bid for government tenders on GeM or CMSS, where BIS registration is frequently a mandatory eligibility criterion.
| Standard | Scope | Applicable To | Status for CDSCO |
| IS 13450 (Part 2/Sec 12): 2023 | Lung ventilators — specific performance, alarms, software (aligns ISO 80601-2-12) | All ventilator types; critical for ICU-grade | Test report required in 2026 dossier |
| IS 7655 | Lung ventilators — electrical safety (aligns IEC 60601-1) | All domestic ventilator manufacturers | Test report referenced in CDSCO checklist |
| IEC 60601-1 | General electrical safety for medical devices | All electrically operated medical devices | Referenced; IS 7655 covers this for India |
| ISO 80601-2-12 | Critical care ventilators — specific requirements | ICU/critical care ventilators | IS 13450 Pt 2/Sec 12 covers this for India |
| IEC 60601-1-2 | Electromagnetic compatibility | All electrical medical devices | Required for EMC compliance evidence |
| ISO 14971 | Risk management for medical devices | All Class C device manufacturers | Required in technical dossier |
| ISO 13485:2016 | Quality Management System | All licensed manufacturers | Effectively required by CDSCO QMS review |
Who Needs a CDSCO Manufacturing License for Ventilators?
Domestic Manufacturers
Any company or individual that manufactures ventilators within India needs a manufacturing license. This applies to:
- Original equipment manufacturers (OEMs) making complete ventilators
- Assemblers combining imported sub-assemblies or components into a finished device
- Contract manufacturers producing ventilators on behalf of another brand (loan license arrangement under Form MD-4)
- R&D entities manufacturing prototypes for clinical investigation (Form MD-5 — test license applies)
Importers
Importers follow a completely separate licensing track. If you are bringing ventilators into India from overseas, you need an Import License under Form MD-14 (for the importer) or Form MD-15 (for the overseas manufacturer). Your Indian entity must have a registered place of business in India and appoint an Indian Authorized Agent. The manufacturing license (Form MD-3/MD-4/MD-9) does not apply to importers.
Traders, Distributors, and Retailers
Traders and distributors do not need a manufacturing license. However, they are legally required to ensure that every ventilator they supply or sell carries a valid CDSCO license number (either manufacturing or import license). Knowingly dealing in unlicensed medical devices is a violation of MDR 2017 and carries penalties.
Eligibility Criteria for the CDSCO Ventilator Manufacturing License
| Eligibility Criterion | Requirement |
| Legal Entity | Proprietorship, Partnership, LLP, Private Limited, or Public Limited company registered in India |
| Manufacturing Premises | Physical manufacturing facility located in India; must meet GMP requirements under Schedule III/V of MDR 2017 |
| Technical Person-in-Charge | At least one qualified technical staff member — degree in Engineering, Applied Sciences, or a relevant technical discipline; must be on the company's rolls |
| Quality Management System | A documented QMS aligned with ISO 13485:2016; CDSCO review effectively requires this level of documentation |
| Technical Documentation | Complete Design Dossier, Risk Management File (ISO 14971), Essential Principles checklist, test reports (IS 13450 Pt 2/Sec 12: 2023, IS 7655, IEC 60601-1-2), IFU, and labels |
| BIS Standard Compliance | Evidence of compliance with IS 13450 (Part 2/Sec 12): 2023 and IS 7655 via NABL-accredited or CDSCO-notified laboratory test reports |
| No Prior Disqualification | Applicant must not have been convicted under the Drugs and Cosmetics Act or have an existing license cancellation order |
Forms Required for CDSCO Ventilator Manufacturing License
| Form | Purpose | Who Uses It |
| Form MD-3 | Grant of license to manufacture medical devices for sale/distribution on own premises | Manufacturers with their own factory/production facility |
| Form MD-4 | Grant of Loan License — manufacturing on licensed premises of another entity | Entities using a contract manufacturing facility; brand owners without own plant |
| Form MD-5 | License to manufacture for clinical investigation/testing purposes only | R&D organisations; clinical trial prototype manufacturers |
| Form MD-6 | Renewal of manufacturing license before expiry | All existing licensed manufacturers approaching expiry date |
| Form MD-7 | Amendment to an existing manufacturing license | When premises, product range, technical person, or QMS changes occur |
| Form MD-9 | The actual manufacturing license granted by CDSCO | This is the document issued to you — not submitted by you |
Key decision: If you own and operate your manufacturing facility, use Form MD-3. If you are manufacturing at another licensed company's premises (contract/loan manufacturing), use Form MD-4. Selecting the wrong form wastes months and forces reapplication.
Documents Required for CDSCO Ventilator Manufacturing License
The document requirement for a Class C ventilator is extensive. This is non-negotiable — the device classification demands it. Prepare each category before you log in to the Sugam portal.
Company and Applicant Documents
- Certificate of Incorporation / Firm Registration / Proprietorship declaration
- PAN Card of the company or proprietor
- GST Registration Certificate
- Board Resolution authorizing the signatory (for companies)
- List of Directors / Partners / Proprietor with contact details
- Power of Attorney if filing through a regulatory consultant or agent
Manufacturing Premises and Site Documents
- Site Master File (SMF) of the manufacturing facility
- Ownership or lease deed / rental agreement for the manufacturing premises
- Scaled floor plan / layout showing production, quality control, storage, and utility areas
- Equipment list with make, model, serial numbers, and current calibration certificates
- HVAC qualification reports where cleanroom conditions are maintained
- Water quality test reports (if water is used in production or component cleaning)
- ETP / waste management compliance certificate as applicable
Technical and Product Documents
- Technical Specifications and Design Dossier for the ventilator (each model separately)
- Risk Management File as per ISO 14971 — hazard analysis, risk controls, residual risk evaluation
- Essential Principles of Safety and Performance checklist (Schedule I of MDR 2017)
- Test Reports from NABL-accredited or CDSCO-notified labs covering IS 13450 (Part 2/Sec 12): 2023, IS 7655, IEC 60601-1-2 (EMC), and ISO 80601-2-12 where applicable
- Biocompatibility assessment for patient-contact components (ISO 10993 series)
- Software validation documentation and cybersecurity assessment (for microprocessor / software-controlled ventilators)
- Clinical evaluation report or clinical data summary
- Instructions for Use (IFU) and User Manual (in English and relevant regional languages)
- Label draft as per Schedule VII of MDR 2017 — including UDI-ready labelling fields
Quality System Documents
- ISO 13485:2016 Certificate from an accredited certification body with valid expiry date
- Complete set of SOPs for manufacturing, QC, complaints, deviations, and recalls
- Batch Manufacturing Record (BMR) template for the ventilator
- Change Control, Non-Conformance, and CAPA SOPs
- Post-Market Surveillance and Vigilance Plan as per MDR 2017 Schedule VIII
Personnel Documents
- Educational qualification certificates of the Technical Person-in-Charge
- Experience letters and appointment letter of the Technical Person
- Company organogram showing QA, Production, Regulatory Affairs, and Service functions
- Training records for all manufacturing and quality personnel
Fee and Declaration Documents
- Government fee payment proof — online payment receipt from Sugam portal
- Signed undertaking and declaration as required under MDR 2017
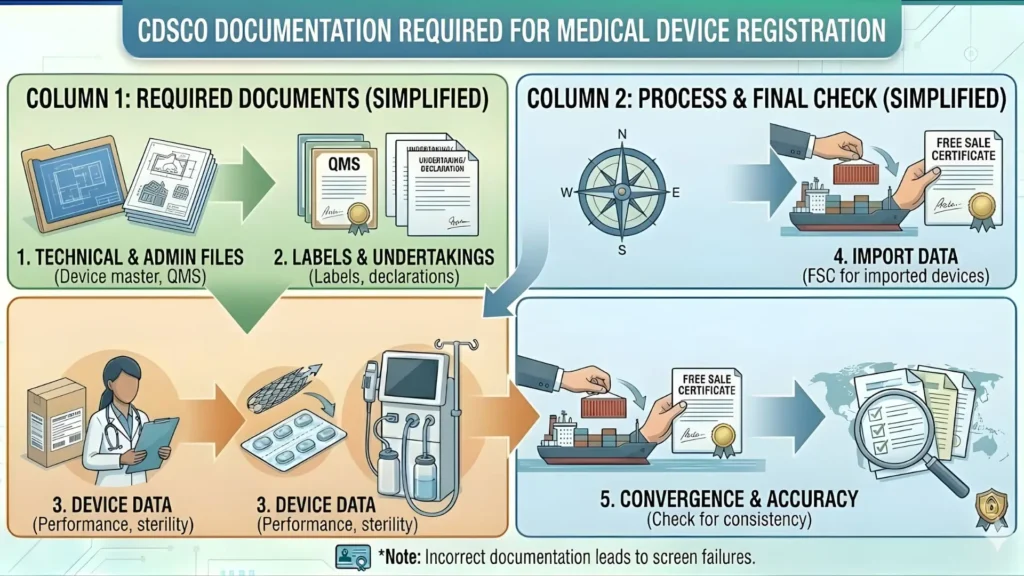

CDSCO Ventilator Manufacturing License: Complete Step-by-Step Process
Step 1: Register on the Sugam Portal
All CDSCO medical device applications are submitted exclusively through the Sugam portal at sugam.gov.in. Create your company account, complete email and mobile verification, and log in. Keep your company CIN or registration number ready. There is no offline application route for manufacturing licenses.
Step 2: Prepare Your Complete Technical Dossier
This is the step that separates successful applicants from those stuck in a loop of CDSCO queries. Before you touch the application form, compile every document listed in Section 8. CDSCO reviewers are thorough and document-specific. Missing test reports, expired ISO 13485 certificates, and incomplete risk management files are the three most common grounds for raising queries. Ensure test reports explicitly reference IS 13450 (Part 2/Sec 12): 2023 — this is what reviewers are checking in 2026.
Step 3: Pay the Application Fee Online
Application fees are paid online through the Sugam portal payment gateway at the time of application submission. Fees are non-refundable once paid. Verify the current fee schedule on cdsco.gov.in before payment, as fees may be revised by government notification.
Step 4: Submit Form MD-7 or Form MD-9 on Sugam
Complete the online application form. Attach all documents in the specified format and file size. Submit the application and immediately save the application reference number. You will need this number for every subsequent communication with CDSCO and for tracking on the portal.
Step 5: Acknowledgement and Initial Scrutiny
CDSCO issues an automated acknowledgement on submission. The application is then assigned to a reviewing officer at CDSCO, New Delhi for initial document scrutiny, typically within 15 to 30 working days. If documents are deficient or queries arise, CDSCO raises them on the Sugam portal and notifies you by email and SMS. Respond within the stipulated timeframe — failure to respond leads to application rejection.
Step 6: Mandatory Site Inspection
For all manufacturing licenses, a physical inspection of your facility is mandatory. CDSCO will schedule and conduct the inspection — either directly by CDSCO inspectors or through designated state inspectors acting on CDSCO's behalf. During inspection, they will assess:
- Physical layout, cleanliness, and environmental controls of the manufacturing area
- Equipment qualification status, calibration records, and maintenance logs
- QMS implementation — are your SOPs actually being followed, or just documented?
- Availability and competence of the Technical Person-in-Charge on the inspection date
- Storage conditions, material traceability, and labelling compliance
- Training records for all production and quality staff
If the inspection reveals non-conformances (NCs), CDSCO issues a formal Inspection Report listing each NC. You must submit a CAPA plan with corrective evidence for each NC within the timeframe specified. For serious NCs, a follow-up re-inspection may be required.
Step 7: CDSCO Technical Review and Approval Decision
Once all queries are closed and the site inspection is satisfactory, CDSCO places the application before the relevant reviewing authority or committee. They evaluate the complete technical dossier, QMS evidence, and inspection outcome. Three outcomes are possible: full approval, conditional approval requiring additional data, or formal rejection with stated reasons.
Step 8: License Grant — Form MD-9
On approval, CDSCO issues the manufacturing license in Form MD-9, which is available for download on the Sugam portal. The MD-9 specifies the licensed product(s) by name and model, the manufacturing premises address, the license validity period, and any conditions of license. This is your operational document — keep it current.

Government Fee Structure for CDSCO Ventilator Manufacturing License
| Application Type | Form | Fee (INR) |
| Grant of manufacturing license (own premises) | Form MD-3 | ₹50,000 |
| Grant of Loan License (contract manufacturing) | Form MD-4 | ₹25,000 |
| Renewal of manufacturing license | Form MD-6 | ₹25,000 |
| Amendment to existing license | Form MD-7 | ₹10,000 |
| Duplicate license | — | ₹5,000 |
| Test/investigation license only | Form MD-5 | ₹15,000 |
Always verify the current fee schedule at cdsco.gov.in before paying, as fees are subject to revision by government notification. Fees once paid through the Sugam portal are non-refundable regardless of application outcome.
License Validity and Renewal Timeline
Manufacturing licenses under MDR 2017 are valid for 5 years from the date of grant unless suspended or cancelled earlier by CDSCO.
| Stage | Action Required | Timeline |
| 90 days before expiry | Begin preparing renewal dossier — update QMS documents, verify ISO 13485 certificate validity, generate updated IS 13450 (Part 2/Sec 12): 2023 compliance evidence if required | Recommended start |
| 60 days before expiry | Submit Form MD-6 with renewal fee on Sugam portal | Ideal filing deadline |
| 30 days before expiry | Absolute last safe date for submission to avoid inspection scheduling risk | Minimum deadline |
| After expiry (with active renewal application) | Continued manufacturing may be permitted at CDSCO discretion pending renewal processing | Subject to CDSCO discretion |
| After expiry (without application) | Manufacturing must cease immediately — constitutes unlicensed activity | Zero tolerance; enforcement action likely |
Even a single day of manufacturing with an expired license — without an active renewal application on record — constitutes a violation of MDR 2017. CDSCO tracks license expiry and can initiate enforcement action without prior notice.
Post-License Compliance Obligations for Ventilator Manufacturers
Receiving the Form MD-9 is not the end of the regulatory journey — it is the beginning of ongoing obligations. CDSCO expects full compliance with the following requirements throughout the license period:
Annual Returns
All licensed manufacturers must file annual returns on the Sugam portal, declaring the quantity and value of ventilators manufactured and supplied during the year. Late or non-filing invites scrutiny.
Post-Market Surveillance (PMS) and Vigilance Reporting
Ventilator manufacturers must maintain an active Post-Market Surveillance system. Any serious adverse event (SAE), device malfunction causing or capable of causing patient harm, or field safety corrective action (FSCA) must be reported to CDSCO within prescribed timelines. The 2025-2026 updates have tightened these timelines for Class C and D life-support devices.
Complaint Handling and Product Recall
A documented complaint management system and product recall procedure must be in place, tested, and ready for execution. Any recall must be notified to CDSCO immediately. Post-recall reports are also required.
GMP Compliance and Unannounced Inspections
CDSCO can and does conduct periodic GMP inspections without advance notice — particularly for life-support device manufacturers. Maintain your facility, SOPs, batch records, and personnel training records in inspection-ready condition at all times. The 2026 guidance has increased the stated frequency of unannounced inspections for Class C manufacturers.
License Amendments (Form MD-7)
Any change to your manufacturing premises, licensed product range, technical personnel, QMS scope, or company details must be formally reported to CDSCO via Form MD-7 before the change is implemented. Implementing unapproved changes — even minor ones — without an amendment is a violation.
UDI Readiness
India is implementing a Unique Device Identification (UDI) framework for high-risk medical devices. Ventilator manufacturers should begin aligning their device labelling, production records, and supply chain traceability systems with UDI requirements now, ahead of mandatory enforcement.
Latest CDSCO Regulatory Updates for Ventilator Manufacturers (2025–2026)
| Update | Details | Impact on Manufacturers |
| IS 13450 (Part 2/Sec 12): 2023 alignment | CDSCO reviewers now expect test reports aligned to this updated BIS standard in new applications and renewals from 2024 onward | Obtain updated test reports from NABL labs covering this standard explicitly |
| Tightened PMSR timelines | Post-market surveillance and vigilance reporting obligations strengthened for Class C and D life-support devices | Review and update your vigilance SOPs; ensure timely incident reporting |
| Increased unannounced GMP inspections | CDSCO has increased frequency of unannounced inspections for life-support device manufacturers | Maintain facility and documentation in permanent inspection-ready condition |
| UDI framework implementation | India moving toward mandatory UDI for high-risk devices; labelling and traceability requirements being phased in | Begin UDI-compatible labelling and product identification practices now |
| CDSCO-GeM integration | Active CDSCO license now a verified eligibility criterion on the Government e-Marketplace portal | Ensure your license is active and correctly reflected on GeM to participate in tenders |
| Sugam portal enhancements | Improved document upload, faster acknowledgements, and online query-response functionality upgraded | Use updated portal features; check Sugam announcements regularly |
| BIS IS 7655 and IS 13450 dual compliance emphasis | CDSCO technical review increasingly expects both standards to be explicitly addressed in the test dossier | Do not rely on one standard alone; obtain reports covering both |
Total Cost Estimation for CDSCO Ventilator Manufacturing License
| Cost Head | Estimated Range (INR) | Notes |
| Government Application Fee (Form MD-3) | ₹50,000 | Non-refundable; payable online on Sugam |
| Testing Charges — IS 13450 (Pt 2/Sec 12): 2023, IS 7655, IEC 60601-1-2 | ₹3,00,000 – ₹8,00,000 | NABL-accredited or CDSCO-notified laboratory; costs vary by number of models and test scope |
| ISO 13485:2016 Certification | ₹1,50,000 – ₹4,00,000 | First-time certification from an accredited body; annual surveillance audit costs additional |
| Technical Documentation and Design Dossier Preparation | ₹1,00,000 – ₹3,00,000 | Depends on in-house capability; outsourcing to a technical writer increases cost |
| Regulatory Consultant Fees | ₹1,50,000 – ₹5,00,000 | Strongly recommended for first-time applicants; can significantly reduce query cycles |
| GMP Facility Upgrades | ₹5,00,000 – ₹50,00,000+ | Highly variable; depends on existing facility condition and gap assessment findings |
| BIS IS 7655 / IS 13450 Registration (voluntary) | ₹50,000 – ₹1,50,000 | Not mandatory for CDSCO license; recommended for government tender eligibility |
| Total Estimated Investment | ₹12,00,000 – ₹70,00,000+ | Range is wide because facility upgrade costs dominate; a purpose-built facility changes the economics significantly |
Conclusion
Getting a CDSCO manufacturing license for ventilators is rigorous — deliberately so. Ventilators are machines that breathe for people who cannot breathe for themselves. The regulatory framework is not bureaucratic obstruction; it is a life-safety system built around the principle that a failed ventilator is not a product liability matter — it is a preventable death.
The manufacturers who succeed in this process — and who sustain compliance over the long term — are not those who found shortcuts. They are the ones who invested in genuine GMP infrastructure, proper test documentation covering IS 13450 (Part 2/Sec 12): 2023 and IS 7655, a real ISO 13485 QMS, and a qualified regulatory team. These same investments are what make their products trusted in clinical settings and competitive in government procurement.
If you are beginning this journey, the most practical first steps are: conduct a GMP gap assessment of your facility, commission testing at an NABL-accredited laboratory covering both IS 13450 (Part 2/Sec 12): 2023 and IS 7655, obtain your ISO 13485 certification, and prepare your complete technical dossier before you open the Sugam portal. Once all of that is genuinely ready, the application process itself is straightforward.
Stay current. CDSCO issues circulars, guidance updates, and standard operating procedure revisions regularly on cdsco.gov.in and the Sugam portal. Regulatory compliance in India's medical device space is a continuous obligation, not a one-time event.
Frequently Asked Questions
Is a CDSCO license required if I am manufacturing ventilators purely for export?
Yes. If manufacturing is carried out in India, a CDSCO manufacturing license is required regardless of whether the product is intended for the domestic market or export. Rule 9 of MDR 2017 does not provide an export exemption for manufacturing.
What is the difference between IS 7655 and IS 13450 (Part 2/Sec 12): 2023?
IS 7655 is the earlier BIS standard for lung ventilators, primarily aligned to IEC 60601-1 for electrical safety. IS 13450 (Part 2/Sec 12): 2023 is the updated and more comprehensive BIS standard, aligned to ISO 80601-2-12, covering ventilator-specific performance requirements including alarm systems, tidal volume accuracy, PEEP delivery, and software validation.
Can a sole proprietor apply for a ventilator manufacturing license?
Yes, a sole proprietor can legally apply. However, they must independently meet all eligibility requirements — qualified technical person-in-charge, GMP-compliant manufacturing premises, complete technical documentation, and accredited test reports.
How long does the entire CDSCO licensing process realistically take?
For a well-prepared application with complete documentation and a GMP-ready facility that passes inspection without major non-conformances, the process typically takes 45 to 90 working days.
Is ISO 13485 certification mandatory?
ISO 13485 is not explicitly stated as a pre-condition in MDR 2017 for obtaining a manufacturing license. However, CDSCO's technical review for Class C devices effectively requires QMS documentation of a depth and rigour that ISO 13485 certification represents.
Can multiple ventilator models be covered under one license?
Yes. Multiple models and variants can be listed under a single Form MD-9 license. However, each model must have its own technical specifications, design dossier, and test reports submitted at the time of application or added via a license amendment (Form MD-7).
Do overseas test lab reports get accepted by CDSCO?
CDSCO generally requires test reports from NABL-accredited or CDSCO-notified testing laboratories in India. Reports from internationally accredited laboratories that are members of the IEC CB scheme may be considered with supporting documentation, but this should be confirmed with the CDSCO reviewing officer before relying solely on overseas reports. The safest approach is Indian NABL lab testing.
What happens if my license expires without renewal?
Manufacturing must cease on the date of expiry if no renewal application is pending. Operating beyond expiry without a filed renewal is a violation of MDR 2017, can result in enforcement action including seizure of goods and cancellation of other licenses, and can harm your eligibility for future applications.
Is BIS product registration under IS 13450 separately required?
For CDSCO licensing purposes, BIS product registration is not a separately mandatory pre-condition. Compliance with IS 13450 (Part 2/Sec 12): 2023 demonstrated through accredited test reports is what the CDSCO dossier requires.
What is the difference between Form MD-3 and Form MD-4?
Form MD-3 is for manufacturers who own and operate their own manufacturing premises. Form MD-4, the Loan License, is for entities that manufacture on the licensed premises of another manufacturer — a contract manufacturing or toll manufacturing arrangement.





































