- MD-14 is the application form and MD-15 is the import license granted by CDSCO for importing medical devices into India.
- Every importer of medical devices must obtain a valid MD-15 license before shipping any regulated device into India.
- The process involves online application through the Sugam portal, document submission, and CDSCO review — typically completed within 30 to 90 working days.
- Non-compliance with MD-14 and MD-15 requirements can lead to seizure of goods, penalties, or cancellation of import rights.
Introduction
If you are planning to import medical devices into India, there is one regulatory step you simply cannot skip — obtaining your MD-14 and MD-15 import license from CDSCO.
It does not matter whether you are a multinational medical device company entering the Indian market for the first time or an established Indian distributor adding a new product line. Without a valid MD-15 import license, your shipment will not clear Indian customs. Full stop.
India regulates the import of medical devices under the Medical Devices Rules, 2017 (MDR 2017), framed under the Drugs and Cosmetics Act, 1940. The Central Drugs Standard Control Organization — commonly known as CDSCO — is the authority responsible for reviewing applications and granting these licenses. The process starts with Form MD-14, which is the application you submit, and ends with Form MD-15, which is the actual import license CDSCO issues to you upon approval.


What you will find here is a complete, step-by-step resource covering every aspect of the MD-14 and MD-15 import license — who needs it, what documents are required, how to apply through the Sugam portal, how much it costs, how long the license remains valid, and how to renew it before it expires.
Everything in this guide reflects the current regulatory requirements as of 2026. Medical device regulation in India has evolved significantly over the last few years, and if you are working with information that is even a year or two old, there is a good chance some of it is already outdated.
What Are MD-14 and MD-15 Import License?
India regulates the import medical devices under the Medical Devices Rules, 2017 (MDR 2017), which falls under the Drugs and Cosmetics Act, 1940. The Central Drugs Standard Control Organization (CDSCO) is the nodal authority responsible for approving and licensing all medical device imports.
Here is how MD-14 and MD-15 are defined in simple terms:
| Form | What It Is | Who Uses It |
| Form MD-14 | Application form submitted by the importer to CDSCO to request an import license for a medical device | Manufacturer's authorized agent or the importer in India |
| Form MD-15 | The actual import license issued by CDSCO upon successful review and approval of MD-14 application | Granted to the importer/authorized agent after approval |


In practice, you fill out MD-14 to apply, and if everything checks out, CDSCO issues you the MD-15 license. Without a valid MD-15, customs authorities in India will not allow the medical devices to clear the port.
Who Needs an MD-14 and MD-15 Import License?
Not everyone importing goods into India needs an MD-15 license — but if your product falls under the category of a regulated medical device, this license is non-negotiable. Here is who needs it:
• Indian companies acting as authorized agents of a foreign manufacturer
• Importers bringing in in-vitro diagnostics (IVDs) regulated under Schedule H of MDR 2017
• Distributors importing Class A, B, C, or D medical devices
• Entities importing combination products where the device component is the primary mode of action
• Companies importing re-usable or single-use surgical instruments classified as medical devices
The following are currently out of scope for MD-14/MD-15 licensing:
• Devices used purely for veterinary purposes (regulated separately)
• Devices imported exclusively for clinical investigation purposes (covered under separate provisions)
• Personal-use imports below a threshold quantity (subject to customs rules)
Pro tip: If you are unsure whether your product qualifies as a medical device under MDR 2017, check the CDSCO device classification database or consult a regulatory affairs expert before initiating the application. Misclassification is one of the most common and costly mistakes importers make.
Eligibility Criteria for MD-14 and MD-15 Import License
Before you start preparing your application, make sure your organization meets these eligibility conditions set by CDSCO:
- The applicant must be a legally registered entity in India — a sole proprietorship, partnership, LLP, or private/public limited company.
- The importer must have a valid Import Export Code (IEC) issued by the Directorate General of Foreign Trade (DGFT).
- For Class B, C, and D devices, the foreign manufacturer must already hold a valid manufacturing license or CE/ISO 13485 certification recognized by CDSCO.
- The applicant must be officially designated as the Authorized Indian Representative (AIR) of the foreign manufacturer through a formal Letter of Authorization (LOA).
- The applicant must have adequate facilities or arrangements to handle, store, and maintain records for the imported devices.
- Previous import licenses should not have been suspended or cancelled for non-compliance.
Documents Required for MD-14 and MD-14 Import License Application
Document preparation is the most time-consuming part of the MD-14 application. Below is a comprehensive checklist so you do not miss anything:
Company and Authorization Documents
• Certificate of Incorporation or Business Registration Certificate
• Import Export Code (IEC) certificate
• Letter of Authorization (LOA) from the foreign manufacturer — must be notarized and apostilled
• Power of Attorney (if applicable)
• PAN card of the company or proprietor
• GST Registration Certificate
Manufacturer Documents
• Manufacturing license or equivalent certificate issued by the competent authority in the country of origin
• ISO 13485 certificate or CE marking certificate (for Class B, C, D devices)
• Free Sale Certificate (FSC) or Certificate of Exportability from the competent authority of the manufacturing country
• WHO-GMP certificate, if applicable
Device-Specific Documents
• Device description, intended purpose, and principle of operation
• Technical documentation and product dossier (as required by device class)
• Labelling and Instructions for Use (IFU) in English
• Risk management summary (as per ISO 14971)
• Performance evaluation or clinical data (for Class C and D devices)
• Shelf life data and storage conditions
Application-Specific Documents
• Duly filled Form MD-14 (online submission via Sugam portal)
• Undertaking for compliance with MDR 2017
• Site Master File (SMF) or establishment details of the importer
• Proof of payment of application fee
Keep all documents organized in both soft copy (PDF format) and hard copy. CDSCO may request originals or certified copies during review.


MD-14 and MD-15 Import License Application Process: Step-by-Step for 2026
The process for applying for an MD-14 import license in India is entirely online through the Sugam portal (sugamapp.nic.in). Here is a clear, step-by-step breakdown:
| Step | Action | Timeline |
| Step 1 | Register on the Sugam portal and create a user account for your company | 1–2 days |
| Step 2 | Log in and select 'New Application' under the Import License module | Same day |
| Step 3 | Fill in Form MD-14 with all device and company details accurately | 2–5 days |
| Step 4 | Upload all supporting documents in the prescribed format | 1–3 days |
| Step 5 | Pay the prescribed application fee online through the portal | Same day |
| Step 6 | Submit the application and note your application reference number | Same day |
| Step 7 | CDSCO scrutiny of application — deficiency notice may be raised if documents are incomplete | 15–30 working days |
| Step 8 | Respond to any deficiency notice within the stipulated time (usually 30 days) | As needed |
| Step 9 | CDSCO review and site inspection (if required for higher-class devices) | 30–60 working days |
| Step 10 | Grant of MD-15 import license — license issued digitally on Sugam portal | Final stage |
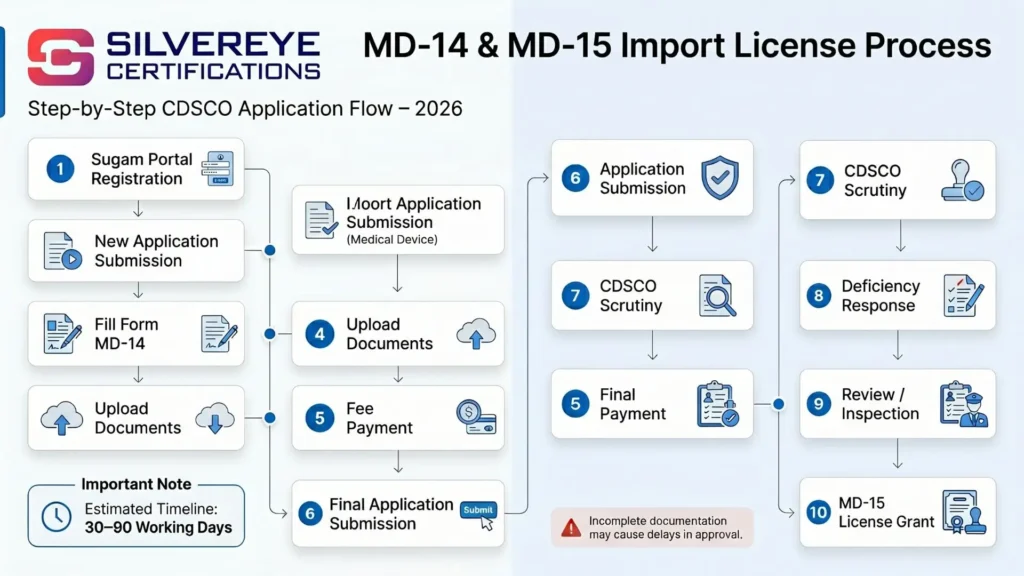

Total expected timeline: 30 to 90 working days for a complete application. If there are deficiencies or additional queries from CDSCO, this timeline may extend. Incomplete applications are the single biggest reason for delays, so invest time upfront in getting the documents right.
MD-14 and MD-15 Import License Fees in India
The fee structure for MD-14 import license applications is determined by the device class. Here is the current fee table based on the Medical Devices Rules, 2017:
| Device Class | Risk Category | Application Fee (INR) | License Fee (INR) |
| Class A | Low Risk | Rs. 3,000 | Rs. 1,000 |
| Class B | Low-Moderate Risk | Rs. 5,000 | Rs. 2,500 |
| Class C | Moderate-High Risk | Rs. 20,000 | Rs. 5,000 |
| Class D | High Risk | Rs. 50,000 | Rs. 10,000 |
| IVDs – Class A | Low Risk | Rs. 3,000 | Rs. 1,000 |
| IVDs – Class D | High Risk | Rs. 50,000 | Rs. 10,000 |
Note: Fees may be revised by the government. Always verify the current fee schedule on the official Sugam portal or CDSCO website before submitting your payment. Fees paid are non-refundable even if the application is rejected.
Validity of MD-14 and MD-15 Import License — How Long Does It Last?
One of the most frequently asked questions about the MD-15 import license is about its validity. Here is what you need to know:
| License Type | Validity Period | Notes |
| Initial MD-15 License | 5 years from date of grant | Applicable to all device classes |
| Renewed MD-15 License | 5 years from date of renewal | Must apply 3 months before expiry |
| License with conditions | As specified in the license | Subject to compliance conditions |
The MD-15 import license is valid for 5 years from the date of issue. After expiry, importers cannot legally import the specified device until a fresh license or renewal is granted. Operating with an expired license is a violation under the Drugs and Cosmetics Act and can invite penal action.
How to Renew MD-14 And MD-15 Import License — Renewal Process
License renewal is not automatic. You have to take proactive steps to avoid a break in your import operations. Here is the renewal timeline and process:
Renewal Timeline
• Start the renewal process at least 3 months before the license expiry date
• Applications submitted before expiry allow you to continue importing under the existing license until a decision is made
• If you miss the deadline and the license expires, you must apply for a fresh license — not a renewal
Renewal Documents Required
• Renewal application in Form MD-14 (same form used for new applications)
• Current MD-15 license copy
• Updated Letter of Authorization from the manufacturer (if the original LOA has expired)
• Updated Free Sale Certificate and manufacturing license
• Declaration of no change in device specifications (or updated dossier if there are changes)
• Proof of renewal fee payment
Renewal Process
- Log in to the Sugam portal
- Select 'Renewal' under the Import License module
- Verify pre-filled details and update any changed information
- Upload updated documents
- Pay the renewal fee
- Submit and track application status
Renewal fees are generally the same as the initial application fees for the respective device class. CDSCO may conduct a review or ask for additional information during the renewal process, especially if there have been any regulatory changes affecting the device category.
Key Benefits of Obtaining MD-14 and MD-15 Import License
Securing your MD-15 import license is not just a legal formality — it opens real business advantages:
| Benefit | Details |
| Legal Compliance | Ensures full compliance with MDR 2017 and Drugs and Cosmetics Act — protects your business from legal action |
| Market Access | Provides authorized access to one of the world's largest and fastest-growing medical device markets |
| Customs Clearance | Enables smooth customs clearance at all Indian ports and airports without delays or seizure risk |
| Business Credibility | Adds regulatory credibility when engaging with hospitals, distributors, and government tenders |
| Brand Protection | Prevents counterfeit or unauthorized imports of the same device by others in the market |
| Tender Eligibility | A valid MD-15 license is often a prerequisite for participating in government procurement tenders |
| Expanded Distribution | Allows nationwide distribution through licensed wholesale dealers and retailers |
Common Reasons for MD-14 Application Rejection or Delay
Understanding why applications get rejected helps you avoid the same mistakes. Here are the most common issues:
• Incomplete or incorrect Form MD-14 — missing mandatory fields or incorrect device classification
• Letter of Authorization not properly notarized and apostilled
• Free Sale Certificate or manufacturing license already expired at the time of submission
• Technical dossier does not meet the requirements for the device class
• Discrepancy between device name/description in LOA and in the product dossier
• Missing Performance Evaluation data for IVDs or clinical evidence for Class C/D devices
• Failure to respond to deficiency notices within the stipulated time
• Fees paid do not match the applicable fee for the declared device class
Always have a qualified regulatory affairs consultant review your application before submission. The cost of a consultant is negligible compared to the cost of delays, rejections, and re-applications.
MD-14 and MD-15 Compliance Requirements After License Grant
Getting the license is not the end of your compliance obligations. Once you hold an MD-15 import license, you must:
• Maintain import records and stock registers for all imported devices
• Ensure all imported devices carry proper labelling as per Schedule I of MDR 2017
• Report any adverse events or device failures to CDSCO within the stipulated timelines
• Allow CDSCO officers to inspect premises, records, and stock upon request
• Inform CDSCO of any changes to device specifications, manufacturer details, or LOA
• Not import or sell devices after the MD-15 license expires
• Maintain a proper cold chain for devices requiring temperature-controlled storage
MD-14 vs MD-15 Import License: Quick Comparison
| Parameter | MD-14 | MD-15 |
| Nature | Application Form | Import License (approval document) |
| Submitted by | Importer / Authorized Agent | Not submitted — issued by CDSCO |
| Purpose | Request for import authorization | Proof of authorization to import |
| Regulatory basis | Medical Devices Rules, 2017 | Medical Devices Rules, 2017 |
| Validity | Not applicable (it's a form) | 5 years from date of issue |
| Portal | Sugam (sugamapp.nic.in) | Issued digitally on Sugam portal |
| Fees payable | Yes — with MD-14 submission | License fee payable after approval |
CDSCO's Role in MD-14 and MD-15 Import License
The Central Drugs Standard Control Organization (CDSCO) is the apex regulatory body for medical devices in India, operating under the Ministry of Health and Family Welfare. CDSCO's Medical Device Division is specifically responsible for:
• Reviewing and processing MD-14 applications
• Issuing MD-15 import licenses
• Conducting inspections of importer premises when necessary
• Maintaining the national database of licensed medical device importers
• Issuing show cause notices and cancelling licenses for non-compliance
• Updating guidelines and policies for medical device regulation
CDSCO headquarters is located in New Delhi, and regional offices in Mumbai, Chennai, Kolkata, Hyderabad, Ahmedabad, and Guwahati assist in processing applications and inspections.
Conclusion: Is Getting MD-14 and MD-15 License Worth the Effort?
Absolutely, yes. India's medical device market is growing at a CAGR of over 15% and is projected to reach USD 50 billion by 2030. Getting your MD-15 import license is the gateway to participating in this growth legally, sustainably, and profitably.
The process can feel complex, especially for first-time importers navigating CDSCO regulations. But with the right documents, a clear understanding of the requirements, and ideally the support of an experienced regulatory consultant, the MD-14 application process is very much achievable.
The key is to start early, prepare thoroughly, and respond promptly to any queries from CDSCO. Your MD-15 import license is not just a regulatory checkbox — it is your license to operate legitimately in one of the most promising medical device markets in the world.
Frequently Asked Questions
What is the difference between MD-14 and MD-15?
MD-14 is the application form you submit to CDSCO to request permission to import medical devices. MD-15 is the actual import license that CDSCO issues to you once your application is approved. Think of MD-14 as the request and MD-15 as the approval.
How long does it take to get an MD-15 import license?
A complete and accurate application typically takes 30 to 90 working days. If CDSCO raises a deficiency notice or requests additional documents, the timeline can extend further. Higher-class devices (Class C and D) may take longer due to the additional technical review involved.
Can a foreign company directly apply for MD-15 in India?
No. A foreign manufacturer cannot directly apply for an MD-15 import license. They must appoint an Authorized Indian Representative (AIR) or a local Indian company through a formal Letter of Authorization (LOA). The AIR applies on behalf of the foreign manufacturer.
Is MD-15 license required for Class A medical devices?
Yes, all regulated medical devices including Class A (low risk) devices require an MD-15 import license if they fall under the notified categories under MDR 2017. However, the documentation and fee requirements are lighter for Class A compared to higher-risk classes.
What happens if I import medical devices without an MD-15 license?
Importing medical devices without a valid MD-15 license is a violation of the Drugs and Cosmetics Act. Consequences include seizure and confiscation of the imported goods, monetary penalties, legal prosecution, and permanent blacklisting from obtaining future licenses.
Can I import multiple devices under a single MD-15 license?
No. Each device or device variant typically requires a separate MD-15 license. If you are importing multiple devices, you need to file separate MD-14 applications for each. Some closely related device variants from the same may be covered under one application at CDSCO's discretion.
What is the validity period of MD-15 import license?
An MD-15 import license is valid for 5 years from the date of issue. You must apply for renewal at least 3 months before the expiry date to avoid a break in your import authorization.
Where do I submit the MD-14 application?
MD-14 applications are submitted online through the Sugam portal at sugamapp.nic.in. Physical submissions are not accepted. You need to create an account on the portal, complete the form, upload documents, and pay the fee — all online.
What is the fee for MD-14 import license application?
The fee depends on the class of the medical device. It ranges from Rs. 3,000 for Class A devices to Rs. 50,000 for Class D devices. Fees must be paid online through the Sugam portal and are non-refundable regardless of the outcome of the application.
Can the MD-15 license be transferred to another company?
No. An MD-15 import license is non-transferable. It is issued specifically to the licensed entity. If the company undergoes a change in ownership, merger, or restructuring, a fresh application may be required, and CDSCO must be informed of any such changes.
Do I need a separate license for each Indian port of entry?
No. An MD-15 license is valid across all ports and customs points in India. You do not need separate licenses for different ports.
What should I do if CDSCO raises a deficiency notice?
If CDSCO raises a deficiency notice, you typically have 30 days to respond and submit the additional or corrected information. Failure to respond within the stipulated time may result in your application being treated as abandoned. Always address deficiency notices promptly and completely.






































