- Class A Medical Device Registration list — every major product category that CDSCO classifies as Class A under India's Medical Devices Rules 2017
- Registration roadmap — exact steps, forms, timelines, and fees to legally register a Class A device with CDSCO in 2026
- Class A vs Class B vs Class C vs Class D — clear comparison so you know exactly where your device sits in India's risk classification framework
- Compliance checklist — mandatory documents, labelling requirements, and post-market obligations for Class A device manufacturers and importers
Why Class A Medical Device Registration Classification Matters for Indian Manufacturers and Importers
If you manufacture or import medical devices in India, the classification your product receives from the CDSCO (Central Drugs Standard Control Organization) directly determines your regulatory workload, your registration timeline, and your legal obligations from the moment you enter the Indian market.


India's medical device regulatory framework is governed by the Medical Devices Rules, 2017 (MDR 2017), notified under the Drugs and Cosmetics Act, 1940. Under this framework, every medical device sold, manufactured, or imported in India is assigned to one of four risk-based classification categories — Class A, Class B, Class C, or Class D — where Class A represents the lowest-risk devices and Class D represents the highest-risk devices.
For anyone dealing with Class A devices, this classification carries a specific set of obligations that are lighter than higher-risk classes but are still legally mandatory. Getting this wrong — either by under-classifying a higher-risk device as Class A, or by missing the registration and compliance steps specific to Class A — can result in import rejections, market withdrawal notices, and legal penalties under Indian law.
India's Medical Device Regulatory Framework: CDSCO and MDR 2017 Explained
Before diving into Class A specifically, it helps to understand the overall regulatory structure. India's medical device regulation has gone through significant transformation in the past decade, moving from a small list of notified devices to a comprehensive, risk-based framework covering all medical devices.
| Regulatory Element | Detail |
| Governing Law | Drugs and Cosmetics Act, 1940 (as amended) |
| Specific Rules | Medical Devices Rules, 2017 (MDR 2017) |
| Regulating Authority | CDSCO — Central Drugs Standard Control Organization under MOHFW |
| Online Portal | SUGAM portal — sugam.gov.in |
| Classification Framework | Four risk-based classes: Class A, B, C, D |
| Applicable Standard | Aligned with GHTF (Global Harmonization Task Force) principles |
| Phase-in Timeline | All medical devices brought under MDR 2017 in phased manner; most now covered |
| 2026 Status | Full regulatory coverage active; post-market surveillance obligations enforced |
India's approach to medical device classification follows the internationally recognized GHTF model, which bases classification on the intended use of the device, the potential risks it poses to the patient or user, the duration of use, and whether the device is invasive, active, implantable, or diagnostic in nature.
India's Four-Tier Medical Device Classification System: Class A to Class D
Understanding where Class A sits within the full classification spectrum helps manufacturers and importers make the right compliance decisions from the start.
| Class | Risk Level | Device Examples | Regulatory Pathway |
| Class A | Low Risk | Bandages, tongue depressors, stethoscopes, examination gloves, wheelchairs, hospital furniture | Self-certification by manufacturer + CDSCO registration (MD 1 / MD 3 form) |
| Class B | Low-Moderate Risk | Hypodermic needles, surgical instruments, suction equipment, hearing aids | CDSCO registration with additional technical documentation |
| Class C | Moderate-High Risk | Lung ventilators, bone fixation plates, haemodialysis machines, blood glucose monitors | Full CDSCO licence with performance testing and clinical data |
| Class D | High Risk | Heart valves, coronary stents, active implantable devices, HIV diagnostics | Most stringent — requires full technical dossier, clinical evaluation, notified body involvement |
Class A devices are the entry point into India's regulated medical device market. They are characterized by non-invasive use, no contact with broken skin or sterile pathways, short contact duration with the body, and no direct dependence of life on their function. However, 'low risk' does not mean 'unregulated.' Every Class A device still requires formal CDSCO registration before it can be legally manufactured, sold, or imported in India.
What is a Class A Medical Device Registration? CDSCO Definition and Risk Criteria
Under Schedule III of India's Medical Devices Rules 2017, a Class A medical device is one that presents low individual risk and low public health risk. The classification rules apply a combination of factors to determine where a device falls — and a device must meet all applicable criteria to be correctly classified as Class A.
CDSCO Classification Factors That Define Class A:
- Intended use is non-invasive — the device does not penetrate the body through a natural body orifice or through the skin
- Contact with intact skin only — it does not come into contact with wounded tissue, mucous membranes, or compromised skin
- Duration is transient or short-term — contact with the patient is brief and not intended to be permanent or long-term
- No energy supply dependency — the device does not require an external power source to perform its primary intended function (or if it does, the function is not life-sustaining)
- Non-sterile supply — the device is generally not required to be supplied in a sterile condition (Class A sterile devices have additional requirements)
- No measuring function with critical precision — the device does not perform a measurement where incorrect precision could cause harm
There is one important sub-classification within Class A that every manufacturer and importer must be aware of: the distinction between Class A standard and Class A sterile.
| Sub-Class | Definition | Additional Requirement | Example Devices |
| Class A (Non-Sterile) | Low-risk device not intended to be supplied in sterile condition | Standard CDSCO registration — MD 1 for manufacturers, MD 3 for importers | Tongue depressors, bandages, hospital furniture, walking aids, stethoscopes |
| Class A (Sterile) | Low-risk device that is supplied in sterile condition — the sterility is a critical characteristic | QMS (Quality Management System) audit required in addition to standard registration; manufacturing site must maintain ISO 13485 or equivalent | Sterile examination gloves, sterile wound dressings, sterile irrigation sets |
Complete Class A Medical Device List Under CDSCO India (2026)
Below is the comprehensive product list covering all major categories and individual devices that fall under Class A classification in India under MDR 2017. This list is aligned with Schedule III classification rules and CDSCO's published guidance. If you are unsure whether a specific device falls under Class A, always seek a formal opinion from a CDSCO-registered regulatory consultant.
1. Wound Care and Dressings
| Product Name | Intended Use | Class A Basis |
| Adhesive bandages (plasters) | Cover minor cuts and abrasions on intact skin | Non-invasive, intact skin contact, no energy source |
| Gauze swabs and dressings (non-sterile) | Wound coverage and absorption | Non-invasive, external use only |
| Sterile gauze dressings | Sterile wound coverage | Class A Sterile — QMS requirement applies |
| Elastic bandages / crepe bandages | Compression and support for musculoskeletal conditions | Non-invasive, external contact only |
| Cotton wool / absorbent cotton | General wound cleaning and absorption | Non-invasive, non-sterile external use |
| Wound closure strips (Steri-strips type) | Approximating wound edges on intact or minor broken skin | Non-invasive, external only |
| Foam dressings (non-medicated, non-sterile) | Absorption and protection for superficial wounds | Non-invasive, external use |
| Alginate dressings (non-active) | Exudate management for surface wounds | Non-invasive, no active ingredient |
| Hydrocolloid dressings (non-medicated) | Moisture-retentive wound environment | Non-invasive, non-sterile, external use |
| Tubular bandages and retention nets | Retention of dressings on body surface | Non-invasive, no direct therapeutic function |
2. Examination and Diagnostic Aids (Non-Measuring)
| Product Name | Intended Use | Class A Basis |
| Stethoscope (acoustic) | Auscultation of body sounds by healthcare professional | Non-invasive, no energy source, no measuring function |
| Tongue depressors (wooden or plastic) | Examination of oral cavity | Non-invasive, transient contact |
| Reflex hammers | Clinical neurological reflex testing | Non-invasive, mechanical only |
| Penlight / clinical examination torch | Visual examination of body orifices | Non-invasive, no therapeutic function |
| Medical loupes (non-powered) | Magnification during clinical examination | Non-invasive, no energy function |
| Otoscope (non-powered, non-illuminated head only) | Examination of ear canal | Non-invasive — note: illuminated electronic versions may be Class B |
| Nasal speculum | Examination of nasal passages | Non-invasive, non-sterile, reusable examination tool |
| Vaginal speculum (reusable, non-sterile) | Gynaecological examination | Non-invasive when used as examination-only instrument |
3. Protective and Barrier Devices
| Product Name | Intended Use | Class A Basis |
| Non-sterile examination gloves (latex / nitrile / vinyl) | Barrier protection during clinical examination | Non-invasive, non-sterile, short-term contact |
| Sterile surgical gloves | Sterile barrier during surgical procedures | Class A Sterile — additional QMS requirement |
| Surgical masks (non-respirator type) | Barrier against droplet contamination | Non-invasive, external protective use |
| Surgical caps and shoe covers | Contamination control in clinical environments | Non-invasive, no patient contact function |
| Medical gowns and drapes (non-sterile) | Staff and patient surface protection | Non-invasive, barrier use only |
| Sterile surgical drapes | Sterile field creation during surgery | Class A Sterile — manufacturing site QMS required |
| Medical face shields | Eye and face splash protection | Non-invasive, external only |
4. Hospital and Patient Care Furniture and Equipment
| Product Name | Intended Use | Class A Basis |
| Hospital beds (manual, non-powered) | Patient positioning and rest during care | Non-invasive, no therapeutic function, no active energy |
| Examination tables / couches | Patient examination and minor procedures | Non-invasive, no active function |
| Hospital trolleys and stretchers (non-motorised) | Patient transport within healthcare facilities | Non-invasive, no therapeutic function |
| Bedpans and urinals | Excretion assistance for bed-bound patients | Non-invasive, no therapeutic function |
| IV stands and drip stands | Support for infusion bags during IV therapy | Non-invasive, no active function — note: IV fluid itself is a drug |
| Bed rails and side guards | Patient fall prevention on hospital beds | Non-invasive, passive safety device |
| Medical waste bins and sharps containers | Segregation and safe disposal of clinical waste | Non-invasive, no patient therapeutic function |
| Medical trolleys and instrument trays | Organisation and transport of surgical instruments | Non-invasive, no therapeutic function |
5. Mobility, Rehabilitation and Assistive Devices
| Product Name | Intended Use | Class A Basis |
| Manual wheelchairs (non-powered) | Mobility assistance for persons with movement limitations | Non-invasive, no active energy source, low individual risk |
| Crutches (axillary and forearm) | Walking aid following injury or surgery | Non-invasive, mechanical support only |
| Walking sticks and canes | Ambulatory support aid | Non-invasive, passive assistive device |
| Zimmer frames / walking frames | Mobility and balance support | Non-invasive, no energy source |
| Cervical collars (soft foam type) | Neck support and mild immobilisation post-injury | Non-invasive, external support, no active function |
| Arm slings and shoulder immobilisers | Post-injury limb support | Non-invasive, external supportive use |
| Orthotic shoe insoles (non-custom, non-prescription) | Arch support and foot pressure distribution | Non-invasive, low-risk external contact |
| Anti-decubitus cushions (static, non-powered) | Pressure redistribution to prevent pressure ulcers | Non-invasive, passive mechanical function |
| Patient hoists (non-powered, manual) | Transfer assistance for patients with limited mobility | Non-invasive, no active energy source |
6. Sterilization, Disinfection and Hygiene Products
| Product Name | Intended Use | Class A Basis |
| Sterilization pouches and bags (medical grade) | Packaging of instruments for sterilisation | Non-invasive, no therapeutic contact with patient |
| Sterilisation indicators (chemical indicator strips) | Verification of sterilisation cycle completion | Non-invasive, monitoring function only |
| Autoclave tape | Visual indicator of steam sterilisation exposure | Non-invasive, no patient contact |
| Medical-grade cleaning brushes for instruments | Decontamination of reusable surgical instruments | Non-invasive, no direct patient function |
| Instrument trays and baskets (sterilisable) | Holding instruments during sterilisation cycles | Non-invasive, no therapeutic function |
7. Ophthalmic Devices (Non-Corrective, Non-Invasive)
| Product Name | Intended Use | Class A Basis |
| Ophthalmic trial lens sets (non-powered) | Vision testing and refraction assessment | Non-invasive, no energy source |
| Trial frames (ophthalmic) | Holding trial lenses during eye examination | Non-invasive, non-therapeutic contact |
| Eye patch (non-sterile) | Occlusion therapy or protection of eye area | Non-invasive, external contact with intact periorbital skin |
| Colour vision test charts (Ishihara plates) | Screening for colour vision deficiency | Non-invasive, no physical patient contact — diagnostic aid only |
8. Dental Devices (Non-Invasive and Non-Active)
| Product Name | Intended Use | Class A Basis |
| Dental mouth mirrors (non-powered) | Visual examination of oral cavity | Non-invasive, examination aid only |
| Dental probes and explorers | Tactile examination of tooth surfaces | Non-invasive, diagnostic use only |
| Dental impression trays (non-custom) | Holding impression material during dental mould-taking | Non-invasive, no active function |
| Dental bibs / patient drapes | Patient protection during dental procedures | Non-invasive, protective barrier use |
| Dental articulating paper | Identifying occlusal contact points | Non-invasive, no therapeutic function |
9. General Single-Use and Disposable Devices (Non-Sterile)
| Product Name | Intended Use | Class A Basis |
| Specimen collection containers (urine pots, stool containers) | Collection of diagnostic specimens from patients | Non-invasive, external collection — patient does not insert the device |
| Spatulas (non-sterile, wooden or plastic) | General sampling and mixing in clinical settings | Non-invasive, transient contact use |
| Cotton buds for clinical use (non-sterile) | Surface swabbing and wound cleaning | Non-invasive, non-sterile external use |
| Disposable aprons (clinical use) | Staff protection from contamination during patient care | Non-invasive, no patient contact function |
| Medical adhesive tape (non-medicated) | Securing dressings and medical tubing to skin surface | Non-invasive, intact skin contact, no active ingredient |
10. Measuring Devices (Non-Critical Precision)
Note: Measuring devices in this category are classified Class A only where inaccurate measurement would not cause direct patient harm. Devices where measurement inaccuracy poses clinical risk (e.g., blood glucose meters, blood pressure monitors) are classified Class B or above.
| Product Name | Intended Use | Class A Basis |
| Non-invasive body measurement tapes (clinical) | Measuring limb circumference, wound dimensions | Non-invasive, no critical precision requirement |
| Height measurement scales (stadiometers) | Measurement of patient height | Non-invasive, no risk from measurement variation |
| Medical weighing scales (mechanical, non-Class III) | General body weight measurement | Non-invasive — Class A where not used for drug dosage calculation |
| Timing devices for clinical use (general purpose) | Timing procedures, respiration rates | Non-invasive, non-critical measurement function |
How to Register a Class A Medical Device with CDSCO: Step-by-Step Process (2026)
Every Class A medical device that is manufactured in India or imported into India must be registered with CDSCO before it can enter the market. There is no exemption for Class A devices — the registration requirement is mandatory across all four classes under MDR 2017.
The specific form you use depends on whether you are a domestic manufacturer or an importer:
- Form MD 1: Used by domestic manufacturers of Class A and Class B medical devices
- Form MD 3: Used by importers of Class A and Class B medical devices
Step 1: Determine Your Device Classification Accurately
Before applying, confirm that your device genuinely falls under Class A. If there is any ambiguity — for example, a device with both non-invasive and minimally invasive use cases, or a device that contacts mucous membranes — seek a pre-submission consultation with a CDSCO-registered regulatory affairs consultant. Incorrect classification is one of the most common grounds for rejection and can require you to restart the entire application process.
Step 2: Register on the SUGAM Portal
All CDSCO medical device applications are submitted online through the SUGAM portal at sugam.gov.in. Create an account for your entity — company, LLP, or individual — and verify your login credentials. Ensure your Import Export Code (IEC) from DGFT and GST registration are current before beginning the application.
Step 3: Prepare and Compile Mandatory Documents
Document completeness is critical. Incomplete applications are returned and restart the clock on your timeline. The mandatory documents for Class A registration differ slightly between manufacturers and importers:
| Document Required | Domestic Manufacturer (MD 1) | Importer (MD 3) |
| Completed application form | Form MD 1 | Form MD 3 |
| Device description and intended use statement | Required | Required |
| Device classification rationale (with Schedule III reference) | Required | Required |
| Declaration of conformity to IS / applicable Indian standards | Required | Required |
| Manufacturing site details and address proof | Required | Not applicable |
| Foreign manufacturer's authorisation / Power of Attorney | Not applicable | Required — must be notarised and apostilled |
| Certificate of Free Sale (CFS) from country of origin | Not applicable | Required — issued by competent authority in manufacturer's country |
| Foreign manufacturer's ISO 13485 or equivalent QMS certificate | Not applicable | Required |
| Label draft compliant with MDR 2017 Schedule V requirements | Required | Required |
| IEC (Import Export Code) from DGFT | Not applicable | Required |
| GST registration certificate | Required | Required |
| Undertaking for post-market surveillance compliance | Required | Required |

Step 4: Submit Application and Pay Fee on SUGAM
Upload all documents in the formats specified by the SUGAM portal (typically PDF for certificates and declarations). Pay the prescribed application fee online. As of current CDSCO schedules, the registration fee for Class A devices is lower than higher-risk classes — but always verify the current fee schedule on the SUGAM portal before submitting, as fees may be revised.
Step 5: CDSCO Review and Grant of Registration
CDSCO will review the submitted application. For Class A devices with complete documentation, the registration process typically takes 30 to 60 working days. If CDSCO raises queries or requests additional information, responding promptly and completely is essential to avoid further delays.
| Application Stage | Typical Timeline | Action Required |
| Document submission on SUGAM | Day 1 | Complete form + upload all documents + pay fee |
| Acknowledgement and application number issued | Within 2 to 5 working days | Save acknowledgement for tracking |
| CDSCO scrutiny and query (if raised) | 15 to 30 working days | Respond to all queries within the stipulated period |
| Registration certificate issued | 30 to 60 working days (from complete submission) | Download certificate from SUGAM portal |
| Registration validity | 5 years (for most Class A devices) | File renewal application before expiry |
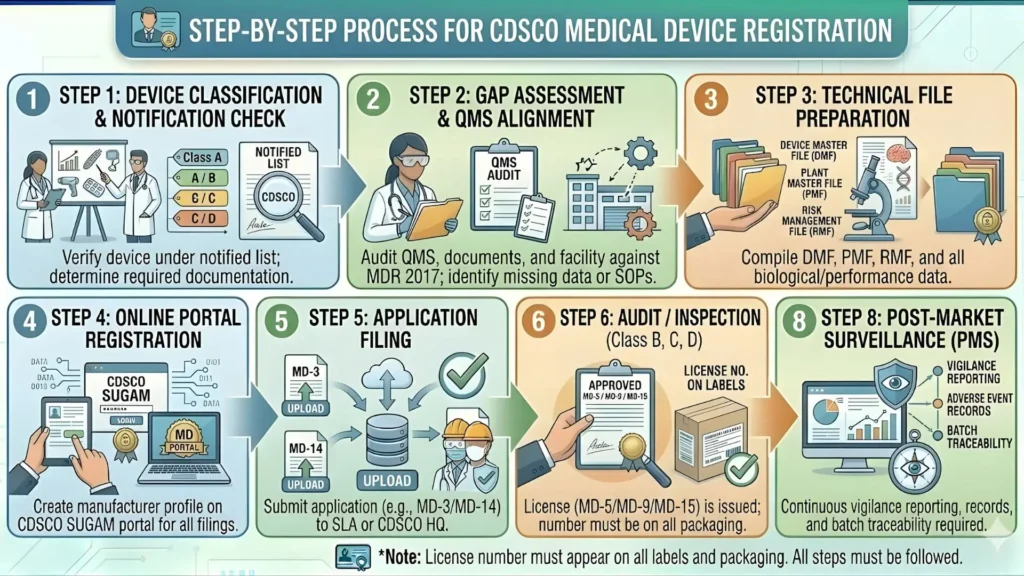

Class A Medical Device Labelling Requirements Under MDR 2017
Every Class A medical device sold in India must carry a label that complies with Schedule V of the Medical Devices Rules 2017. Labelling non-compliance is a significant source of regulatory action even for low-risk devices. Your label must include all of the following:
- Name of the medical device — as it appears in the registration certificate
- Name and address of the manufacturer — full legal name and country of manufacture
- Name and address of the Indian importer — mandatory for all imported devices
- Unique Device Identifier (UDI) — as per CDSCO UDI implementation timeline
- Batch or lot number — for traceability purposes
- Date of manufacture — month and year at minimum
- Expiry date or shelf life — where applicable to the device type
- Intended use statement — brief, accurate description of the device's medical purpose
- Instructions for use or reference to Instructions for Use (IFU) document — if full IFU cannot fit on the label
- Storage conditions — temperature range, humidity, light sensitivity where relevant
- Single-use symbol (if applicable) — internationally recognized symbol \u2613 for single-use devices
- Sterile symbol (if applicable) — for Class A Sterile devices, the sterile packaging indicator
- CDSCO registration number — must appear on the label once registration is granted
For imported Class A devices, labels must be in English. If the original foreign label does not meet all Indian requirements, a sticker overlay or re-labelling process must be completed before the product enters the retail or clinical distribution chain.
Post-Market Surveillance and Ongoing Compliance for Class A Devices
Registration does not end your compliance obligations. Under MDR 2017, all registered medical device holders — including Class A — must maintain active post-market surveillance. While Class A requirements are less intensive than Class C or D, they are still legally binding.
| Obligation | Class A Requirement | Consequences of Non-Compliance |
| Periodic Safety Update Reports (PSUR) | Required — frequency as per CDSCO guidance | Registration suspension or cancellation |
| Vigilance Reporting (adverse events) | All serious incidents must be reported to CDSCO within prescribed timelines | Legal action under DCA 1940 |
| Field Safety Corrective Action (FSCA) | Recalls, field corrections, or safety notices must be reported and coordinated with CDSCO | Market withdrawal and penalties |
| Device Modifications | Any change to intended use, design, or materials may require fresh registration or variation application | Selling unregistered variant is a regulatory violation |
| Record Keeping | Maintain distribution records for traceability — minimum 5 years for Class A | Inability to support recall or investigation |
| Registration Renewal | Apply for renewal before the 5-year registration expiry | Lapsed registration means product cannot be legally sold |
Class A vs Class B Medical Devices: Key Differences Every Indian Manufacturer Must Know
The distinction between Class A and Class B is one of the most common points of confusion in Indian medical device regulation. Both use the same application forms (MD 1 for manufacture, MD 3 for import), but the documentary requirements and scrutiny levels differ. Getting the classification wrong — especially classifying a Class B device as Class A — is a serious regulatory risk.
| Comparison Parameter | Class A | Class B |
| Risk Level | Low risk | Low-to-moderate risk |
| Invasiveness | Non-invasive only | May be minimally invasive |
| Body Contact | Intact skin or no body contact | May contact mucous membranes or internal body surfaces |
| Sterility Requirement | Generally non-sterile (exception: Class A Sterile sub-class) | More devices are sterile-supplied |
| Examples | Bandages, stethoscopes, wheelchairs, tongue depressors | Hypodermic needles, suction catheters, surgical instruments, hearing aids |
| QMS Requirement | Required for Class A Sterile only | Required for all Class B devices |
| Technical Documentation Depth | Standard — device description, intended use, conformity declaration | More detailed — including biocompatibility data where relevant |
| Application Form | MD 1 (manufacture) / MD 3 (import) | MD 1 (manufacture) / MD 3 (import) |
| Typical Registration Timeline | 30 to 60 working days | 45 to 90 working days |
The single most practical rule of thumb: if your device ever enters a body orifice, contacts non-intact skin, is implanted even temporarily, or directly sustains a physiological function, it almost certainly does not belong in Class A. When in doubt, classify higher and discuss with a regulatory consultant — reclassifying downward later is easier than being caught with an under-classified device.
Common Mistakes in Class A Medical Device Registration (And How to Avoid Them)
After reviewing hundreds of Class A device applications, regulatory professionals in India consistently see the same errors. Here are the most common mistakes and how to avoid them:
- Misclassifying a Class B device as Class A: The most consequential error. A hypodermic needle, for example, is Class B not Class A, despite appearing straightforward. Always reference Schedule III of MDR 2017 before assuming Class A. If the device contacts non-intact tissue, a natural orifice, or the bloodstream at any stage of use, it is not Class A.
- Submitting incomplete labelling drafts: CDSCO frequently returns applications because the label draft does not include all Schedule V mandatory elements. Draft your label before starting the application — do not leave it as an afterthought.
- Power of Attorney not apostilled: For imported Class A devices, the manufacturer's authorisation document must be notarised in the country of origin and apostilled (or legalised for non-Hague Convention countries). A simple notarised copy is insufficient.
- Using an outdated Certificate of Free Sale: The CFS must be current — typically issued within 12 months of the application date. An expired CFS will result in rejection.
- Failing to account for Class A Sterile requirements: Importers of sterile Class A devices sometimes submit a standard MD 3 without providing the manufacturer's ISO 13485 QMS certification. CDSCO requires QMS evidence for Class A Sterile devices specifically.
- Not updating registration after product modifications: Any change to the device name, intended use, material composition, design, or manufacturer details requires a variation application. Continuing to sell the original registered product after significant modification without notifying CDSCO is a regulatory violation.
Conclusion: Getting Class A Medical Device Compliance Right in India
Class A may be the lowest-risk tier of India's medical device classification system, but 'low risk' in the regulatory sense is not the same as 'low stakes' in the commercial sense. An unregistered Class A device cannot legally enter the Indian market regardless of how safe or well-established the product is internationally.
The good news is that for most Class A devices with complete documentation, the CDSCO registration pathway is straightforward. Korean, European, US, and Indian manufacturers alike have navigated it successfully thousands of times. The key is approaching it methodically — correct classification first, complete documentation second, SUGAM submission third — and not underestimating the detail that goes into labelling and post-market obligations.
India's medical device market continues to grow rapidly in 2026, driven by expanding healthcare infrastructure, rising domestic manufacturing capability, and increasing regulatory sophistication. Building a clean, compliant Class A portfolio today — with properly registered devices, correctly labelled products, and maintained post-market surveillance records — is the foundation for long-term market success.
Frequently Asked Questions
Is registration mandatory for all Class A devices in India?
Yes, without exception. Under MDR 2017, every Class A device must be registered with CDSCO before it can be manufactured, imported, or sold in India. "Low risk" does not mean unregulated — there are no blanket exemptions. Always verify the current compliance status for your device category on the CDSCO website before entering the market.
Does one MD 3 registration cover multiple product variants?
Not automatically. Each distinct product generally needs its own MD 3 registration. Certain size variations of the same device may be grouped under one registration, but this is decided case by case by CDSCO. If you have a product range, get regulatory advice before assuming a single registration covers all variants.
What are the penalties for selling a Class A device without CDSCO registration?
Serious ones. Under the Drugs and Cosmetics Act 1940, penalties include product seizure, market withdrawal orders, fines, and potential criminal prosecution. CDSCO actively conducts market surveillance — unregistered device sellers have faced enforcement action. It is not a theoretical risk.
How long is a Class A device registration valid, and when should I renew?
Registration is valid for 5 years. Start your renewal application at least 3 to 4 months before expiry — a lapsed registration means your product cannot legally be sold until renewal is granted, and CDSCO processing takes time.
Does a CE mark or US FDA clearance waive CDSCO registration?
No. CE and FDA approvals support your application as evidence of conformity, but they do not replace Indian registration. You must complete the full SUGAM submission and receive a CDSCO certificate before selling in India — no shortcuts exist regardless of international approvals held.
Can the same MD 3 registration cover both sterile and non-sterile versions of a device?
No. Sterile and non-sterile versions are treated as separate products with different risk profiles and documentation requirements. The sterile version additionally requires ISO 13485 QMS evidence from the manufacturer. Each version needs its own application and registration certificate.
Who is legally responsible for CDSCO registration — the Indian importer or the foreign manufacturer?
The Indian importer (or Indian authorised representative) owns and files the MD 3 application and holds the registration certificate. The foreign manufacturer provides supporting documents — CFS, ISO 13485, Power of Attorney — but has no direct standing with CDSCO. Foreign manufacturers without an Indian entity cannot apply directly.
Is SUGAM the only route for Class A device registration?
Yes. All applications must be submitted through the SUGAM portal at sugam.gov.in. Physical or offline submissions are not accepted. The portal handles the entire process — filing, document upload, fee payment, query response, and certificate download. If you encounter technical portal issues, document them and contact CDSCO's helpdesk immediately.
Do Indian distributors and retailers need a separate licence to sell Class A devices?
Yes. The product registration (MD 1 or MD 3) is held by the manufacturer or importer. Distributors and retailers must separately obtain a medical device sales licence from the State Licensing Authority (SLA) under MDR 2017. Selling without this licence is a violation even if the product itself is fully registered with CDSCO.
How long must an importer maintain records after registration is granted?
A minimum of 5 years. Records must include batch numbers, quantities, supply dates, and customer details for every consignment. Complaint records, adverse event reports, and any field safety corrective action documentation must also be retained. These must be available on demand during a CDSCO inspection or product recall — failure to produce them carries significant enforcement risk.





































