- CDSCO Import License For Ventilators are Class C (high-risk) notified medical devices under India's Medical Devices Rules, 2017 — importing them without a valid CDSCO import license is a punishable offence under the Drugs and Cosmetics Act, 1940, regardless of the country of manufacture.
- The import license process involves two connected applications: Form MD-14 filed by the Indian importer, and Form MD-15 filed by the overseas (foreign) manufacturer to register with CDSCO — both must be approved before any commercial import can begin.
- Technical compliance with BIS IS 13450 (Part 2/Sec 12): 2023 and IS 7655 must be demonstrated through accredited test reports; CDSCO reviewers are now specifically checking for IS 13450 (Part 2/Sec 12): 2023 alignment in 2026 import dossiers.
- 2026 update: CDSCO has tightened post-market surveillance (PMSR) obligations for imported Class C devices, is implementing UDI (Unique Device Identification) requirements for high-risk devices, and has strengthened the Authorized Indian Agent (AIA) accountability framework for overseas manufacturers.
Introduction
Importing ventilators into India is one of the most tightly regulated commercial activities in the country's healthcare supply chain — and justifiably so. Ventilators are life-support devices. A poorly documented import, a device that hasn't been properly tested, or a shipment that bypasses licensing not only violates Indian law but puts patients at serious risk.
If you are an Indian company that wants to import ventilators from an overseas manufacturer, or if you are a foreign company that wants to sell your ventilators in the Indian market, this guide is written specifically for you. The CDSCO import license for ventilators involves two distinct but interconnected applications — Form MD-14 for the Indian importer and Form MD-15 for the overseas manufacturer — and both need to be understood together.
This is a complete, factual, and practitioner-oriented guide covering the full import licensing pathway as of 2026: legal framework, BIS standard requirements including IS 13450 (Part 2/Sec 12): 2023, eligibility, documents, step-by-step process, fees, timelines, post-license obligations, and the latest regulatory updates from CDSCO. No shortcuts, no outdated information.
Understanding the CDSCO Import License for Ventilators
Under Rule 27 of the Medical Devices Rules, 2017 (MDR 2017), no person shall import any notified medical device into India without a valid license granted by the Central Licensing Authority (CLA), which is CDSCO, New Delhi. Ventilators are specifically listed as notified medical devices under Schedule I of MDR 2017 and classified as Class C — the high-risk category.


The import licensing structure for ventilators is a two-track system:
- Form MD-14: The import license application filed by the Indian importer. This is the license that authorizes the Indian entity to bring ventilators into India for commercial distribution and sale.
- Form MD-15: The registration application filed by the overseas (foreign) manufacturer. This establishes CDSCO's recognition of the foreign manufacturer, confirms the product's safety and performance credentials, and is linked to the MD-14 license.
Both tracks are filed on the Sugam portal (sugam.gov.in). The Indian importer cannot operate without an MD-14 license, and the foreign manufacturer's products cannot be legally imported without MD-15 registration. In practice, both applications are usually prepared and submitted simultaneously or in a coordinated sequence.
Key distinction: The manufacturing license (Form MD-3/MD-4/MD-9) is for domestic manufacturers only. If you are importing ventilators — not making them in India — your pathway is Form MD-14 (importer) and Form MD-15 (overseas manufacturer). These are two completely different regulatory tracks.
What Is a Ventilator Under Indian Regulatory Law?
Before starting the import license process, confirm that your product is classified as a ventilator under Indian medical device law. The classification determines your regulatory track, applicable fees, documentation depth, and oversight frequency.
Under Schedule I of MDR 2017, ventilators are a notified Class C medical device. The regulatory term covers a wide range of respiratory support devices:
- Invasive mechanical ventilators — ICU-grade, requiring endotracheal intubation
- Non-invasive ventilators — BiPAP and CPAP devices for respiratory therapy
- Transport and emergency ventilators
- High-frequency oscillatory ventilators (HFOV) for neonatal and adult critical care
- Neonatal and paediatric ventilators
- Home-care ventilators for chronic respiratory conditions
- Anaesthesia workstations with integrated mechanical ventilation function
- Portable ventilators for sub-acute and step-down care settings
If your imported device delivers, assists, or controls mechanical ventilation — even as a secondary or combined function — CDSCO may classify it as a ventilator. Always confirm your device classification with CDSCO before initiating the import license application. An incorrect classification wastes months and money.
Legal and Regulatory Framework for Importing Ventilators into India
| Regulation / Standard | Relevance to Ventilator Import |
| Drugs and Cosmetics Act, 1940 | Primary statute; importing without license is a criminal offence under this Act |
| Medical Devices Rules, 2017 (MDR 2017) | Governs import classification, application process, standards compliance, and post-market obligations |
| Schedule I (MDR 2017) | Lists ventilators as notified medical devices; Class C classification |
| Rule 27 (MDR 2017) | The specific rule mandating a license for import of notified medical devices |
| IS 13450 (Part 2/Sec 12): 2023 | Updated BIS standard for lung ventilators aligned to ISO 80601-2-12; test reports now expected in 2026 import dossiers by CDSCO reviewers |
| IS 7655 | Earlier BIS standard for lung ventilators aligned to IEC 60601-1; still referenced in CDSCO checklists alongside IS 13450 |
| IEC 60601-1 | International electrical safety standard for medical devices; referenced by CDSCO |
| ISO 80601-2-12 | International standard for critical care ventilators; IS 13450 Pt 2/Sec 12 is the Indian equivalent |
| IEC 60601-1-2 | Electromagnetic compatibility requirements; required in import technical dossier |
| ISO 14971 | Risk management for medical devices; mandatory for Class C import dossier |
| ISO 13485:2016 | QMS standard; overseas manufacturers must demonstrate QMS compliance as part of MD-15 |
| CDSCO Guidance Documents | Application checklists, SOPs, and circulars on cdsco.gov.in; must be followed for current applications |
| DPCO / NPPA | Price control regulations; may apply to imported ventilators if placed under essential medical device price list |
BIS Standards for Imported Ventilators — IS 13450 (Part 2/Sec 12): 2023 and IS 7655
This is the section most importers and overseas manufacturers underestimate, and it is responsible for a significant share of CDSCO queries and application delays in 2025–2026. Indian BIS standards apply to imported ventilators just as much as domestically manufactured ones.


IS 13450 (Part 2/Sec 12): 2023 — The Standard CDSCO Reviewers Are Now Checking
IS 13450 (Part 2/Sec 12): 2023 is the Bureau of Indian Standards (BIS) specification for lung ventilators for medical use, published in 2023. It is the Indian adoption of ISO 80601-2-12 — the international standard for critical care ventilators — and represents a comprehensive upgrade from earlier ventilator standards. The 2023 edition addresses:
- Alarm system performance — priority levels, response times, and alarm pause functions for ICU ventilators
- Tidal volume and respiratory rate accuracy across varying patient lung compliance conditions
- PEEP (Positive End-Expiratory Pressure) delivery accuracy and stability
- Inspired oxygen concentration accuracy (FiO2 delivery)
- Software and firmware validation requirements for electronically controlled ventilators
- Electromagnetic compatibility (EMC) performance in hospital environments — particularly important in ICU settings with multiple electrical devices
- Usability and human factors engineering documentation
- Biocompatibility of patient-contact components such as breathing circuits, valves, and masks
For import license applications filed in 2026, CDSCO reviewers are now specifically checking whether your technical dossier — specifically the test reports section — explicitly references IS 13450 (Part 2/Sec 12): 2023. Test reports that reference only IEC 60601-1 or ISO 80601-2-12 without the IS 13450 designation may trigger a query asking for IS 13450 compliance evidence. Get this right before you submit.
IS 7655 — Still Referenced, Still Required
IS 7655 is the earlier BIS standard for lung ventilators for medical use, aligned to IEC 60601-1 (general electrical safety for medical devices). While IS 13450 (Part 2/Sec 12): 2023 is now the more specific and updated ventilator standard, IS 7655 remains referenced in CDSCO application checklists. A complete 2026 import dossier should carry test reports addressing both standards. Importers relying only on their overseas manufacturer's IEC 60601-1 report — without explicit IS 7655 and IS 13450 (Part 2/Sec 12): 2023 alignment — commonly face queries.
Does BIS Product Registration Apply to Imported Ventilators?
As of 2026, BIS product registration under IS 13450 or IS 7655 is not a separate mandatory pre-condition for obtaining a CDSCO import license. What CDSCO requires within the import dossier is test report evidence of compliance with these standards from an accredited laboratory. However:


If you plan to supply ventilators to government agencies through the Government e-Marketplace (GeM), Central Medical Services Society (CMSS), or state procurement agencies, BIS product registration is frequently a mandatory tender eligibility requirement — separate from and additional to your CDSCO import license.
Voluntary BIS registration under IS 13450 (Part 2/Sec 12): 2023 or IS 7655 strengthens market credibility, supports tender eligibility, and demonstrates long-term quality commitment to hospital procurement teams.
| Standard | Scope | Applicable To Imports | CDSCO Status 2026 |
| IS 13450 (Part 2/Sec 12): 2023 | Lung ventilators — performance, alarms, software, EMC (aligns ISO 80601-2-12) | All imported ventilator types; critical for ICU/critical care | Test report required in import dossier — reviewers checking explicitly in 2026 |
| IS 7655 | Lung ventilators — electrical safety (aligns IEC 60601-1) | All imported ventilators with electrical function | Referenced in CDSCO checklist; test report expected alongside IS 13450 |
| IEC 60601-1 | General electrical safety for medical devices | All electrically operated medical devices | Referenced; IS 7655 covers this for Indian compliance |
| ISO 80601-2-12 | Critical care ventilators — specific requirements | ICU / critical care ventilators | IS 13450 (Part 2/Sec 12): 2023 is the Indian version |
| IEC 60601-1-2 | EMC requirements | All electrical medical devices | Required for electromagnetic compatibility evidence |
| ISO 14971 | Risk management | All Class C medical devices | Mandatory in import technical dossier |
| ISO 13485:2016 | QMS standard | Overseas manufacturer (MD-15) | QMS certificate from overseas manufacturer required |
Who Needs a CDSCO Import License for Ventilators?
Indian Importers (Form MD-14)
Any Indian company or individual bringing ventilators from overseas into India for commercial distribution and sale requires a CDSCO import license. This includes:
- Sole distributors and exclusive importers appointed by overseas ventilator manufacturers
- Multi-brand medical equipment trading companies importing from various overseas suppliers
- Hospitals or healthcare systems that import ventilators directly for their own use and also sell or supply to affiliates
- Indian subsidiary companies of overseas ventilator manufacturers who manage the import function for the Indian market
The Indian importer must have a registered business entity in India, a physical place of business, and must appoint an Authorized Indian Agent (AIA) for each overseas manufacturer whose products they are importing. In many cases, the importer itself acts as the AIA — but this relationship must be formally documented and notarised.
Overseas (Foreign) Manufacturers (Form MD-15)
Every overseas manufacturer whose ventilators are being imported into India must separately register with CDSCO by filing Form MD-15 through their Authorised Indian Agent. This registration establishes CDSCO's recognition of the overseas manufacturer and creates an accountability chain for product safety, adverse event reporting, and field safety corrective actions in the Indian market.
An overseas manufacturer with multiple ventilator models must ensure each model is listed in the MD-15 registration. Adding new models after initial registration requires an amendment, not a fresh application.
Domestic Manufacturers — Not This Track
If you manufacture ventilators in India, your route is Form MD-3 (own premises) or Form MD-4 (loan/contract manufacturing), not MD-14/MD-15. The import license framework applies exclusively to devices manufactured outside India and imported for commercial sale.
Re-importers and Third-Country Importers
If ventilators are manufactured in one country but imported into India via a third country (e.g., assembled in Germany, re-exported via Singapore into India), the import license still applies. The original manufacturer's country and the country of shipment are both relevant in the MD-15 registration. CDSCO tracks country of origin, not just country of export.
Form MD-14 and Form MD-15
Form MD-14 — The Indian Importer's Application
Form MD-14 is the application form filed by the Indian importer to obtain a license to import a notified medical device — in this case, ventilators — for sale and distribution in India. Key details about MD-14:
- Filed on the Sugam portal by the Indian importer's registered entity
- Covers all ventilator models from a specific overseas manufacturer that the importer wishes to bring in
- Requires the importer to declare their Authorized Indian Agent relationship with the overseas manufacturer
- Must be supported by the overseas manufacturer's Form MD-15 registration (or simultaneous application)
- Results in the actual import license (Form MD-16) being issued by CDSCO on approval
- The licensed entity on Form MD-14 is the Indian importer — they are legally responsible for the product in India
Form MD-15 — The Overseas Manufacturer's Registration
Form MD-15 is filed by the overseas manufacturer through their Authorised Indian Agent to register the manufacturer and their specific ventilator products with CDSCO. Key details about MD-15:
- Filed on the Sugam portal by the Indian AIA on behalf of the overseas manufacturer
- Establishes CDSCO's recognition of the foreign manufacturer's facility, quality systems, and product technical details
- Requires submission of the overseas manufacturer's home country regulatory approval or Certificate of Free Sale (CFS) — typically from their national health authority
- Must include test reports, design dossier, and quality system documentation for each ventilator model being registered
- Creates a manufacturer-product-agent accountability record in CDSCO's database
- On approval, a Certificate of Registration is issued to the overseas manufacturer
Important: MD-14 and MD-15 are related but filed as separate applications on the Sugam portal. In practice, the Indian importer coordinates both filings because they are the Authorized Indian Agent in most cases. CDSCO links both applications during review.
| Feature | Form MD-14 (Indian Importer) | Form MD-15 (Overseas Manufacturer) |
| Who files it | Indian importer / authorized representative | Overseas manufacturer's Authorised Indian Agent (usually the importer) |
| Purpose | Grant of import license for the Indian market | Registration of overseas manufacturer and product with CDSCO |
| Resulting document | Form MD-16 Import License | Certificate of Registration for overseas manufacturer |
| Key content | Importer's business details, AIA relationship, product list, fees | Manufacturer's facility details, regulatory approvals, technical dossier per product |
| BIS / technical standards | Referenced and confirmed in application | Test reports covering IS 13450 (Pt 2/Sec 12): 2023, IS 7655, IEC 60601-1-2 submitted here |
| Validity period | 5 years from date of issue | 5 years from date of issue |
| Renewal | Form MD-6 equivalent for importers | Renewal of manufacturer registration |
Eligibility Criteria for CDSCO Ventilator Import License
Eligibility for Indian Importer (Form MD-14)
| Eligibility Criterion | Requirement |
| Legal Entity | Proprietorship, Partnership, LLP, Private Limited, or Public Limited company registered in India under applicable law |
| Registered Place of Business | Must have a physical office or place of business in India; P.O. Box addresses are not accepted |
| Authorised Indian Agent | Must have a formal, notarised Authorised Indian Agent agreement with each overseas manufacturer whose products are being imported |
| No Prior Disqualification | Importer must not have been convicted under the Drugs and Cosmetics Act or have an active license cancellation order |
| Storage Facility | Must have adequate, documented storage facilities appropriate for medical device storage (temperature-controlled where applicable for ventilator accessories) |
| Qualified Person | Must have a technically competent person associated with the import business; CDSCO reviews credentials during application |
Eligibility for Overseas Manufacturer (Form MD-15)
| Eligibility Criterion | Requirement |
| Manufacturing Facility | Must have a legally established manufacturing facility in the country of origin; facility address must match regulatory approvals |
| Home Country Regulatory Approval | Must hold marketing authorisation, CE marking, FDA clearance/approval, or equivalent regulatory approval from the country of manufacture or a recognised jurisdiction |
| Certificate of Free Sale (CFS) | CFS issued by the competent authority in the country of manufacture — confirming the device is freely sold in that market |
| Quality Management System | ISO 13485:2016 certification or equivalent QMS recognised by CDSCO; QMS certificate must be valid and not expired |
| Technical Documentation | Complete technical dossier per ventilator model including IS 13450 (Part 2/Sec 12): 2023 and IS 7655 test reports, design dossier, risk management file, IFU, labelling |
| Authorised Indian Agent | Must appoint an AIA with a registered office in India through a notarised agreement |
| No Active CDSCO Ban | Overseas manufacturer must not be subject to any CDSCO import alert, product ban, or recall notice in India |
Complete Document Checklist for CDSCO Ventilator Import License
The document requirement for importing a Class C medical device like a ventilator is extensive. Prepare every document before starting the Sugam portal application — incomplete submissions are the primary cause of delays.
Indian Importer Documents (Form MD-14)
- Certificate of Incorporation / Firm Registration / Proprietorship declaration of the Indian importing entity
- PAN Card of the company or proprietor
- GST Registration Certificate
- Board Resolution authorizing the signatory for CDSCO applications
- List of Directors, Partners, or Proprietor with contact information
- Notarized Authorized Indian Agent agreement between the Indian importer and the overseas manufacturer
- Power of Attorney if filing through a regulatory consultant or third-party agent
- Details of the Indian importer's storage facilities (address, storage conditions, area)
- Details of the qualified technical/responsible person associated with the import business
- Government fee payment receipt (online payment through Sugam portal)
- Declaration and undertaking as required under MDR 2017
Overseas Manufacturer Documents (Form MD-15)
- Proof of legal establishment of the overseas manufacturing entity in their home country
- Valid marketing authorization / regulatory approval from the home country regulatory authority (e.g., CE Certificate, US FDA 510(k)/PMA, TGA approval, or equivalent)
- Certificate of Free Sale (CFS) issued by the competent authority in the country of manufacture — apostilled or notarized as required
- ISO 13485:2016 certificate of the overseas manufacturing facility — must be from an accredited certification body with valid expiry date
- Full product list with model numbers, nomenclature, and intended use for each ventilator variant being registered
- Copy of the notarized Authorized Indian Agent appointment letter from the overseas manufacturer
Technical and Product Documents (per ventilator model)
- Technical Specifications and Design Dossier for each ventilator model
- Test report covering IS 13450 (Part 2/Sec 12): 2023 — from an NABL-accredited laboratory or a CDSCO-notified testing laboratory in India, or from an internationally accredited IEC CB scheme laboratory (subject to CDSCO acceptance)
- Test report covering IS 7655 compliance — separately referenced alongside IS 13450
- Test report covering IEC 60601-1-2 (electromagnetic compatibility)
- Risk Management File as per ISO 14971 — hazard analysis, FMEA, risk controls, and residual risk summary
- Essential Principles of Safety and Performance checklist (Schedule I of MDR 2017)
- Biocompatibility assessment for patient-contact components per ISO 10993 series
- Software validation documentation and cybersecurity risk assessment for software-controlled ventilators
- Clinical evaluation report or clinical data summary demonstrating safety and performance in clinical use
- Instructions for Use (IFU) and User Manual in English — and any regional Indian language version if required for end-user communication
- Label draft and packaging label as per Schedule VII of MDR 2017 — with UDI-compatible fields
- Post-Market Surveillance (PMS) and vigilance plan for the Indian market
Labelling Requirements Under MDR 2017 Schedule VII
- Every imported ventilator sold in India must carry a label compliant with Schedule VII of MDR 2017. The label must include:
- Name and address of the importer in India (not just the overseas manufacturer)
- Name and address of the overseas manufacturer
- Generic name and brand name of the device
- Model number and serial number or batch/lot number
- Date of manufacture and date of expiry or shelf life (where applicable)
- CDSCO import license number
- Country of origin
- 'Caution' statement if applicable — medical device for professional use only
- UDI — Unique Device Identifier fields (being phased in under 2026 guidelines)
Critical compliance point: Every ventilator carton and device must carry the Indian importer's name, address, and CDSCO license number on the label. Devices without the Indian importer's details on the label — even if the device itself has valid CE marking — cannot be legally sold in India.

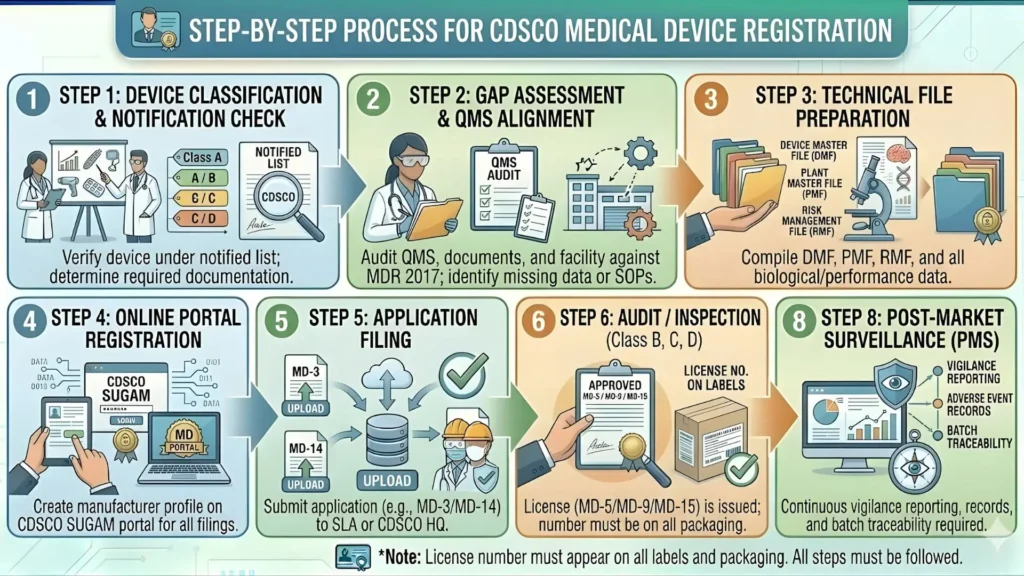
Step-by-Step Process: CDSCO Import License for Ventilators
Step 1: Confirm Device Classification
Before anything else, confirm that your product is a Class C ventilator under Schedule I of MDR 2017. If you are importing a combination device or a device with ancillary ventilation function, get a formal classification opinion from a qualified regulatory consultant. Misclassification is expensive to correct mid-process.
Step 2: Appoint an Authorized Indian Agent (AIA)
If the overseas manufacturer does not already have an AIA in India, this is the first practical step. The AIA is the legally accountable representative of the overseas manufacturer in India for CDSCO purposes. The AIA appointment must be documented through a formal notarised agreement specifying the scope of authority, duration, and responsibilities including adverse event reporting and recall cooperation.
Step 3: Register on the Sugam Portal
Both the Indian importer and the AIA (for the overseas manufacturer's MD-15 application) must register on the Sugam portal at sugam.gov.in. Create company-level accounts, verify email and mobile numbers, and complete profile setup. There is no offline or email-based application route for import licenses.
Step 4: Prepare the Complete Technical Dossier
Compile all documents listed in Section 8 before opening the application form. This includes IS 13450 (Part 2/Sec 12): 2023 test reports, IS 7655 test reports, CE certificate or equivalent, CFS, ISO 13485 certificate, AIA agreement, and Schedule VII-compliant label drafts. An incomplete dossier will generate queries and extend your timeline by weeks or months.
Step 5: Pay Application Fees
Application fees for both MD-14 and MD-15 are paid online through the Sugam portal payment gateway at the time of submission. Fees are non-refundable. Verify the current fee schedule on cdsco.gov.in before payment.
Step 6: Submit Form MD-14 and Form MD-15 on Sugam
Complete and submit both applications on the Sugam portal. Attach all documents in the required format and within specified file size limits. Record both application reference numbers — you will need them for tracking and all CDSCO correspondence.
Step 7: CDSCO Acknowledgement and Document Scrutiny
CDSCO issues automated acknowledgements on submission. Applications are then assigned to reviewing officers at CDSCO, New Delhi. Initial document scrutiny typically takes 15 to 30 working days. If documents are deficient or queries arise, CDSCO raises them on the Sugam portal and notifies you via email and SMS. Query responses must be submitted within the stipulated timeframe — typically 30 days — or the application is rejected.
Step 8: Technical Review of Product Dossier
For Class C ventilators, CDSCO conducts a detailed technical review of the product dossier — not merely a procedural document check. Reviewers assess the IS 13450 (Part 2/Sec 12): 2023 and IS 7655 test reports, risk management file, clinical evaluation data, software documentation, and label compliance. This is where incomplete or outdated BIS standard test reports are caught.
Step 9: Possible Site Inspection of Importer's Premises
For import licenses, CDSCO may — and increasingly does — conduct a site inspection of the Indian importer's registered premises and storage facility. During inspection, they assess storage conditions, document management practices, recall readiness, and the importer's awareness of post-market surveillance obligations. Be prepared.

Government Fee Structure for CDSCO Ventilator Import License
| Application Type | Form | Applicable Fee (INR) |
| Grant of import license (Indian importer) | Form MD-14 | ₹50,000 per manufacturer (for Class C devices) |
| Registration of overseas manufacturer | Form MD-15 | ₹50,000 per manufacturer |
| Renewal of import license | Renewal application | ₹25,000 |
| Amendment to import license | Amendment application | ₹10,000 |
| Duplicate import license | — | ₹5,000 |
| Additional product (added via amendment) | Form MD-7 equivalent | Per CDSCO notification |
Fees are non-refundable once paid on the Sugam portal. Always verify the current fee schedule at cdsco.gov.in before initiating payment, as the Government of India may revise fees through notification without individual communication to applicants.
Import License Validity and Renewal
CDSCO import licenses for ventilators (Form MD-16) are valid for 5 years from the date of issue. The overseas manufacturer's Certificate of Registration (from Form MD-15) is also valid for 5 years and must be renewed in parallel with the import license.
| Stage | Action Required | Timeline |
| 90 days before expiry | Start preparing renewal dossier — update ISO 13485 certificate, verify AIA agreement validity, check IS 13450 (Pt 2/Sec 12): 2023 test report currency | Recommended start date |
| 60 days before expiry | Submit renewal application with updated fee on Sugam portal | Ideal filing deadline |
| 30 days before expiry | Last safe date for submission to avoid import disruption during review | Minimum deadline |
| After expiry with active renewal application | Continued import may be permitted at CDSCO discretion during renewal review period | Subject to CDSCO discretion |
| After expiry without renewal application | All imports must cease immediately; any shipments in transit may be detained at customs | Zero tolerance enforcement |
An expired import license means CDSCO's system no longer recognizes your right to import. Customs authorities can and do cross-check CDSCO import license status for notified medical devices at the port of entry. Importing with an expired license — even if goods are in transit — constitutes a violation of MDR 2017.
Latest CDSCO Regulatory Updates for Ventilator Importers (2025–2026)
| Update | Details | What Importers Must Do |
| IS 13450 (Part 2/Sec 12): 2023 emphasis | CDSCO reviewers now specifically look for this standard in import dossiers alongside IS 7655 — applications without explicit IS 13450 alignment are generating queries | Obtain updated test reports from NABL or IEC CB accredited labs referencing IS 13450 (Part 2/Sec 12): 2023 explicitly before filing |
| Tightened PMSR timelines | Vigilance reporting obligations for Class C devices tightened; faster adverse event reporting required | Review and update your PMS and vigilance SOPs; train your regulatory team on new timelines |
| AIA accountability strengthened | CDSCO has issued guidance reinforcing that AIAs are jointly accountable for adverse event reporting, recalls, and PMS obligations in India | Ensure your AIA agreement is updated and the AIA has documented procedures for CDSCO compliance |
| UDI implementation | UDI framework being phased in for Class C and D devices; labelling and traceability requirements taking effect | Work with overseas manufacturer to add UDI codes to device labels; update inventory traceability systems |
| Increased import inspections | CDSCO has increased physical inspections of importers' premises, warehouses, and distribution facilities | Keep storage records, import records, and device traceability documents in permanent inspection-ready condition |
| CDSCO-GeM integration | Active CDSCO import license status is now cross-verified on the Government e-Marketplace portal | Ensure your MD-16 license status is current and accurately reflected on GeM for uninterrupted government tender participation |
| BIS dual-standard compliance | Both IS 13450 (Pt 2/Sec 12): 2023 and IS 7655 test reports now expected in the same dossier | Do not rely on a single standard's test report; ensure both are present and from accredited laboratories |
Benefits of Obtaining a CDSCO Import License for Ventilators
Beyond the obvious legal requirement, holding a valid CDSCO import license delivers tangible commercial and operational advantages:
- Legal market access — You can import, stock, and sell ventilators in India without risk of seizure, penalties, or criminal prosecution.
- Government tender eligibility — CDSCO import license is a mandatory eligibility criterion for GeM portal, CMSS, ESIC, CGHS, and state government procurement tenders for ventilators. Without it, you cannot participate.
- Hospital procurement preference — Procurement committees in accredited hospitals (NABH, JCI) require valid CDSCO licensing from suppliers as part of their vendor qualification process.
- Brand credibility — An active CDSCO license signals regulatory seriousness to hospital administrations and clinical teams who evaluate imported ventilator brands.
- Customs clearance — A valid MD-16 import license enables smooth customs clearance at Indian ports. Shipments without valid licensing face detention, demurrage costs, and possible destruction of goods.
- Investment and partnership readiness — Indian distributors and institutional buyers require verified CDSCO licensing before signing distribution or supply agreements with overseas ventilator brands.
Total Cost Estimation for CDSCO Ventilator Import License
| Cost Head | Estimated Range (INR) | Notes |
| Government Fee — Form MD-14 | ₹50,000 | Per overseas manufacturer; non-refundable |
| Government Fee — Form MD-15 | ₹50,000 | Per overseas manufacturer registration; non-refundable |
| IS 13450 (Pt 2/Sec 12): 2023 and IS 7655 Testing | ₹3,00,000 – ₹8,00,000 | NABL or IEC CB accredited laboratory; cost varies by number of models and test scope |
| Technical Documentation Preparation | ₹1,00,000 – ₹3,00,000 | Design dossier, risk file, IFU India adaptation; cost depends on in-house capability |
| Regulatory Consultant Fees | ₹1,50,000 – ₹5,00,000 | Strongly recommended for first-time applicants and for overseas manufacturer coordination |
| AIA Agreement Notarisation and Apostille | ₹30,000 – ₹1,50,000 | Depends on country of overseas manufacturer; apostille costs vary by jurisdiction |
| Label Artwork and Schedule VII Adaptation | ₹20,000 – ₹80,000 | Re-labelling for Indian regulatory compliance per model |
| Annual Returns and Ongoing Compliance | ₹30,000 – ₹1,00,000 per year | Regulatory consultant support for annual filings and query management |
| Total Estimated Investment (First Year) | ₹7,00,000 – ₹18,00,000 | Excludes physical inventory, import duties, and logistics costs |
Conclusion
The CDSCO import license process for ventilators is demanding — but it is designed to be. When a ventilator enters an Indian ICU, the clinical team trusts that it has met documented safety and performance standards, that someone in India is accountable for its post-market performance, and that there is a functioning system for reporting and recalling it if something goes wrong. The MD-14 and MD-15 licensing framework creates that accountability chain.
For importers, the key to a smooth process is preparation before submission — not after. That means having your IS 13450 (Part 2/Sec 12): 2023 test reports in hand, a properly executed AIA agreement, a valid CE certificate and CFS, a current ISO 13485 certificate from your overseas manufacturer, and a Schedule VII-compliant label draft ready before you log in to the Sugam portal.
For overseas manufacturers looking to enter the Indian ventilator market, understanding that India has its own BIS standards — IS 13450 (Part 2/Sec 12): 2023 and IS 7655 — and that these are not automatically satisfied by your existing CE or FDA documentation is the single most important insight this guide can offer. Commission IS 13450-specific testing early, appoint a capable and India-experienced AIA, and treat your Indian regulatory dossier as a standalone document, not a copy of your European or US submission.
India's ventilator market is growing, driven by expanding ICU capacity, increased critical care awareness, and aggressive government procurement programmes. The regulatory pathway is clear and well-documented. Compliance is the entry price, and once you are in, it is a market worth being in.
Frequently Asked Questions
What is the difference between Form MD-14 and Form MD-15?
Form MD-14 is filed by the Indian importer and results in the import license (Form MD-16) that authorizes the Indian entity to bring ventilators into India. Form MD-15 is filed by the overseas manufacturer through their Authorized Indian Agent and results in a Certificate of Registration for the foreign manufacturer. Both are required for legal commercial import of ventilators.
Can I import ventilators for demonstration or clinical evaluation without an import license?
For genuine clinical evaluation or investigational use, a separate import permission under Form MD-13 (for import of devices for investigation or clinical evaluation) may apply. This is a different and more restrictive track. Importing under this category for purposes other than genuine clinical investigation is a violation. For all commercial import including demos and trials ahead of commercial sale, an MD-14 import license is required
Which BIS standards apply to imported ventilators?
IS 13450 (Part 2/Sec 12): 2023 — the updated BIS standard aligned to ISO 80601-2-12 — and IS 7655 — aligned to IEC 60601-1 — are the primary BIS standards relevant to imported ventilators in India.
Do imported ventilators need BIS product registration separately?
BIS product registration under IS 13450 or IS 7655 is not a separately mandatory pre-condition for obtaining a CDSCO import license in 2026. However, for government supply contracts through GeM, CMSS, or state procurement, BIS product registration is frequently a mandatory tender eligibility criterion.
How long is a CDSCO ventilator import license valid?
The import license (Form MD-16) is valid for 5 years from the date of issue. The overseas manufacturer's Certificate of Registration (from MD-15) is also valid for 5 years. Apply for renewal of both at least 60 to 90 days before expiry to avoid any lapse in your authorisation to import.
Can one Indian importer hold licenses for multiple overseas ventilator manufacturers?
Yes. An Indian importer can hold separate MD-14 import licenses for multiple overseas manufacturers. Each manufacturer relationship requires a separate MD-14 application, a separate AIA agreement, and separate government fees.
What happens if my overseas manufacturer updates their product — say, a new software version?
Any significant change to the ventilator — including firmware updates that affect safety or performance parameters, hardware revisions, new alarm logic, or changes to patient-contact components — must be reported to CDSCO through a formal amendment application (Form MD-7 equivalent for importers).
Can an overseas manufacturer appoint multiple AIAs in India?
An overseas manufacturer can appoint one Authorised Indian Agent at a time for a specific product or product range. However, they can appoint different AIAs for different product lines or geographical territories — but each AIA relationship requires a separate, formally executed agreement
What happens if CDSCO issues an import alert against my ventilator brand?
A CDSCO import alert effectively suspends the import license for the affected product. All shipments in transit may be detained at Indian customs, and existing stock may be subject to recall. The importer and AIA are jointly responsible for coordinating with CDSCO and the overseas manufacturer to address the alert.
Is an overseas manufacturer's CE mark or FDA clearance sufficient for CDSCO import approval?
CE marking or US FDA clearance is necessary but not sufficient for CDSCO import approval. These approvals are considered strong supporting evidence of product safety and quality and are required as part of the MD-15 dossier.





































