- In-Vitro Diagnostic Devices (IVDs) follow a separate, dedicated classification track under MDR 2017 — distinct from general medical devices — with Class A through D based on risk and intended use.
- Every IVD importer in India must apply for an import license using Form MD-14, but IVDs require additional product-specific documents like performance evaluation data, clinical sensitivity/specificity reports, and reference material details.
- The Form MD-14 process for IVDs differs from general medical devices in its documentation depth, testing requirements, and the regulatory scrutiny applied — especially for HIV, hepatitis, and blood screening kits (Class D IVDs).
- As of 2026, all four classes of IVDs are under mandatory licensing — skipping or delaying the process can result in customs holds, financial penalties, and reputational damage with Indian hospital and laboratory buyers.
Introduction
If you are an importer of In-Vitro Diagnostic Devices — ranging from blood glucose meters and pregnancy test kits to PCR diagnostics and HIV screening assays — you already know that India is one of the fastest-growing diagnostic markets in the world. But with that growth comes a very structured regulatory environment that you cannot afford to navigate casually.
Most importers are broadly aware of the CDSCO import license process under Form MD-14. What many do not realize, however, is that IVDs are not simply lumped into the general medical device category. They have their own dedicated classification system, their own risk parameters, and their own set of documentation requirements that differ in meaningful ways from non-IVD medical devices.


This guide is specifically designed for importers, regulatory affairs professionals, and authorized Indian agents dealing with IVD import license India requirements in 2026. We cover everything from the ground up — what IVDs are, how they are classified under MDR 2017, what Form MD-14 requires specifically for IVDs, and how the process differs from importing general medical devices.
Whether you are bringing in rapid antigen test kits, clinical chemistry analyzers, molecular diagnostic systems, or point-of-care devices, this guide gives you a clear and accurate picture of what the Indian regulatory framework demands.
What Are In-Vitro Diagnostic Devices (IVDs)?
In-Vitro Diagnostic Devices (IVDs) are medical devices — reagents, kits, instruments, systems, or software — intended to examine specimens derived from the human body for the purpose of diagnosis, monitoring, treatment decisions, or screening of disease.
The phrase 'in-vitro' literally means 'in glass' — referring to tests performed outside the body, on samples like blood, urine, saliva, tissue, or other biological fluids. This is what distinguishes IVDs from implants or surgical instruments.
| Common IVD Categories |
| Blood glucose monitoring systems (glucometers + test strips)Pregnancy, ovulation test kits HIV, Hepatitis B, Hepatitis C, syphilis rapid test kits PCR, molecular diagnostic kits (including COVID-19 and respiratory panels) Clinical chemistry, hematology analyzers Tumor markers, oncology diagnostics Thyroid function, cardiac biomarker, hormone assay kits Drug of abuse screening kits Urine analyzers, dipstick test strips Microbiological culture media and identification systems. |
IVDs touch almost every branch of healthcare — from primary health centers using simple rapid tests to tertiary hospitals running high-throughput automated analyzers. That breadth, combined with the direct impact these devices have on patient diagnosis and treatment, is precisely why India's MDR 2017 gives them their own classification framework.
Regulatory Framework for IVDs in India
IVDs were officially brought under the Medical Devices Rules, 2017 (MDR 2017) — notified under the Drugs and Cosmetics Act, 1940. Before MDR 2017, only a limited list of IVDs (like HIV and Hepatitis kits) were regulated. The 2017 rules expanded coverage significantly, and subsequent notifications in 2020 and 2022 brought additional IVD categories into the fold.
| Regulatory Element | Details |
| Governing Act | Drugs and Cosmetics Act, 1940 |
| Applicable Rules | Medical Devices Rules, 2017 (MDR 2017) |
| IVD-Specific Schedule | Schedule II-C (IVD Classification under MDR 2017) |
| Regulatory Authority | CDSCO — Central Drugs Standard Control Organisation |
| Licensing Authority | Central Licensing Authority (CLA) / DCGI for Class B, C, D IVDs |
| Application Form | Form MD-14 (Import License Application) |
| License Issued In | Form MD-15 |
| Portal for Application | SUGAM (sugam.mohfw.gov.in) |
| Key Amendments | MDR Amendment 2020, 2022 notifications expanding IVD coverage |
IVD Classification Under MDR 2017: Class A Through D
This is where IVDs diverge from general medical devices. MDR 2017 uses Schedule II-C to classify IVDs into four risk-based classes. Unlike the classification of general devices, IVD classification is driven by factors like the consequences of incorrect results, the intended user (professional vs. self-test), the analyte being detected, and whether the device is used for life-threatening disease screening.
IVD Class A — Low Individual Risk, Low Public Health Risk
These are general-purpose laboratory instruments and reagents that pose low individual and public health risk. Errors in results have limited direct clinical impact.
| Criterion | Details |
| Risk Profile | Low individual and low public health risk |
| Regulatory Scrutiny | Lowest — declaration of conformity by manufacturer |
| Examples | General laboratory instruments, culture media (non-diagnostic specific), specimen containers, collection tubes (general purpose), lab staining solutions, distilled water for diagnostic use |
| Intended User | Primarily trained laboratory professionals |
| Self-Test Permitted | Generally not applicable |
IVD Class B — Low Individual Risk, Moderate Public Health Risk
These IVDs have a moderate potential for public health impact, often because they detect conditions that — if missed — could lead to wider community spread or delayed treatment.
| Criterion | Details |
| Risk Profile | Low individual risk, moderate public health risk |
| Regulatory Scrutiny | Moderate — requires technical documentation and performance data |
| Examples | Pregnancy test kits, urinalysis test strips (professional use), general haematology analyzers, biochemistry analyzers (non-critical analytes), blood grouping (non-ABO/Rh), anti-nuclear antibody tests |
| Intended User | Primarily trained professionals; some OTC variants |
| Self-Test Permitted | Some sub-categories (e.g., pregnancy kits in OTC format) |
IVD Class C — High Individual Risk, Moderate-High Public Health Risk
These are diagnostics for serious conditions where a wrong result — false negative or false positive — can have significant clinical consequences for the individual patient.
| Criterion | Details |
| Risk Profile | High individual risk, moderate-high public health risk |
| Regulatory Scrutiny | High — performance evaluation, clinical validation, possible audit |
| Examples | Blood glucose monitoring systems (self-test), PSA tests, HbA1c testing kits, thyroid function tests, cardiac biomarker kits (troponin, BNP), coagulation analyzers, tumor marker assays, drug of abuse screening (self-test), fertility hormone assays |
| Intended User | Professional and self-test (home use) categories |
| Self-Test Permitted | Yes — specific performance criteria required for home-use versions |
IVD Class D — Highest Individual and Public Health Risk
Class D IVDs represent the most critical category. These are devices used to screen blood for transfusion-transmitted infections, detect life-threatening diseases where a missed diagnosis has irreversible consequences, or where the disease has high epidemic potential. CDSCO applies the strictest regulatory scrutiny to this class.
| Criterion | Details |
| Risk Profile | Highest individual and public health risk |
| Regulatory Scrutiny | Strictest — full technical file, clinical data, third-party testing, site audit |
| Examples | HIV 1 and 2 diagnostic kits, Hepatitis B surface antigen (HBsAg) detection kits, Hepatitis C antibody tests, HTLV I and II, West Nile Virus tests, ABO blood grouping for transfusion, Rh typing for transfusion, compatibility testing for blood transfusion, malaria detection kits (transfusion screening), CD4 counting kits |
| Intended User | Exclusively trained medical / laboratory professionals |
| Self-Test Permitted | No — professional use only |
| Why Class D IVD Compliance is Non-Negotiable |
| An incorrect result from a Class D IVD — such as a missed HIV or Hepatitis B infection in donated blood — can have catastrophic consequences for transfusion recipients. CDSCO mandates third-party performance evaluation by a government-approved or accredited testing laboratory for Class D IVDs before the import license is granted. Any attempt to import Class D IVDs without a valid import license is treated as a serious regulatory offense under the Drugs and Cosmetics Act. |
How IVD Classification Differs from General Medical Device Classification
This is a point of confusion for many regulatory professionals, especially those transitioning from general medical device work into IVD regulation. The classification logic is fundamentally different:
| Classification Factor | General Medical Devices | IVDs |
| Primary Risk Driver | Physical interaction with body (contact duration, invasiveness) | Consequence of erroneous diagnostic result + disease severity |
| Schedule Reference in MDR 2017 | Schedule II-A and II-B | Schedule II-C (dedicated IVD schedule) |
| Self-Test Consideration | Generally not a primary classification factor | Critical factor — home-use IVDs have stricter performance requirements |
| Public Health Risk Weight | Lower weight in classification | High weight — especially for epidemic-potential diseases |
| Software Considerations | IEC 62304, SOUP consideration | IVD software classified separately under companion diagnostic rules |
| Testing Laboratory Involvement | Not always mandatory for lower classes | Third-party lab testing mandatory for Class D IVDs |
| Clinical Data Requirement | Required for Class C and D | Required from Class C onwards; Class D requires extensive clinical validation |
Form MD-14 for IVD Import: How It Works
All IVD importers in India must submit Form MD-14 to CDSCO to obtain the import license (issued in Form MD-15). The basic framework is the same as for general medical devices — you apply via the SUGAM portal, pay the applicable fee, and await CDSCO review. But what goes inside the application is where IVDs diverge significantly.
The depth of documentation, the performance data requirements, and the specific technical standards that apply to IVDs make their Form MD-14 dossier considerably more complex than a typical general medical device application. Let us walk through it step by step.
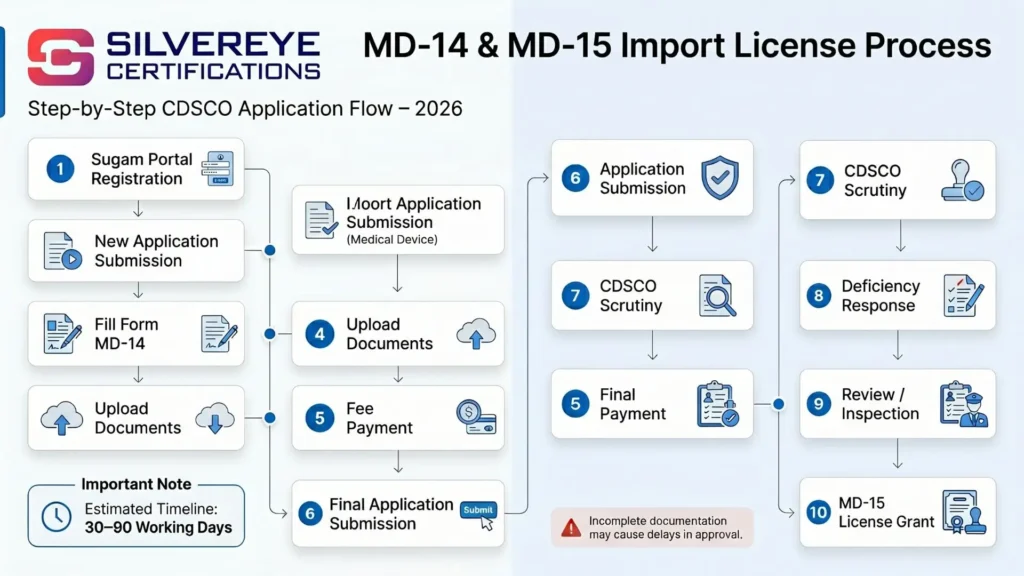
Step-by-Step Form MD-14 Process for IVD Importers
| Step | Action | IVD-Specific Notes |
| 1 | Classify the IVD under Schedule II-C of MDR 2017 | Check if device is an instrument, reagent, kit, or system — each may classify differently |
| 2 | Verify if already listed as notified IVD | CDSCO maintains a list of specifically notified IVDs — confirm current status on cdsco.gov.in |
| 3 | Appoint Authorized Indian Agent (AIA) | AIA must be a resident Indian entity with a written, apostilled agreement from the foreign manufacturer |
| 4 | Prepare the Technical Dossier | IVDs require performance evaluation data, clinical validation, reference materials — see detailed list below |
| 5 | Get third-party performance testing done (Class D) | Testing by CDSCO-approved or accredited laboratory — essential before submission for Class D |
| 6 | Register and log in to SUGAM portal | sugam.mohfw.gov.in — create/use existing account |
| 7 | Fill and submit Form MD-14 online | Select correct IVD category; upload all documents in PDF format |
| 8 | Pay the prescribed government fee | Based on device class — non-refundable |
| 9 | Respond to CDSCO deficiency/query letters | IVD applications often receive technical queries on performance data — respond within given timeline |
| 10 | Manufacturing site inspection (if required) | Mandatory for Class D; possible for Class C depending on device type and risk profile |
| 11 | Receive Import License in Form MD-15 | Valid for 3 years from date of grant |

Documents Required for Form MD-14: IVD-Specific Requirements
IVD applications share some common documents with general medical devices, but also require a distinct set of IVD-specific technical and clinical documents. Here is a comprehensive breakdown:
Section A: Standard Documents (Common with General Medical Devices)
| Document | Notes |
| Duly filled Form MD-14 | Signed by authorized signatory; IVD product category clearly indicated |
| Covering letter on applicant letterhead | Addressed to Central Licensing Authority, CDSCO |
| Proof of Indian entity registration | Company incorporation / partnership deed / proprietorship certificate |
| State Drug License / Medical Device License | For importer's premises — storage and import authorization |
| Technically Qualified Person (TQP) credentials | Pharmacy graduate / medical graduate / relevant science degree required |
| Authorization letter from foreign manufacturer | In prescribed format; apostilled; must specifically mention the IVD product(s) |
| Certificate of Incorporation of foreign manufacturer | Registered in country of origin |
| Free Sale Certificate (FSC) | Issued by competent authority in manufacturing country — confirming legal market authorization |
| ISO 13485 Certificate / GMP Certificate | Current and valid manufacturing quality certificate |
Section B: IVD-Specific Technical Documents
| Document | Class Applicability | Notes |
| Performance Evaluation Report | A, B, C, D | Analytical performance: sensitivity, specificity, precision, accuracy, linearity, measuring range |
| Clinical Validation / Clinical Sensitivity & Specificity Data | C and D (mandatory); B (recommended) | Data from clinical specimens — must meet CDSCO minimum performance standards |
| Reference Materials / Calibrators Documentation | B, C, D | Details of reference standards, WHO reference preparations used for calibration |
| Intended Use and Indications Statement | All classes | Detailed, unambiguous statement — includes analyte, matrix, clinical purpose |
| Summary of Safety and Performance (SSP) | C and D | Structured summary of device performance, clinical evidence, and risk management |
| Risk Management File Summary | B, C, D | Per ISO 14971 — residual risk acceptability statement |
| Instructions for Use (IFU) — Draft Indian Labeling | All classes | Must comply with MDR 2017 Schedule IV; language requirements |
| Analytical Interference Data | B, C, D | Documented testing for common interferences (hemolysis, lipemia, icterus, etc.) |
| Stability Data | All classes | Shelf life data under specified storage conditions; in-use stability for opened reagents |
| Cut-off Determination Data (for qualitative tests) | B, C, D | How positive/negative cut-off was established; statistical justification |
| Waived / POC Testing Validation (if applicable) | C (self-test) | Additional data required if device is intended for home / point-of-care use |
| Third-Party Performance Test Report from CDSCO-approved lab | D (mandatory) | Separate from manufacturer's data — independent laboratory confirmation |
Section C: Market Authorization Documents
| Document | Notes |
| CE-IVD Certificate (EU IVDR or IVDD) / US FDA 510(k) or PMA / TGA / Health Canada approval | Market authorization from reference regulatory authority — at least one major market clearance strongly preferred |
| Technical File Index | Summary list of technical file contents — not the full file, but an index demonstrating its existence |
| Declaration of Conformity | Manufacturer's declaration that the IVD conforms to applicable standards and MDR 2017 requirements |
| CE-IVD Notified Body Certificate (if applicable) | For devices requiring Notified Body review under EU IVDR — especially Class C and D equivalents |


| Important Note on EU IVDR vs IVDD |
| The European Union transitioned from the In Vitro Diagnostic Directive (IVDD, 98/79/EC) to the In Vitro Diagnostic Regulation (IVDR, EU 2017/746) with full enforcement from May 2022.CDSCO has started recognizing EU IVDR certificates. However, during the IVDR transition period, some devices still carry IVDD certificates. Always check the current CDSCO guidance on which EU market authorization documents are accepted — this is a live area of regulatory development. |
Form MD-14 for IVDs vs General Medical Devices: Key Differences
This is the section most importers need most urgently. If you have previously handled Form MD-14 for general medical devices and are now moving into IVDs, here is a precise comparison of where the two tracks diverge:
| Comparison Point | General Medical Devices | IVDs |
| Classification Schedule | Schedule II-A / II-B of MDR 2017 | Schedule II-C of MDR 2017 (dedicated IVD schedule) |
| Primary Risk Factor in Classification | Nature and duration of body contact, invasiveness | Severity of disease detected, consequence of wrong result, public health impact |
| Performance Evaluation Data | Not universally required for all classes | Required for ALL classes from A to D; depth increases with class |
| Clinical Sensitivity/Specificity Data | Required only for Class C and D (therapeutic devices) | Required for Class C and D IVDs; strongly recommended for Class B |
| Third-Party Lab Testing Requirement | Rare — case-by-case basis | Mandatory for Class D IVDs before application is accepted |
| Reference Material Documentation | Not applicable | Required — IVDs must reference WHO or national reference preparations where available |
| Self-Test / Home-Use Classification Impact | Minimal impact on risk class | Moves device to higher scrutiny class; additional performance validation needed |
| Analytical Interference Documentation | Relevant mainly for devices with cross-reactions | Mandatory for most IVDs — hemolysis, lipemia, drugs, high-dose hook effect |
| Cut-off Validation Data | Not applicable | Required for qualitative IVDs — statistical methodology must be documented |
| In-Use Stability Data | Storage conditions data sufficient | Separate in-use stability testing required — how long opened reagent remains valid |
| Companion Diagnostic (CDx) Consideration | Not applicable to most devices | Companion diagnostics linked to specific therapeutics have additional CDSCO requirements |
| Post-Market Surveillance Requirements | Required for Class C and D | More intensive for IVDs — especially Class D; batch testing records mandatory |
Eligibility Criteria for IVD Import License via Form MD-14
The eligibility requirements for IVD import mirror the general Form MD-14 criteria, but with some additional layers specific to diagnostics:
For the Foreign IVD Manufacturer
- Must be a legally registered manufacturer in the country of origin.
- ISO 13485 certification is expected; for Class D IVDs, compliance with ISO 15189 (medical laboratory quality) principles in manufacturing is an advantage.
- Must have existing market authorization (CE-IVD, FDA clearance, or equivalent) — especially for Class C and D IVDs.
- Must authorize an Indian agent in writing, with specific mention of the IVD products and scope of authorization.
- Must be prepared for manufacturing site inspection for Class C and D IVDs — CDSCO or its authorized inspection bodies may conduct on-site or remote audits.
For the Authorized Indian Agent / Importer
- Must be a registered Indian entity — company, LLP, or individual with valid legal status.
- Must hold a valid Drug License / Medical Device License from the State Licensing Authority for the import and storage premises.
- Storage premises must meet temperature and environmental conditions required for IVDs — particularly for cold-chain products (2°C–8°C or -20°C storage).
- Must appoint a Technically Qualified Person (TQP) — typically a pharmacy graduate, medical graduate, or life sciences graduate with relevant experience.
- Must have cold chain infrastructure documented if the IVD requires refrigerated or frozen storage and transport.
- Must be capable of maintaining post-market surveillance records and filing adverse event reports as required for the IVD class.
Government Fees for IVD Import License (Form MD-14) — 2026
The fee structure for IVD import license applications follows the same Schedule VII of MDR 2017 that applies to general medical devices. However, fees are applied per device/product application, and IVDs — especially Class C and D — often involve multiple products, making total cost a significant planning factor.
| IVD Class | Application Fee (per product) | Key Notes |
| Class A IVD | Rs. 5,000 per device | Lowest fee bracket; general lab instruments and basic reagents |
| Class B IVD | Rs. 10,000 per device | Moderate fee; pregnancy kits, urinalysis, general hematology |
| Class C IVD | Rs. 25,000 per device | High fee; glucometers, cardiac markers, hormone assays |
| Class D IVD | Rs. 50,000 per device | Highest fee; HIV kits, blood screening assays, transfusion testing |
| License Renewal | Approx. 50% of original fee | Check latest government gazette notification |
| Amendment to License | Varies by amendment type | Consult CDSCO circular for current amendment fee schedule |
| Fee Planning Tip for IVD Importers |
| If you are importing a diagnostic analyzer along with its reagent kits, each product line (instrument, individual reagent kit, calibrator, control) may require a separate Form MD-14 application with its own fee. Plan your application strategy carefully — grouping instrument systems with their reagent families is sometimes possible, but depends on how CDSCO classifies the system vs. its accessories. All fees are paid online through SUGAM and are non-refundable, even if the application is rejected or withdrawn. |
Validity and Renewal of IVD Import License
The import license for IVDs (issued in Form MD-15) is valid for 3 years from the date of grant — same as general medical devices. However, IVD-specific renewal considerations make early action even more important.
| Parameter | Details |
| License Validity | 3 years from date of issue |
| Recommended Renewal Start | 3 to 6 months before expiry |
| Critical Documents to Renew | ISO 13485/GMP certificate, Free Sale Certificate, CE-IVD certificate (check for IVDR transition status), Authorization letter from manufacturer |
| Third-Party Test Report (Class D) | May need to be updated at renewal if original testing is more than 3 years old |
| Cold Chain Compliance Review | Re-verify that storage and transport infrastructure still meets IVD temperature requirements |
| Impact of Expired License | Cannot legally import; customs may hold or reject consignments |
| Post-Expiry Re-application | Treated as fresh application — significantly longer timeline than timely renewal |
| IVD Renewal Watch-Out |
| For Class D IVDs in particular — the technical standards and performance benchmarks can evolve. At renewal, CDSCO may ask for updated performance data if significant regulatory updates have occurred since your original license was granted. The EU IVDR classification has reclassified several IVDs upward in risk class compared to IVDD. If your device's EU classification has changed since the original license, your renewal strategy may need to reflect this change in your Indian application as well. |
Post-Approval Compliance for IVD Importers
Holding a valid IVD import license is not a one-time achievement. Indian regulations — and global best practice — require ongoing compliance that active importers must manage systematically:
- Batch Release Records: Every imported batch of IVDs must have complete documentation — lot number, manufacturing date, expiry date, quantity imported, and certificate of analysis from the manufacturer.
- Quality Control Testing at Importation: For Class D IVDs, CDSCO may require testing of imported batches at government-approved laboratories before distribution — do not plan to sell immediately upon arrival.
- Adverse Event Reporting: If a distributed IVD causes or is suspected to have contributed to an incorrect diagnosis leading to patient harm, this must be reported to CDSCO within prescribed timelines.
- Field Safety Corrective Actions (FSCA): Any recall, market withdrawal, or field safety notice issued by the manufacturer globally must be mirrored with appropriate action in India and reported to CDSCO.
- Labeling Compliance: Every IVD pack must carry Indian-market labeling compliant with MDR 2017 Schedule IV — including instructions in English (and Hindi where required), storage conditions, lot number, and importer details.
- Cold Chain Documentation: For temperature-sensitive IVDs, maintain temperature records throughout the storage and distribution chain. Breaks in cold chain must be documented and products evaluated before further distribution.
- Annual Returns: Depending on licensing conditions, annual returns detailing import quantities, sales data, and adverse event summary may need to be filed with CDSCO.
- License Amendment for Changes: Changes to product design, labeling, manufacturing site, storage conditions, or importer details require a license amendment before implementation.
Special IVD Categories with Additional Regulatory Requirements
Some IVDs go beyond the standard Form MD-14 framework due to their specific nature, intended use, or public health significance:
1. HIV and Blood Screening IVDs
HIV and blood-screening IVDs are Class D and among the most tightly regulated. In addition to the standard Form MD-14 dossier, CDSCO has historically required:
- Prior approval under specific government notification for HIV diagnostic kits.
- Performance evaluation against Indian epidemiology — the device must detect the HIV strains prevalent in the Indian population.
- In some cases, approval from the National AIDS Control Organization (NACO) technical committee in addition to CDSCO clearance.
2. Point-of-Care (POC) and Self-Test IVDs
POC diagnostics and self-test IVDs — like home glucose monitors, home pregnancy tests, and OTC rapid tests — fall under Class C or higher and require additional documentation:
- Usability studies demonstrating that lay users (not laboratory professionals) can operate the device correctly without errors.
- Labeling specifically designed for non-expert users — with warnings, failure mode messaging, and referral recommendations.
- Performance data under conditions of actual home use (environmental variation, user error tolerance).
3. Companion Diagnostics (CDx)
A Companion Diagnostic is an IVD used to identify patients likely to benefit from (or at risk from) a specific therapeutic. CDSCO has been aligning with global CDx frameworks and the requirements include:
- Formal CDx designation and co-development relationship with the linked therapeutic device or drug.
- Clinical validation data specifically generated in the context of the linked treatment decision.
- Coordination between the CDx import application and the corresponding drug or biologic regulatory pathway in India.
4. Nucleic Acid Testing (NAT) and Molecular Diagnostics
PCR kits, RT-PCR systems, and other NAT-based diagnostics — especially those used for blood bank screening or infectious disease panels — face additional requirements:
- Detailed validation data covering sensitivity of detection (limits of detection), specificity across genotypes, and cross-reactivity panel testing.
- Instrument-reagent system compatibility documentation if the kit is designed for a specific platform.
- Labeling requirements specific to NAT systems, including contamination prevention warnings.
Common Mistakes IVD Importers Make in the Form MD-14 Process
Learning from others' experience can save months of delay. These are the most frequently seen issues in IVD import license applications:
- Wrong IVD Classification: Classifying under the general medical device schedule instead of Schedule II-C, or classifying at a lower risk class than warranted — CDSCO will flag this and may return the application.
- Missing Performance Evaluation Data: Submitting a dossier with only CE or FDA approval certificates but without standalone performance evaluation reports — approval certificates are supporting documents, not substitutes for performance data.
- Outdated Clinical Data: Submitting clinical validation data that is more than 5 years old without a contemporaneous performance update — CDSCO may question the current relevance of the data.
- No Third-Party Lab Report for Class D: Attempting to submit a Class D IVD application without the independent performance test report from a CDSCO-approved laboratory — this is a non-starter.
- Inadequate Indian Labeling: Draft Indian labels that do not include all Schedule IV mandatory elements, or that are direct translations of English labels without proper localization for the Indian market.
- Cold Chain Gap in Premises: Declaring storage premises for refrigerated or frozen IVDs without verified cold chain infrastructure documentation — CDSCO inspectors and state licensing authorities check this.
- Failing to Differentiate Reagent Kits from Instruments: Treating an analyzer system and its consumable reagent kits as a single product in the application — they often require separate applications.
SUGAM Portal: Practical Guide for IVD Importers
| Action | Portal Path / Guidance |
| Create New Account | Register as 'Importer' entity type — use company PAN or CIN for identity verification |
| Select Application Type | Choose 'Import License' > 'Medical Devices' > Select IVD category correctly |
| Document Upload Format | PDF — individual file size usually capped at 10–25MB; compress large documents |
| Fee Payment | Integrated payment gateway — net banking, NEFT, debit/credit card accepted |
| Track Application Status | SUGAM dashboard shows real-time status: Submitted → Under Review → Deficiency → Approved |
| Respond to Deficiency | Log in, navigate to your application, attach responses and revised documents — do not create a new application |
| Download Approved License | Form MD-15 is available as a digitally signed PDF on the SUGAM portal post-approval |
| Renewal Application | Navigate to existing license record > select Renewal > update/reupload documents as required |
Conclusion: Getting Your IVD Import Strategy Right for 2026
India's IVD import license framework under MDR 2017 is thorough, and for good reason. Diagnostic devices directly influence clinical decisions. A missed HIV diagnosis from a substandard test kit or an incorrect blood glucose reading in a diabetic patient can have serious, sometimes irreversible consequences. The regulatory framework exists to prevent exactly this.
For importers, the key takeaway is this: IVDs are not a sub-category of general medical devices — they are a parallel track with their own classification logic, their own documentation depth, and their own performance verification standards. Treating an IVD import application like a routine medical device application is the fastest path to rejection.
Whether you are importing rapid antigen kits, high-throughput clinical chemistry systems, or life-saving blood screening assays, approaching the Form MD-14 IVD import process with the right preparation — complete documentation, correctly classified devices, verified cold chain infrastructure, and realistic timelines — is what separates importers who grow in the Indian market from those stuck in regulatory limbo.
Start early, classify correctly, build a complete dossier, and partner with regulatory experts who understand both the technical and procedural nuances of CDSCO's IVD licensing framework. That is the surest way to secure your import license and build a sustainable diagnostics business in India.
Frequently Asked Questions
Are all IVDs required to have an import license in India?
As of 2026, all four classes of IVDs (Class A through D) are under mandatory import licensing. The phased implementation under MDR 2017 has been completed, meaning there is no longer a category of IVDs that can be imported without a valid Form MD-14 import license.
What is Schedule II-C under MDR 2017 and why does it matter?
Schedule II-C is the dedicated IVD classification schedule under the Medical Devices Rules, 2017. It defines the four risk classes (A, B, C, D) specifically for IVDs using criteria unique to in-vitro diagnostics — such as disease severity, consequence of incorrect results, and public health risk.
Does a CE-IVD or FDA clearance eliminate the need for Indian clinical data?
No. CE-IVD, FDA clearance, or other international approvals are important supporting documents but do not replace the requirement for clinical performance data as part of the Form MD-14 dossier.
Who can conduct the third-party performance testing required for Class D IVDs?
The independent performance testing for Class D IVDs must be conducted by a laboratory approved or recognized by CDSCO, or by a NABL-accredited laboratory that meets CDSCO's technical requirements.
Can one Form MD-14 application cover an IVD analyzer and all its reagent kits?
Generally, no. An IVD analyzer (the instrument) and its reagent kits are typically treated as distinct products by CDSCO, especially if the reagents have different risk classifications or different intended uses.
Is cold chain infrastructure mandatory for all IVD import license applications?
Not for all IVDs, but mandatory for those with specific temperature requirements. If your IVD requires storage at 2°C–8°C (refrigerated), -20°C (frozen), or other controlled temperatures, your import premises must be equipped accordingly and documented as part of the Form MD-14 application.
Can a foreign manufacturer apply directly for IVD registration in India without an Indian agent?
Yes — this is done via Form MD-41 (application for registration certificate), which is a separate route from Form MD-14. Under the MD-41 route, the foreign manufacturer applies directly under their own name for a Registration Certificate (Form MD-42).
How long does it take to get an IVD import license in India?
Timelines vary by class. Class A and B IVDs typically see approvals in 3 to 6 months under normal circumstances. Class C IVDs can take 6 to 9 months, sometimes longer. Class D IVDs — with third-party testing requirements and possible manufacturing site inspections — often take 9 to 18 months or more
What is the post-market batch testing requirement for imported Class D IVDs?
For Class D IVDs such as HIV and Hepatitis kits, CDSCO may require batch-wise testing of imported consignments at approved government laboratories before the product can be distributed.






































