- Form MD-14 For Medical Device Import is the official CDSCO application form required to obtain an import license for medical devices in India.
- It applies to all foreign manufacturers and their authorized Indian agents importing Class A, B, C, and D medical devices.
- The license issued under Form MD-14 is valid for 3 years from the date of grant and must be renewed before expiry.
- Non-compliance with the MD-14 process can result in shipment rejection, fines, or a ban on future imports.
Introduction
If you are planning to import medical devices into India, you have probably come across the term Form MD-14 at some point. And if you are confused about what it actually is, what it covers, and how to apply for it, you are not alone. Many importers, distributors, and even regulatory professionals find the process a bit overwhelming the first time around.
India's medical device regulatory landscape changed significantly after the Medical Devices Rules, 2017 (MDR 2017) came into force. Before that, the rules were scattered, and enforcement was inconsistent. After MDR 2017, every medical device — whether manufactured domestically or imported from abroad — requires proper regulatory approval before it can enter the Indian market.


Form MD-14 sits at the center of this framework for importers. Think of it as the gateway application form that sets the entire import licensing process in motion. Without it, your device cannot legally be imported, stored, sold, or distributed in India.
This guide covers everything you need to know about Form MD-14 — from what it is and who needs it, to the documents required, fees involved, step-by-step process, validity, renewal, and the most commonly asked questions from real importers.
What is Form MD-14?
Form MD-14 is the prescribed application form under the Medical Devices Rules, 2017 used to apply for an import license for medical devices in India. It is submitted to the Central Drugs Standard Control Organization (CDSCO), which is the national regulatory authority for drugs and medical devices under the Ministry of Health and Family Welfare, Government of India.
In simpler terms, if a foreign manufacturer or an authorized Indian importer wants to bring a medical device into India and sell it commercially, they must fill out and submit Form MD-14 to get the necessary import license.
Once approved, CDSCO issues the license in Form MD-15, which is the actual Import License certificate. But Form MD-14 is the starting point — it is the application that triggers the review, evaluation, and approval process.
| Important Clarification |
| Form MD-14 = Application form for import license (what you submit).Form MD-15 = Import License certificate (what you receive after approval).These two are closely linked but are not the same document. This distinction matters when you are tracking your application status or renewing your license. |
Legal Basis and Regulatory Authority
The requirement for Form MD-14 is grounded in the Medical Devices Rules, 2017, which were notified under the Drugs and Cosmetics Act, 1940. These rules regulate the import, manufacture, sale, and distribution of medical devices in India.
| Regulatory Element | Details |
| Governing Act | Drugs and Cosmetics Act, 1940 |
| Applicable Rules | Medical Devices Rules, 2017 (MDR 2017) |
| Regulatory Authority | CDSCO (Central Drugs Standard Control Organisation) |
| Licensing Authority | Central Licensing Authority (CLA) — DCGI |
| Form Name | Form MD-14 (Application for Import License) |
| License Issued In | Form MD-15 |
| Applicable Classes | Class A, B, C, and D Medical Devices |
| Last Updated Framework | MDR Amendment 2020 and subsequent notifications |
Who Needs to Submit Form MD-14?
Not everyone involved in the medical device supply chain needs to file Form MD-14. It applies specifically to:
- Foreign manufacturers who want to import their medical devices into India for commercial sale.
- Authorized Indian agents or importers who are officially appointed by the foreign manufacturer to handle the import process on their behalf.
- Companies importing medical devices for re-labeling, repacking, or local distribution under their own brand name.
- Importers dealing in notified medical devices that require mandatory registration or licensing under CDSCO regulations.
It is important to note that Class A and Class B medical devices (lower risk categories) were gradually brought under the mandatory licensing framework through phased implementation. As of 2026, all four classes — A, B, C, and D — require proper import licensing via Form MD-14 for import into India.
Understanding Medical Device Classification in India
Before diving into the Form MD-14 process, it helps to understand how India classifies medical devices. The risk-based classification system under MDR 2017 categorizes devices into four classes:
| Class | Risk Level | Examples | Regulatory Scrutiny |
| Class A | Low Risk | Tongue depressors, bandages, examination gloves | Least — Self-certification possible |
| Class B | Low-Moderate Risk | Hypodermic needles, suction equipment, basic diagnostic kits | Moderate |
| Class C | Moderate-High Risk | Lung ventilators, bone fixation plates, diagnostic imaging equipment | High |
| Class D | Highest Risk | Heart valves, implantable defibrillators, HIV diagnostic kits | Strictest |


Higher-class devices undergo more rigorous scrutiny, which means the Form MD-14 process for Class C and D devices involves more documentation, clinical data, and longer review timelines compared to Class A or B.
Key Benefits of Obtaining an Import License Through Form MD-14
Many importers view the MD-14 process as a compliance burden. But there is a flip side to it — having a valid import license actually works in your favor in several meaningful ways:
- Legal Market Access: A valid license allows uninterrupted importation and commercial sale of your medical device across India without risk of customs detention or seizure.
- Builds Buyer Trust: Hospitals, distributors, and procurement committees in India increasingly ask for regulatory proof before purchasing imported devices. An MD-15 license (obtained via MD-14) is that proof.
- Protects Against Counterfeits: CDSCO registration creates an official record of your device's specifications, reducing the risk of counterfeit products entering the market under your brand.
- Government Tender Eligibility: Many central and state government procurement tenders require proof of valid CDSCO licensing. Without it, you cannot bid.
- International Credibility: Regulatory approval from CDSCO, a recognized authority, adds to the device's global compliance credentials.
- Smooth Customs Clearance: With a valid import license on record, customs clearance at Indian ports becomes significantly faster and less likely to face holds.
Eligibility Criteria for Form MD-14
To apply for an import license via Form MD-14, the applicant must meet certain conditions set by CDSCO. These are non-negotiable requirements:
For the Foreign Manufacturer
- Must be a legally registered company in the country of origin.
- Must have necessary approvals or market authorization in their home country (in most cases).
- Must designate an authorized Indian agent who will act as the local point of contact for CDSCO.
- Manufacturing facility must comply with applicable quality standards (ISO 13485 or equivalent Good Manufacturing Practices).
For the Authorized Indian Agent / Importer
- Must be a resident Indian individual or a company registered in India.
- Must have a valid written agreement or authorization letter from the foreign manufacturer.
- Must possess or apply for a Drug License / Medical Device License from the State Licensing Authority (where applicable).
- Must have adequate premises for storage of medical devices as per prescribed standards.
- Must employ a technically qualified person (graduate in pharmacy, medicine, or relevant science) as required under MDR 2017.
Documents Required for Form MD-14 Submission
This is probably the section most importers spend the most time on — and for good reason. Document preparation is the most time-intensive part of the MD-14 process. Here is a detailed breakdown of what you will typically need:
Application-Level Documents
| Document | Details / Notes |
| Duly filled Form MD-14 | Signed by the authorized signatory of the applicant entity |
| Covering letter on company letterhead | Addressed to the Central Licensing Authority, CDSCO |
| Proof of entity registration in India | Certificate of Incorporation / Partnership Deed / Proprietorship proof |
| Drug License / Medical Device License (State) | Issued by State Licensing Authority — required for warehouse/import premises |
| Site Master File (SMF) or Undertaking for premises | Details of import storage premises |
| Technically Qualified Person (TQP) details | Educational qualification certificates and appointment letter |
Foreign Manufacturer-Related Documents
| Document | Details / Notes |
| Authorization letter from foreign manufacturer | Authorizing the Indian agent to apply on their behalf — notarized and apostilled |
| Certificate of Incorporation of foreign manufacturer | Registered in the country of origin |
| Free Sale Certificate (FSC) | Issued by competent authority in the country of origin or manufacturing country |
| ISO 13485 or GMP Certificate | Valid manufacturing quality certificate |
| Manufacturing site audit report (if applicable) | For Class C and D devices — audit by CDSCO or accredited body |
| Declaration of Conformity | Manufacturer's declaration of compliance with applicable standards |
Product-Specific Documents
| Document | Details / Notes |
| 510(k) / CE Certificate / Equivalent approval | Market approval in US, EU, or other reference countries |
| Intended Use and Indications | Clear product description with intended use statement |
| Technical File / Design Dossier summary | Technical specifications of the device |
| Labeling and packaging details | Proposed Indian labeling complying with MDR 2017 Schedule IV requirements |
| Performance testing data | Analytical / performance / safety test reports |
| Clinical evaluation report (for Class C and D) | Clinical data supporting safety and performance |
| Shelf life / stability data | Required for devices with defined shelf life |
| Biocompatibility data (for implants or body-contact devices) | As per ISO 10993 series |


| Pro Tip for Importers |
| All foreign documents need to be apostilled or legalized as per the country of origin. CDSCO has become stricter about this in recent years. Documents submitted in a language other than English must be accompanied by a certified English translation. Keep all documents ready in both physical and digital format — the SUGAM portal (CDSCO's online portal) requires digital uploads. |
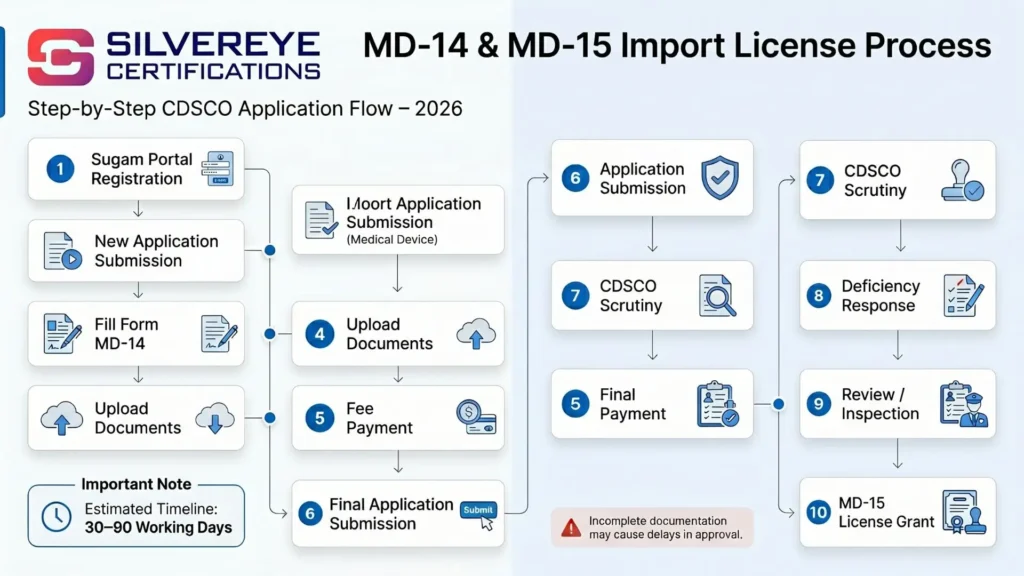
Step-by-Step Process for Form MD-14 Application
Here is how the actual Form MD-14 import license process works from start to finish in 2026:
| Step | Action | Who Does It | Notes |
| 1 | Classify the Medical Device | Importer / Regulatory Consultant | Refer to MDR 2017 Schedule I and CDSCO device classification list |
| 2 | Check if Device is Notified / Already Approved | Importer | Use CDSCO website — some devices may already be registered under the manufacturer's name |
| 3 | Gather All Required Documents | Importer + Foreign Manufacturer | Allow 4–8 weeks for foreign documents with apostille |
| 4 | Register on SUGAM Portal | Importer | sugam.mohfw.gov.in — create applicant account |
| 5 | Fill and Submit Form MD-14 Online | Importer | Fill all fields carefully; upload all supporting documents |
| 6 | Pay the Prescribed Government Fee | Importer | Online payment via SUGAM portal |
| 7 | CDSCO Technical Scrutiny | CDSCO | Documents verified by Central Licensing Authority team |
| 8 | Deficiency Letter (if any) | CDSCO | Reply within the specified timeline — usually 30 days |
| 9 | Manufacturing Site Inspection (if required) | CDSCO | Mandatory for Class C and D; may be waived for some Class B |
| 10 | Grant of Import License in Form MD-15 | CDSCO | Typically 3–9 months depending on device class |

Form MD-14 Government Fees
The fee structure for import license applications under Form MD-14 is set by the central government. Here is a general overview based on the Schedule VII of MDR 2017 and subsequent amendments:
| Fee Category | Amount (INR) | Notes |
| New Import License Application — Class A | Rs. 5,000 per device | Subject to revision by government notification |
| New Import License Application — Class B | Rs. 10,000 per device | Subject to revision by government notification |
| New Import License Application — Class C | Rs. 25,000 per device | Subject to revision by government notification |
| New Import License Application — Class D | Rs. 50,000 per device | Subject to revision by government notification |
| License Renewal Fee | Typically 50% of original fee | Check latest government notification before applying |
| Amendment to Existing License | Varies by amendment type | Consult CDSCO latest circular |
| Fee Notice |
| Fees are paid online through the SUGAM portal at the time of application submission. Fees are non-refundable even if the application is rejected or withdrawn. Always verify the current fee schedule on the CDSCO official website (cdsco.gov.in) before submitting, as the government periodically revises the fee structure through official Gazette notifications. |
Validity of the Import License (Form MD-15)
The import license granted on the basis of Form MD-14 (issued in Form MD-15) is generally valid for 5 years from the date of grant.
| Parameter | Details |
| License Validity | 5 years from date of issue |
| Renewal Window | Application must be submitted before expiry (ideally 3-6 months in advance) |
| Grace Period | Limited or none — expired licenses cannot be used for import |
| Post-Expiry Import | Not permitted — customs may reject consignments against expired license |
| License Transferability | Not transferable — linked to specific importer and foreign manufacturer |
| Changes to Device / Manufacturer | Require amendment to existing license or fresh application |
Renewal Process and Timeline for Form MD-14 License
Renewing your import license is just as important as getting it the first time. Many importers delay renewal and end up creating compliance gaps that can be costly. Here is what you need to know:
When to Apply for Renewal
- Start the renewal process at least 3 to 6 months before the license expiry date.
- CDSCO recommends submitting the renewal application before expiry so that imports can continue without interruption under the previously held license while the renewal is being processed.
- If the license has already expired, you may need to reapply as a fresh application — which takes significantly longer.
Renewal Application Steps
- Log in to the SUGAM portal using your existing credentials.
- Navigate to the renewal section and select your existing license.
- Upload updated documents — especially renewed ISO 13485/GMP certificates, updated Free Sale Certificate, and renewed authorization letter from foreign manufacturer.
- Pay the applicable renewal fee online.
- Submit and track the application on SUGAM. Respond promptly to any deficiency raised by CDSCO.
| Renewal Tip |
| Even if your device specifications have not changed, some supporting documents from the foreign manufacturer (like GMP certificate, Free Sale Certificate) may have their own expiry dates. Make sure all such documents are current at the time of renewal submission — expired supporting documents are the most common reason for renewal delays. |
Common Reasons for Form MD-14 Application Rejection or Delay
Knowing what goes wrong for other applicants can save you significant time and money. Here are the most frequent issues CDSCO raises:
- Incomplete Documentation: Missing documents or documents without proper apostille or notarization.
- Expired Supporting Documents: ISO certificates, Free Sale Certificates, or GMP certificates that are past their validity date at the time of submission.
- Mismatch in Product Details: Product description, intended use, or device name in Form MD-14 not matching the technical documents.
- Authorization Letter Issues: Authorization from foreign manufacturer not in the prescribed format, missing specific authorization scope, or not apostilled.
- Incorrect Device Classification: Applying under the wrong risk class — this can trigger re-evaluation or outright rejection.
- Labeling Non-Compliance: Proposed Indian labeling not meeting MDR 2017 Schedule IV requirements (language, mandatory information, etc.).
- Storage Premises Non-Compliance: State licensing authority requirements for storage conditions not met — especially for temperature-sensitive devices.
- Incomplete SUGAM Upload: Technical issues or incomplete document uploads on the portal that go unnoticed before submission.
How to Apply on the SUGAM Portal
CDSCO conducts the entire Form MD-14 import license application process through its online portal called SUGAM. Here is a quick orientation for first-time applicants:
| Portal Feature | Details |
| Portal Name | SUGAM (System for Unified Grant and Management) |
| Website | sugam.mohfw.gov.in |
| Managed By | Ministry of Health and Family Welfare, Government of India |
| Application Submission | Fully online — no physical submission required for most applications |
| Document Upload | PDF format, usually individual file size limits apply |
| Payment | Online payment gateway integrated into the portal |
| Status Tracking | Real-time tracking of application status after submission |
| Communication | CDSCO raises queries and sends deficiency notices through the portal |
Form MD-14 vs Other CDSCO Forms
Importers often get confused between different CDSCO forms. Here is a quick comparison to set the context straight:
| Form | Purpose | Who Uses It |
| Form MD-14 | Application for Import License for Medical Devices | Importers / Authorized Indian Agents |
| Form MD-15 | Import License Certificate (granted after MD-14 approval) | Issued by CDSCO to the applicant |
| Form MD-41 | Application for Registration of Medical Devices (for foreign manufacturers) | Foreign manufacturers registering directly |
| Form MD-42 | Registration Certificate for Medical Devices | Issued by CDSCO post MD-41 review |
| Form MD-3 | Application for Manufacture License (domestic) | Indian manufacturers |
| Form MD-7 | Application for Loan License (domestic manufacture) | Loan license applicants |
Recent Regulatory Updates Affecting Form MD-14 Process (2025–2026)
Staying current with regulatory changes is part of being a responsible importer. Here are some key developments that have shaped the MD-14 landscape in recent times:
- Phased Implementation Completion: As of 2026, Class A and Class B devices are now fully under mandatory licensing, closing the earlier gap where only notified devices required licensing.
- Enhanced Digital Processing on SUGAM: CDSCO has improved SUGAM's backend to enable faster deficiency resolution and better applicant-CDSCO communication.
- Risk-Based Audit for Class B: A new risk-based audit protocol was introduced for certain Class B devices, which may require an on-site inspection of the foreign manufacturing facility.
- Tightened Labeling Requirements: CDSCO issued revised guidelines on labeling, making it mandatory to include QR codes linking to product information for certain device categories.
- Import of Investigational Devices: Revised provisions under MDR for importing medical devices for clinical investigation now have a separate, faster track distinct from the Form MD-14 commercial import process.
| Stay Updated |
| Regulations evolve. Always check the CDSCO official website (cdsco.gov.in) and SUGAM portal for the latest circulars, guidance documents, and amendments before starting or renewing your Form MD-14 application in 2026. |
Conclusion: Getting Form MD-14 Right the First Time
Form MD-14 is not just a bureaucratic formality — it is the regulatory foundation on which your entire medical device import business in India rests. Getting it right matters, not just for initial market entry, but for long-term compliance, customer trust, and growth in one of the world's largest medical device markets.
The process can be complex, especially if you are dealing with Class C or D devices or if your foreign manufacturer is new to the Indian regulatory framework. But with the right preparation — understanding the documentation requirements, meeting eligibility criteria, using the SUGAM portal correctly, and staying on top of renewal timelines — it is absolutely manageable.
If you are serious about importing medical devices into India in 2026 and beyond, treat the Form MD-14 for medical device import process as a strategic priority, not a box-ticking exercise. A well-managed import license keeps your supply chain smooth, your customers confident, and your business on the right side of one of the fastest-evolving regulatory environments in Asia.
Frequently Asked Questions
What is the difference between Form MD-14 and Form MD-41?
Form MD-14 is the application to import medical devices into India, while Form MD-41 is the application to sell, stock, or distribute (wholesale) medical devices within India
How long does it take to get approval on Form MD-14?
The timeline varies by device class. Class A and B devices may get approval in 3 to 6 months under normal circumstances. Class C and D devices typically take 6 to 12 months, and can extend further if a manufacturing site inspection is required or if deficiencies arise.
Can I import a medical device before my Form MD-14 is approved?
Technically, you cannot import devices after the license expires. However, if you have applied for renewal before the expiry date, CDSCO may allow continued imports while the renewal is pending — but this is not guaranteed. It is safest to apply for renewal at least 3 to 6 months before expiry.
Is a separate Form MD-14 needed for each product or each device model?
Generally, each distinct medical device or product variant that has a separate intended use or differs materially in composition or design requires a separate application
Does the foreign manufacturer need to be physically present in India for the process?
No. The foreign manufacturer does not need to be present in India. The authorized Indian agent manages the entire process on their behalf.
Can a single Indian importer hold licenses for devices from multiple foreign manufacturers?
Yes. An Indian importer can hold multiple Form MD-14 import licenses — one for each device or manufacturer relationship.
What is the Free Sale Certificate, and why is it required for Form MD-14?
A Free Sale Certificate (FSC) is an official document issued by the regulatory or governmental authority in the country of origin, confirming that the medical device is legally marketed and sold freely in that country.
Can Form MD-14 be submitted in physical format, or is it only online
As of 2024 onwards, CDSCO primarily processes Form MD-14 applications through the SUGAM online portal. Physical submissions are no longer the standard procedure






































