- CDSCO Medical Device Registration For Oncology devices in India are classified under Class A to Class D — with most cancer diagnostic and therapeutic devices falling under Class C or Class D, requiring CDSCO Central License.
- CDSCO's SUGAM online portal is the mandatory submission platform for all oncology device registration applications in India as of 2026 — paper filings are no longer accepted.
- International oncology device manufacturers with FDA, CE Mark, TGA, Health Canada, or PMDA approval can apply for a clinical data waiver, significantly cutting registration time and cost.
- Registration costs for oncology devices under CDSCO range from approximately INR 50,000 for Class B devices to INR 5 lakh or more for Class D devices requiring clinical trial data.
Introduction
If you are a manufacturer, importer, or distributor planning to bring an oncology medical device to the Indian market, understanding the CDSCO medical device registration process is the single most important step in your market entry strategy. India is one of the fastest-growing medical device markets in the world, and oncology — driven by a rising cancer burden, expanding hospital infrastructure, and government-backed schemes like Ayushman Bharat — is among the most active segments.


The regulatory authority that governs this space is the Central Drugs Standard Control Organization (CDSCO), operating under India's Ministry of Health and Family Welfare. CDSCO regulates all medical devices — including oncology diagnostic systems, cancer surgical instruments, radiation therapy equipment, and IVD cancer assays — under the Medical Devices Rules, 2017 (MDR 2017), last amended in 2020.
This guide covers everything you need to know about CDSCO medical device registration for oncology in 2026 — from device classification and documentation requirements to SUGAM portal submission, timelines, costs, clinical data waivers, and the most common approval pitfalls. If you are serious about getting your cancer device approved in India, this is your starting point.
What Is CDSCO Medical Device Registration for Oncology?
CDSCO medical device registration for oncology is the formal regulatory process through which a manufacturer, importer, or authorized agent obtains a license to legally import, manufacture, or sell a cancer-related medical device in India. The process is governed by the Medical Devices Rules, 2017, notified under the Drugs and Cosmetics Act, 1940, and administered by CDSCO at the central level as well as State Licensing Authorities (SLAs) at the state level, depending on device risk classification.
All medical devices — including oncology products — that fall within the scope of India's regulated device list must be registered before they can be placed on the Indian market. Non-compliance results in seizure, import ban, and significant financial penalties. For foreign manufacturers, the entire registration process must be conducted through an India-based Authorized Indian Representative (AIR).
2026 Update: As per the latest CDSCO circular, the scope of regulated medical devices in India has been progressively expanded. If your oncology device was previously unregulated under India's older framework, it is highly advisable to verify current classification status on the CDSCO website or consult a regulatory affairs expert before assuming exemption.
CDSCO Device Classification for Oncology — Class A to Class D
Under the Medical Devices Rules, 2017, all medical devices — including oncology devices — are classified into four risk-based categories. This classification determines which regulatory authority handles the application, what documentation is required, and whether clinical data must be submitted or can be waived.
| Class | Risk Level | Oncology Device Examples | Licensing Authority | Clinical Data |
| Class A | Low | Surgical gauze, specimen containers, oncology patient monitoring consumables | State Licensing Authority (SLA) | Not required |
| Class B | Low-Moderate | Biopsy needles, surgical forceps, infusion sets used in chemotherapy | State Licensing Authority (SLA) | Not required |
| Class C | Moderate-High | Radiotherapy planning software, oncology-specific infusion pumps, diagnostic ultrasound for cancer, IVD cancer screening tests | CDSCO Central Licence | Required or waivable |
| Class D | High | Linear accelerators (LINAC), implantable brachytherapy seeds, robotic oncology surgery systems, AI-based cancer diagnostic SaMD | CDSCO Central Licence | Mandatory or waivable via reference country approval |
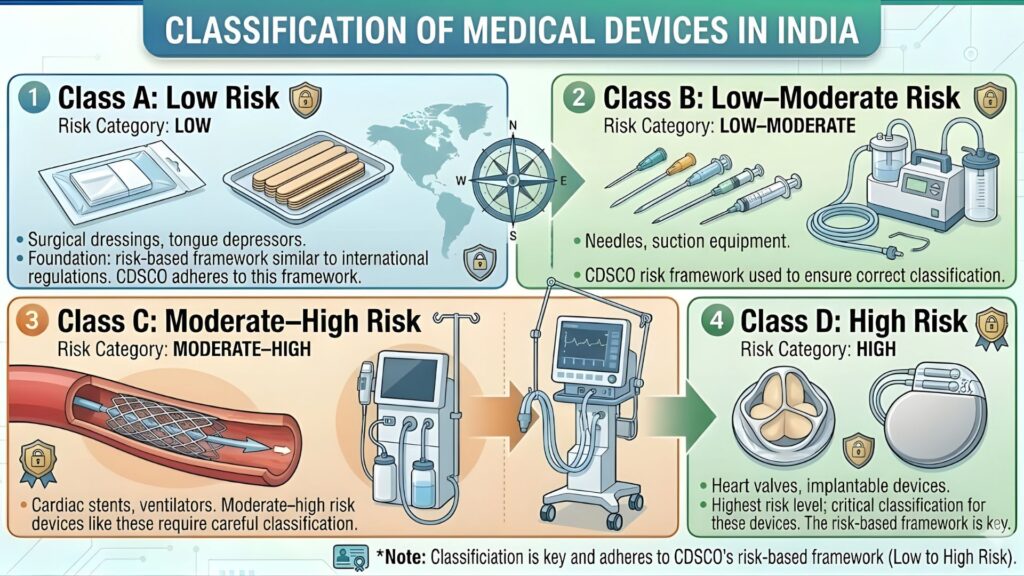

Most oncology therapeutic and diagnostic devices — including radiation systems, cancer biomarker assays, positron emission tomography (PET) components, and computer-aided detection (CAD) tools for cancer — fall under Class C or Class D. Manufacturers must ensure correct classification before beginning documentation, as incorrect classification is one of the leading causes of CDSCO registration delays.
| Important: Oncology combination products — those that pair a medical device with a drug or biologic (such as drug-eluting beads for chemoembolisation or radioactive seeds for brachytherapy) — require a dual regulatory strategy under both CDSCO and the Central Drugs Standard Control Organization's pharmaceutical division. This significantly increases complexity and timeline. |
Who Needs to Apply for CDSCO Oncology Device Registration?
Understanding who bears the registration obligation is critical before beginning the process. Under Indian law, the following parties are required to obtain CDSCO registration for oncology devices:
- Indian manufacturers: Any company manufacturing a regulated oncology device in India must obtain a manufacturing license from the State Licensing Authority (for Class A/B) or CDSCO (for Class C/D) before commercial production and sale.
- Foreign manufacturers / importers: Any foreign company wishing to import and sell an oncology device in India must appoint an Authorized Indian Representative (AIR) and obtain an import license under Form MD-14 or MD-15 depending on device class.
- Indian importers / distributors: Entities importing oncology devices on behalf of a foreign manufacturer must hold a valid import license and ensure the foreign manufacturer holds valid registration.
- Repacking / relabelling entities: Companies that repack or relabel regulated oncology devices in India are also subject to CDSCO licensing requirements.
| Key Point for Foreign Oncology Device Manufacturers (2026): If you are a foreign manufacturer, you do not directly apply to CDSCO. Your AIR applies on your behalf. The AIR must be a legal entity registered in India and must submit a duly notarized and apostilled Power of Attorney along with the registration application. Choosing the right AIR with oncology regulatory experience is a strategic decision, not just an administrative one. |
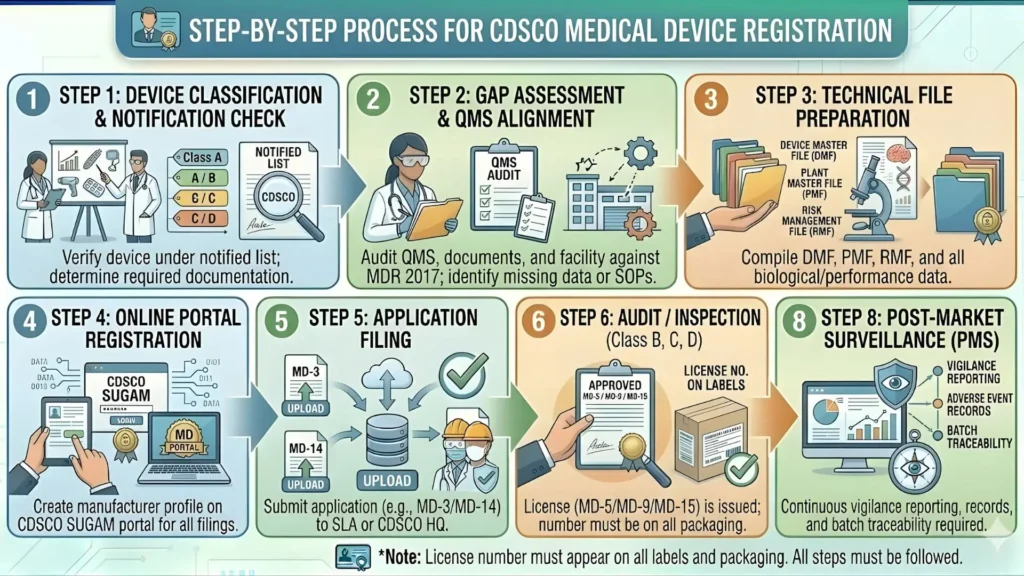
CDSCO SUGAM Portal — How to Submit Your Oncology Device Application
All CDSCO medical device registration applications — including those for oncology devices — must be filed online through the SUGAM portal (sugam.gov.in). The portal manages the entire lifecycle of the application, from initial submission and fee payment to query responses, additional information requests, and final license issuance.
Step-by-Step SUGAM Submission Process for Oncology Devices
- Create and verify your SUGAM account: Register as a manufacturer or importer on the SUGAM portal. Foreign manufacturers must register through their AIR's account. Ensure all company details, contact information, and GST/DUNS numbers are accurate before filing.
- Select the correct application form: Use Form MD-14 for import license (Class C and D oncology devices) or Form MD-41 for manufacturing license. Selecting the wrong form results in outright rejection and restarts the clock.
- Upload the complete technical dossier: All documentation — device description, risk management file, quality management system certificates, clinical data or waiver justification, labelling, and test reports — must be uploaded in the specified PDF format. CDSCO does not accept incomplete submissions.
- Pay the prescribed government fee: Fee payment is made online through the SUGAM portal payment gateway. Government registration fees for Class C and Class D oncology devices range from INR 50,000 to INR 5,00,000 depending on device category and applicant type (Indian vs. foreign manufacturer).
- Track application status and respond to queries: CDSCO officers raise queries (Additional Information requests) through the portal. All responses must be submitted within the specified time window. Missing a query deadline results in application rejection.
- Receive Grant of Import License or Manufacturing License: Upon successful review, CDSCO issues the licence electronically through SUGAM. The licence is valid for 5 years and must be renewed before expiry.

| Common SUGAM Portal Mistake for Oncology Devices: Many applicants upload unattested or non-notarised documents, use incorrect form numbers for their device class, or submit incomplete Annexures. CDSCO's online system has strict file format requirements — all documents must be in PDF/A format, and each file must not exceed the size limit specified in the submission guidelines. Always review the current SUGAM submission checklist on CDSCO's official website before filing. |
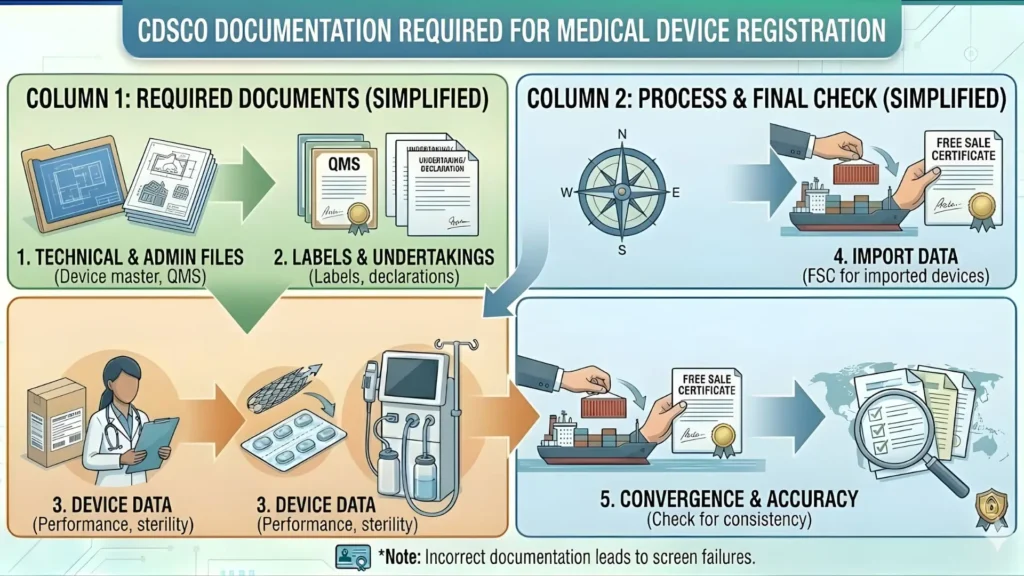
Required Documentation for CDSCO Oncology Device Registration
The documentation requirements for CDSCO oncology device registration vary based on device classification, whether the applicant is an Indian or foreign manufacturer, and whether clinical data is being submitted or a waiver is being claimed. Below is a comprehensive checklist applicable to Class C and Class D oncology devices — the categories covering the vast majority of cancer diagnostic and therapeutic products.
Core Technical Dossier Requirements
- Device description and intended use: A detailed technical description of the oncology device, its mechanism of action, intended patient population (including cancer type and stage where relevant), and indications for use.
- Risk management file: Prepared in accordance with ISO 14971:2019. Must address oncology-specific risks including radiation exposure (for radiotherapy devices), cytotoxic drug contact (for chemotherapy delivery devices), and software failure modes (for AI-based cancer diagnostics).
- Quality Management System certificate: ISO 13485:2016 certificate from an accredited certification body. CDSCO accepts certificates issued by recognized bodies. For foreign manufacturers, the QMS certificate must be in the name of the manufacturing facility, not just the parent company.
- Performance and safety test reports: Electrical safety (IEC 60601 series), electromagnetic compatibility (IEC 60601-1-2), biocompatibility (ISO 10993 series for contact devices), sterility validation (for sterile oncology devices), and any oncology-specific performance standards applicable to your device type.
- Labelling and Instructions for Use (IFU): Must comply with CDSCO labelling requirements under Schedule V of the Medical Devices Rules, 2017. Labels must include the device name, model, manufacturer name and address, manufacturing date, shelf life (if applicable), and statutory warnings. IFU must be in English.
- Clinical data or waiver justification: For Class C and Class D oncology devices. If submitting clinical data, it must include clinical investigation reports or published peer-reviewed literature demonstrating safety and performance in the intended oncology indication. If claiming a waiver, a formal waiver application with supporting reference country approval documents must be included.
- Free Sale Certificate (FSC): For foreign manufacturers, a Certificate of Free Sale from the country of manufacture, duly notarized and apostilled, confirming the device is legally marketed in its home market.
- Authorized Indian Representative (AIR) agreement: Notarized Power of Attorney, AIR appointment letter, and AIR's Indian business registration documents.
- Post-Market Surveillance (PMS) plan: Mandatory for Class C and Class D oncology devices. Must outline the manufacturer's plan for monitoring device performance in the Indian market post-approval, including adverse event reporting procedures under CDSCO's vigilance framework.

Additional Requirements for Specific Oncology Device Categories
| Oncology Device Type | Additional CDSCO Requirement |
| Radiation therapy devices (LINAC, brachytherapy) | Atomic Energy Regulatory Board (AERB) clearance required in addition to CDSCO registration. AERB governs radiation-emitting equipment in India independently. |
| AI-based oncology diagnostics (SaMD) | Software documentation per IEC 62304, intended use validation, algorithm performance data on Indian patient population strongly recommended |
| In vitro diagnostic oncology devices (cancer biomarker assays, liquid biopsy) | Registered under MDR 2017 Schedule — must specify diagnostic claim precisely. Companion diagnostics linked to specific drugs require cross-referencing with drug approval. |
| Single-use oncology devices | Sterility and packaging validation mandatory. Re-use prohibition labelling required per CDSCO guidelines on single-use devices. |
| Oncology implants (stents, seeds) | Biocompatibility full battery per ISO 10993, implant-specific risk analysis, and clinical data from studies in oncology patients required. |
CDSCO Clinical Data Waiver for Oncology Devices — The Fast Track to Indian Market
One of the most strategically important provisions in India's Medical Devices Rules, 2017 for oncology device manufacturers is the clinical data waiver. For international manufacturers who have already obtained regulatory approval for their oncology device in a reference country, CDSCO may waive the requirement to conduct separate clinical investigations in India — dramatically reducing both the time and cost of registration.
Reference Countries Accepted by CDSCO for Clinical Data Waiver
| Reference Country / Region | Regulatory Body | Approval Document Required |
| United States of America | U.S. Food and Drug Administration (FDA) | 510(k) Clearance letter, PMA Approval Order, or De Novo Grant |
| European Union | CE Mark under EU MDR 2017/745 or MDD | CE Certificate + Declaration of Conformity |
| Australia | Therapeutic Goods Administration (TGA) | ARTG (Australian Register of Therapeutic Goods) entry certificate |
| Canada | Health Canada | Medical Device Licence (MDL) |
| Japan | Pharmaceuticals and Medical Devices Agency (PMDA) | Shounin (approval certificate) or Ninsho (certification) |
To claim a clinical data waiver, the applicant must submit the following as part of the CDSCO registration application through the SUGAM portal:
- A certified copy of the reference country regulatory approval (e.g., FDA 510(k) clearance letter), apostilled or notarised as required
- A formal written justification explaining why the clinical data from the reference country is applicable to the Indian population and patient setting
- Evidence that the device is currently being sold and actively used in the reference country market
- A statement confirming the device has not been withdrawn from any market due to safety concerns
- An Indian-specific risk summary addressing any population-specific considerations — particularly relevant for oncology devices used in cancer types with different prevalence profiles in India versus Western markets
| Strategic Insight for 2026: If your oncology device holds both FDA clearance and CE Mark, lead with the FDA approval in your CDSCO waiver application. FDA approval is the most commonly cited reference in CDSCO waiver grants for Class C and Class D oncology devices. Ensure your approval documents are recent — CDSCO reviewers have been known to raise queries on reference approvals older than five years. |
CDSCO Oncology Device Registration — Timelines and Costs in 2026
One of the first questions every oncology device manufacturer asks is: how long will this take, and what will it cost? Here is a realistic breakdown based on current CDSCO processing realities in 2026.
| Device Class | Regulatory Path | Realistic Timeline | Key Timeline Drivers |
| Class A | State Licensing Authority | 1–3 months | Documentation completeness |
| Class B | State Licensing Authority | 2–6 months | SLA workload, documentation quality |
| Class C (with waiver) | CDSCO Central | 6–14 months | CDSCO officer assignment, query cycles |
| Class C (clinical trial) | CDSCO Central | 18–36 months | Clinical investigation approval + trial conduct |
| Class D (with waiver) | CDSCO Central | 9–18 months | Document quality, reference approval recency |
| Class D (clinical trial) | CDSCO Central | 30–54 months | CDSCO trial approval, trial conduct, data analysis |
These timelines assume a complete and well-prepared application at the time of submission. Each CDSCO query cycle — where CDSCO raises an Additional Information (AI) request and the applicant must respond — typically adds 2–4 months to the timeline. Complex Class D oncology devices have seen as many as three or four query cycles before final approval. Working with an experienced CDSCO regulatory consultant who specializes in oncology submissions is one of the most effective ways to reduce these delays.
Government Registration Fees — CDSCO 2026
| Application Type | Device Class | Government Fee (INR) | Notes |
| Import Licence (Foreign Manufacturer) | Class C | INR 50,000 per device | Per device model — not per product family |
| Import Licence (Foreign Manufacturer) | Class D | INR 1,00,000 per device | Additional AERB fees apply for radiation devices |
| Manufacturing Licence (Indian Manufacturer) | Class C | INR 20,000 | Payable to CDSCO via SUGAM portal |
| Manufacturing Licence (Indian Manufacturer) | Class D | INR 50,000 | Site inspection fees additional |
| Licence Renewal | Class C / Class D | 50% of original fee | Must be filed 6 months before expiry |
| Licence Amendment (change in model/specification) | Any Class | 25% of original fee | Per amendment request |
| Important Cost Note: Government fees are only one part of the total registration cost. Professional costs for regulatory affairs consultants, document preparation, notarisation, apostille, translation (if applicable), ISO 13485 certification (if not already held), test report generation, and clinical data compilation typically add INR 3 lakh to INR 25 lakh or more to the overall budget for a Class D oncology device registration in India. |
CDSCO Registration for Specific Oncology Device Categories
Oncology is a broad clinical field, and the regulatory requirements vary meaningfully across different device categories. Here is a focused breakdown of what CDSCO expects for the most commonly registered cancer device types in India.
1. Radiation Oncology Devices (LINAC, Brachytherapy, Cobalt-60 Systems)
Radiation therapy devices are among the most tightly regulated oncology products in India. In addition to CDSCO registration under Class D, all radiation-emitting devices require separate clearance from the Atomic Energy Regulatory Board (AERB) under the Atomic Energy (Radiation Protection) Rules, 2004. AERB clearance must be obtained before the device can be installed or operated at any Indian healthcare facility.
Manufacturers must ensure that both the CDSCO import license and the AERB equipment approval are coordinated, as hospitals purchasing these devices require both certificates before commissioning. AERB site approvals are also required for each installation, which means the regulatory burden extends beyond the manufacturer to the end-user facility.
2. Oncology In Vitro Diagnostic (IVD) Devices
Cancer IVDs — including HER2 amplification testing kits, BRCA mutation assays, liquid biopsy platforms, circulating tumour DNA (ctDNA) detection systems, and PSA immunoassay kits — are regulated as Class C or Class D devices under the Medical Devices Rules, 2017, scheduled under the notified IVD list.
For oncology IVDs, CDSCO requires detailed analytical performance data including sensitivity, specificity, limit of detection, precision, and interference studies. For companion diagnostics tied to specific oncology drugs, the IVD registration must reference the associated drug's approval. CDSCO expects performance data from studies conducted on or validated against Indian patient samples for novel oncology IVDs — a requirement that catches many international manufacturers off guard.
3. AI-Powered Oncology Diagnostic Software (SaMD)
Artificial intelligence-based cancer diagnostic tools — such as AI-assisted mammography CADe systems, AI lung nodule detection software, AI pathology slide analysis platforms, and AI-based colonoscopy detection tools — are classified as Software as a Medical Device (SaMD) and fall under CDSCO regulation when they meet the definition of a medical device under Indian law.
CDSCO's 2023 guidance on SaMD (aligned with IMDRF SaMD framework) requires manufacturers to submit IEC 62304 software lifecycle documentation, algorithm validation reports, cybersecurity risk analysis, and — for AI oncology tools specifically — evidence that the algorithm's performance has been validated on data representative of Indian patients. This last requirement is a significant consideration for international AI oncology products developed and trained primarily on Western datasets.
4. Oncology Surgical and Interventional Devices
Minimally invasive oncology devices — including tumour ablation probes (radiofrequency, microwave, cryoablation), laparoscopic oncology instruments, endoscopic resection devices, and robotic surgery systems used in cancer procedures — are classified as Class B to Class D depending on their risk profile and contact nature.
For Class C and Class D surgical oncology devices, CDSCO requires biocompatibility testing per ISO 10993 (particularly for devices that contact tissue or blood), sterility validation for sterile devices, and electrical safety data per IEC 60601-1. Robotic surgery systems carry the most demanding documentation burden and typically require clinical data from peer-reviewed literature or clinical investigations specifically in oncology surgical settings.
Common Mistakes That Delay CDSCO Oncology Device Approval
Based on real-world experience with CDSCO submissions across the oncology spectrum, here are the most consistent errors that result in query letters, application rejections, or multi-year delays.
- Incorrect device classification: Classifying a Class D oncology device as Class C to reduce regulatory burden is a common and costly error. CDSCO officers scrutinize the risk justification carefully — especially for implantable and radiation-emitting oncology devices. Incorrect classification leads to rejection and restart.
- Appointing an under-resourced AIR: Foreign oncology device manufacturers sometimes appoint AIRs based purely on cost rather than competence. An AIR with no CDSCO oncology experience will be unable to respond effectively to technical queries, delaying approvals significantly.
- Incomplete clinical data waiver package: Submitting only the reference country approval letter without the supporting justification, active marketing evidence, and population-specific risk summary is the most common reason CDSCO rejects clinical data waiver claims for oncology devices.
- Non-compliant labelling and IFU: Labels that do not include all Schedule V mandatory elements, or IFUs that describe oncology indications not covered by the reference country approval, are consistent sources of CDSCO queries.
- Missing AERB coordination for radiation oncology devices: Filing CDSCO registration for a linear accelerator or brachytherapy system without an AERB strategy in place leads to a situation where the device is CDSCO-approved but cannot be legally operated in India. Both approvals must be planned together.
- Outdated ISO 13485 certificate: CDSCO routinely rejects applications where the QMS certificate has expired or is nearing expiry (within 3 months). The certificate must remain valid throughout the entire review period.
- Failure to address Indian population-specific considerations: For AI oncology devices and oncology IVDs, CDSCO expects some acknowledgement of performance data or validation in the Indian patient population. Submitting data exclusively from Western patient cohorts without addressing Indian demographic applicability is a growing reason for AI queries in 2026.
Conclusion
CDSCO medical device registration for oncology in India is a structured, rules-based process — and in 2026, it is more rigorous and more consequential than at any point in the past decade. The Indian cancer device market is growing fast, the regulatory framework is maturing rapidly, and CDSCO's expectations for documentation quality, clinical evidence, and post-market accountability are rising in step.
The manufacturers and importers who succeed in this environment are the ones who take classification seriously from day one, who invest in qualified documentation and experienced AIRs, and who approach CDSCO registration as a long-term market access strategy rather than a one-time filing exercise. The clinical data waiver pathway remains one of India's most valuable market entry tools for internationally approved oncology devices — but it must be used correctly to deliver its full benefit.
Whether you are registering a radiation therapy system, an AI-based cancer diagnostic tool, an oncology IVD, or a minimally invasive cancer surgical device, the regulatory pathway to the Indian market is navigable. The requirements are clear. The process is defined. With the right preparation and the right regulatory partner, CDSCO approval for your oncology device is within reach.
| Ready to register your oncology device in India? Partnering with a CDSCO-experienced regulatory affairs consultant who specialises in oncology submissions can reduce your timeline by 30–40%, improve first-cycle approval rates, and help you avoid the costly query cycles that delay most first-time applicants. Engage your regulatory team before finalising your dossier — not after your first CDSCO rejection. |
Frequently Asked Questions
How long does CDSCO take to approve a Class D oncology device in 2026?
For a Class D oncology device with a valid clinical data waiver, realistic approval timelines in 2026 range from 9 to 18 months from complete application submission. For devices requiring clinical investigations, add 18–36 months for trial conduct and data analysis on top of the CDSCO review period. Application quality is the single biggest controllable factor — a complete, well-organised dossier with no documentation gaps significantly reduces query cycles.
Can I register my oncology device in India without clinical trials if I have FDA approval?
Yes, in most cases. CDSCO's clinical data waiver provision allows Class C and Class D oncology device manufacturers who hold FDA clearance or approval to apply for exemption from conducting Indian clinical investigations. The waiver is not automatic — you must formally apply with supporting documentation including the FDA approval certificate, evidence of active marketing in the US, and an Indian population-specific risk justification. CDSCO has the discretion to accept or reject waiver requests and may request bridging data even with FDA approval for certain novel oncology device categories.
Does my linear accelerator or brachytherapy device need AERB approval as well as CDSCO?
Yes, absolutely. Radiation-emitting oncology devices require both CDSCO import/manufacturing licence and AERB equipment type approval under Indian atomic energy regulations. These are two separate regulatory processes managed by two different government bodies. AERB approval must be obtained before the device can be imported, installed, or operated at any Indian healthcare facility. Many manufacturers make the mistake of securing CDSCO approval first and then discovering the AERB process adds a further 6–18 months. Plan both processes concurrently from day one.
What is an Authorized Indian Representative and do I really need one?
For foreign oncology device manufacturers, the Authorised Indian Representative (AIR) is a mandatory legal requirement under the Medical Devices Rules, 2017 — not an optional convenience. You cannot apply for CDSCO registration directly as a foreign company. Your AIR is the legal entity that submits the application, receives CDSCO correspondence, and holds primary regulatory accountability in India on your behalf. Choosing an AIR with specific CDSCO oncology device experience is important — a poorly qualified AIR directly impacts your approval timeline and query resolution quality.
How often do I need to renew my CDSCO oncology device license?
CDSCO import and manufacturing licenses for medical devices — including oncology devices — are valid for 5 years from the date of issue. Renewal applications must be submitted through the SUGAM portal at least 6 months before the expiry date. Failure to renew on time results in license lapse, meaning you are no longer legally authorized to import or sell the device in India until the renewed license is issued. Plan your renewal well in advance — CDSCO's renewal processing times can run 3–6 months.
Are AI-based oncology diagnostic tools regulated by CDSCO in India
Yes. AI-based oncology diagnostic software that meets the definition of a medical device under the Medical Devices Rules, 2017 — specifically software intended to diagnose, detect, monitor, or predict cancer or cancer-related conditions — is regulated by CDSCO as Software as a Medical Device (SaMD). Classification is typically Class C or Class D depending on the intended use and risk level. CDSCO expects software lifecycle documentation per IEC 62304, algorithm validation reports, and — increasingly in 2026 — performance data from studies conducted on Indian patient data to support registration.





































