- Every stethoscope sold or imported in India must be registered under CDSCO — no exceptions as of 2026.
- Stethoscopes fall under Class A (low risk) medical devices — registration is mandatory but the process is relatively straightforward.
- An LMPC (Legal Metrology Packaged Commodities) certificate is additionally required for stethoscopes sold as packaged consumer goods.
- New 2026 CDSCO notifications have tightened post-market surveillance and labelling norms — this guide covers everything you need to stay compliant.
Introduction
If you are a manufacturer, importer, or distributor planning to sell stethoscopes in India, you already know that regulatory compliance is not optional. The Central Drugs Standard Control Organization (CDSCO) brought Medical Devices under a structured licensing framework through the Medical Devices Rules, 2017, and stethoscopes — classified as Class A low-risk devices — became fully notified under Schedule I. What does this mean for your business? It means you cannot legally manufacture, import, sell, stock, or distribute a stethoscope in India without a valid registration certificate.
This pillar guide walks you through every single aspect of the process — from understanding what type of certification applies to your product, to eligibility criteria, documents, step-by-step process, realistic cost estimates, timelines, validity, and renewal. We have also incorporated the latest 2026 regulatory updates so the information you are reading right now is current and accurate.
Why Is Medical Device Registration for Stethoscopes Mandatory in India?
India's medical device regulatory landscape changed fundamentally with the Medical Devices Rules, 2017 (MDR 2017), which came fully into force under the Drugs and Cosmetics Act, 1940. The rules created a risk-based classification system for medical devices, and stethoscopes — being diagnostic instruments — were placed under Schedule I as notified medical devices.


Before this, stethoscopes sat in a grey zone. Many manufacturers and importers operated without specific device registration, relying only on import licenses or general trade compliance. That window closed. By 2022, CDSCO made it mandatory for all Class A and Class B devices to obtain either a manufacturing license (Form MD-5) or an import license (Form MD-14), depending on whether the product is made in India or brought in from abroad.
Selling an unregistered stethoscope in India today can result in:
- Seizure and recall of the product
- Cancellation of import or distribution licenses
- Penalties under the Drugs and Cosmetics Act
- Criminal liability for responsible persons in the company
Classification of Stethoscope as a Medical Device in India
Understanding the risk classification of a stethoscope is the first step before you do anything else. The Medical Devices Rules, 2017 follow a four-tier risk classification:
| Class | Risk Level | Examples | Regulatory Pathway |
| Class A | Low Risk | Stethoscopes, bandages, tongue depressors | Self-certification + CDSCO Registration |
| Class B | Low-Moderate Risk | Hypodermic needles, syringes | CDSCO Registration |
| Class C | Moderate-High Risk | Ventilators, bone fixation plates | CDSCO Registration + Audit |
| Class D | High Risk | Pacemakers, HIV test kits | CDSCO Registration + Stringent Review |
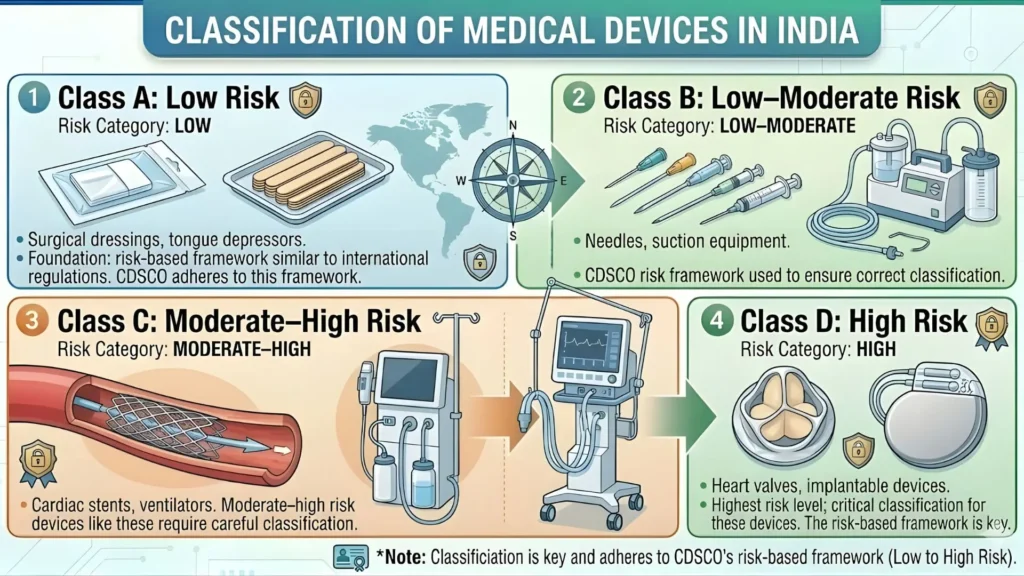

A standard acoustic stethoscope (used for listening to heart, lung, and bowel sounds) is classified as a Class A medical device under Schedule I of the MDR 2017, with the product code typically falling under 'diagnostic equipment — non-electric.' Electronic stethoscopes with amplification circuits may be classified as Class B in some cases — always confirm your specific product code with a regulatory consultant before filing.
Types of Certificates / Licenses Required for Stethoscopes
This is where many people get confused — there is not one single certificate for stethoscopes. Depending on your business activity (manufacturing, importing, or distributing) and how the product is sold (B2B or retail packaged), you may need one or more of the following:
CDSCO Manufacturing License (Form MD-5) — For Indian Manufacturers
If you manufacture stethoscopes in India, you need a manufacturing license issued in Form MD-5 by the State Licensing Authority (SLA) under CDSCO guidance. This authorizes you to make and sell the device within India and, with the right endorsements, export it as well.
CDSCO Import License (Form MD-14) — For Importers
If you import stethoscopes from outside India — whether from China, Germany, the USA, or anywhere else — you need an import registration certificate issued in Form MD-14 by the Central Licensing Authority (CLA), which is the CDSCO. This is the most common pathway for businesses sourcing stethoscopes internationally.
LMPC Certificate (Legal Metrology Packaged Commodities)
If the stethoscope is sold as a packaged commodity to end consumers (hospitals, clinics, retail pharmacies, or online platforms), the importer or manufacturer also needs an LMPC Certificate under the Legal Metrology (Packaged Commodities) Rules, 2011. This certificate ensures that the product packaging carries all mandatory declarations such as:
- Name and address of the importer / manufacturer
- Country of origin
- Net quantity / weight
- Maximum retail price (MRP) inclusive of all taxes
- Month and year of manufacture / import
- Customer care contact number
The LMPC certificate is issued by the Controller of Legal Metrology in the respective state. For importers, it is a mandatory pre-import requirement because customs will check for it during clearance.
BIS Certification (If Applicable)
The Bureau of Indian Standards (BIS) currently does not mandate compulsory BIS certification for stethoscopes under any Quality Control Order as of 2026. However, voluntary BIS marking (IS standard) can strengthen your market position, especially for government tenders.
ISO 13485 — Quality Management System
While not a government-issued license, ISO 13485 certification (Quality Management System for Medical Devices) is increasingly required by large hospital procurement departments and is often asked for during CDSCO audits as evidence of good manufacturing practice. If you are exporting as well, it is practically mandatory.
Eligibility Criteria for Stethoscope Registration
Who can apply for a stethoscope registration or license in India? The eligibility requirements differ slightly based on the license type:
Eligibility for Import License (Form MD-14)
| Criterion | Requirement |
| Applicant Type | Company or firm registered in India (Pvt Ltd, LLP, Partnership, Proprietorship) |
| Authorised Indian Representative (AIR) | Mandatory if the foreign manufacturer has no Indian entity |
| Foreign Manufacturer Audit (FSC) | Free Sale Certificate from country of origin is typically required |
| Technical Responsible Person | Person with relevant qualification overseeing regulatory affairs |
| Principal Manufacturer Agreement | Notarised agreement between the Indian importer and foreign manufacturer |
Eligibility for Manufacturing License (Form MD-5)
| Criterion | Requirement |
| Applicant Type | Indian entity (Pvt Ltd, LLP, etc.) |
| Premises | Manufacturing premises meeting MDR 2017 Schedule II requirements |
| Technical Staff | Qualified technical person on record (diploma/degree in relevant field) |
| Quality System | GMP-compliant quality management system in place |
| State Jurisdiction | Application filed with the SLA of the state where manufacturing happens |
Eligibility for LMPC Certificate
| Criterion | Requirement |
| Applicant Type | Importer, packer, or manufacturer selling packaged goods to end consumers |
| Business Registration | Valid GST registration and trade licence |
| Product Labels | Sample labels complying with Legal Metrology (Packaged Commodities) Rules, 2011 |
| State of Application | Applied in the state where your registered office is located |
Documents Required for Stethoscope Registration
Getting your documents right before you apply is the single most important thing you can do to avoid delays. CDSCO and state authorities are strict about incomplete submissions. Here is a comprehensive list:
Documents for CDSCO Import License (Form MD-14) — Stethoscope
- Duly filled application in Form MD-14 on the SUGAM portal
- Certificate of Incorporation / Business Registration of the Indian applicant
- Free Sale Certificate (FSC) or Certificate of Marketability from the country of origin — authenticated / apostilled
- ISO 13485 certificate of the foreign manufacturer (if available)
- Notarized appointment letter / agreement between Indian importer and foreign manufacturer
- Product literature, brochure, and technical specification sheet
- Labelling artwork and draft label compliant with MDR 2017 Schedule III
- Performance test reports (if required for the specific device class)
- Declaration of conformity by the manufacturer
- Audited financial statements of the Indian applicant company (last 2 years)
- Power of Attorney in favour of the authorized signatory
- Proof of premises (rent agreement / ownership document)
- GST registration certificate
- PAN card of the entity
- Cancelled cheque for payment reference
Documents for LMPC Certificate — Stethoscope
- Application form as prescribed by the state legal metrology department
- Certificate of Incorporation / firm registration
- GST registration certificate
- Import Export Code (IEC) — mandatory for importers
- Sample product label with all mandatory declarations printed
- Invoice copy of sample product (for importers)
- Proof of business address (utility bill / rent agreement)
- Authorization letter if applying through a consultant


Documents for Manufacturing License (Form MD-5)
- Application in Form MD-3 (application for new license) on SUGAM portal
- Site Master File (SMF) — details of manufacturing premises
- Layout plan of the manufacturing facility
- List of machinery and equipment with technical specifications
- Details of the technical responsible person with qualification certificates
- Standard Operating Procedures (SOPs) for key manufacturing and QC processes
- Product technical file including risk analysis (ISO 14971)
- Performance / functional test reports
- Stability data (where applicable)
- Declaration of conformity
- Business registration, GST, PAN
- State pollution control board consent (if applicable)
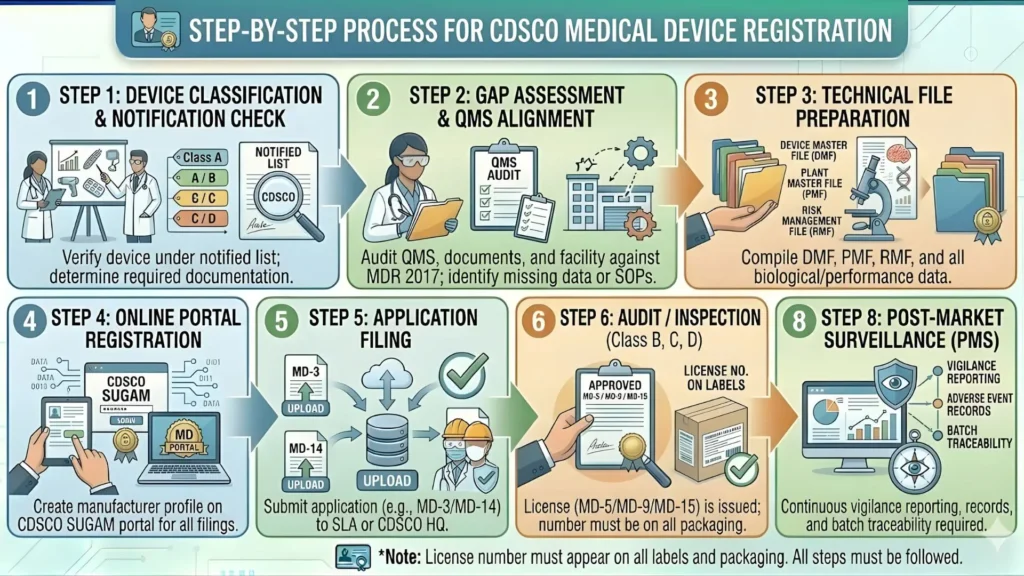
Step-by-Step Process for Medical Device Registration of Stethoscope
Let us walk through the complete process, step by step, so you know exactly what to expect at each stage.
Step 1: Determine Your Product Classification
Confirm whether your stethoscope is Class A (acoustic, standard) or Class B (electronic/amplified). Check the CDSCO notified list under Schedule I. If unsure, file a classification query through the CDSCO grievance portal before applying.
Step 2: Create Your SUGAM Account
SUGAM is CDSCO's online portal (sugam.cdscoonline.gov.in) where all medical device applications are submitted. Create an account for your organisation, complete the profile, and get your login credentials verified.
Step 3: Prepare Technical Documentation
Compile your technical file — product specifications, test reports, labelling artwork, manufacturer certificates, and all documents listed in Section 5 above. Keep a checklist so nothing is missed.
Step 4: Submit the Application on SUGAM Portal
Log in to SUGAM and navigate to the Medical Devices section. Select the appropriate form (MD-14 for import, MD-3 / MD-5 for manufacturing). Fill in all details carefully. Upload soft copies of all documents. Pay the prescribed government fee online.
Step 5: Scrutiny by CDSCO Officers
CDSCO officers conduct a preliminary scrutiny of the application. If documents are found incomplete or there are technical queries, you will receive a deficiency letter (DL) on SUGAM. You must respond within the specified time (usually 30 days) with the corrected information. Multiple rounds of deficiency are possible for complex applications.
Step 6: Technical Evaluation
For Class A devices like stethoscopes, the technical evaluation is relatively simple — there is no mandatory clinical trial data requirement. The officer verifies product safety, labelling compliance, and the authenticity of the Free Sale Certificate (for imports).
Step 7: Grant of License / Registration Certificate
Once satisfied, the licensing authority issues the Import Registration Certificate in Form MD-14 (for importers) or the Manufacturing Licence in Form MD-5 (for manufacturers). The certificate is made available for download on the SUGAM portal.
Step 8: Apply for LMPC Certificate (Parallel Process)
Simultaneously or immediately after CDSCO registration, apply to the State Legal Metrology Department for the LMPC certificate. This is a separate process run by a different government authority. Most states have online portals now. The process typically takes 15 to 30 working days.

| Stage | Authority | Typical Duration |
| SUGAM account creation and document prep | Applicant | 3–7 days |
| Application submission | CDSCO / SLA | 1 day |
| Preliminary scrutiny | CDSCO officer | 15–30 days |
| Deficiency response (if any) | Applicant | 15–30 days (per round) |
| Technical evaluation and approval | CDSCO CLA / SLA | 30–60 days |
| Licence issuance | CDSCO / SLA | 7–10 days after approval |
| LMPC Certificate (parallel) | State Legal Metrology | 15–30 working days |
Government Fee / Cost for Stethoscope Registration
One of the most searched questions is: how much does it cost to register a stethoscope in India? Let us break down the government fees as well as typical professional service charges.
| Certificate / Licence Type | Government Fee (Approx.) | Professional / Consultant Fee (Approx.) |
| CDSCO Import Licence — Class A (MD-14) | INR 5,000 per product | INR 20,000 – 50,000 |
| CDSCO Manufacturing Licence — Class A (MD-5) | INR 3,000 – 5,000 per product | INR 25,000 – 60,000 |
| LMPC Certificate (per state) | INR 300 – 1,000 per state | INR 3,000 – 8,000 |
| ISO 13485 Certification (if applicable) | INR 80,000 – 2,00,000 (full audit) | Included in certification body fee |
| FSC / Apostille Charges (for imports) | Varies by country of origin | INR 5,000 – 15,000 |
Note: Government fees mentioned above are indicative and subject to revision by CDSCO. Always verify the latest fee structure on the SUGAM portal before filing. Fees for multiple products or multiple states may vary.
Timeline for Stethoscope Registration in India
How long does medical device registration take for a stethoscope? The honest answer is: it depends on how well-prepared your documentation is. Here is a realistic estimate:
- Best-case scenario (complete documents, no deficiency): 60 to 90 days
- Typical scenario (one round of deficiency): 90 to 150 days
- Worst-case scenario (multiple rounds of deficiency or incomplete FSC): 6 to 12 months
The LMPC certificate process runs in parallel and typically takes 15 to 30 working days if all documents are in order, so it does not add to the critical path.
One practical tip: start your CDSCO registration process at least 3 to 4 months before your planned product launch date. Rushing regulatory processes rarely works in your favour.
Validity and Renewal of Stethoscope Registration
| Licence / Certificate Type | Validity Period | Renewal Requirement |
| CDSCO Import Licence (MD-14) — Class A | Perpetual (no fixed expiry) unless cancelled | Renewal not required unless there is a product change; surrender if stopping business |
| CDSCO Manufacturing Licence (MD-5) | Perpetual unless suspended / cancelled | Periodic renewal of premises licence as per state rules (typically 5 years) |
| LMPC Certificate | 1 year (some states issue multi-year) | Annual renewal before expiry |
| ISO 13485 Certification | 3 years (with annual surveillance audits) | Re-certification audit every 3 years |
A common misconception is that the CDSCO import license expires every year. It does not. The license is perpetual for Class A devices, but you must intimate CDSCO if:
- The product undergoes a significant design or specification change
- The foreign manufacturer changes
- The Indian importer entity undergoes a change in ownership or constitution
- You decide to stop importing the product
Benefits of Completing Stethoscope Registration
Regulatory compliance is not just about avoiding penalties. There are real, tangible business benefits to having your stethoscope properly registered:
- Legal market access: You can legally sell across all Indian states without risk of seizure
- Hospital and government tenders: Most government tenders require a valid CDSCO license — you simply cannot bid without it
- E-commerce listing: Platforms like Amazon India, Flipkart, and 1mg require proof of device registration for medical product listings
- Brand trust and credibility: Registered products build trust with doctors, hospitals, and distributors
- Export facilitation: A valid Indian manufacturing license helps you obtain a Free Sale Certificate from CDSCO if you want to export
LMPC Certificate for Stethoscope
Since a lot of our readers are specifically looking for the LMPC certificate for stethoscopes, let us go deeper on this particular certification.
What is LMPC Certification?
LMPC stands for Legal Metrology Packaged Commodities. Under the Legal Metrology Act, 2009 and the Legal Metrology (Packaged Commodities) Rules, 2011, any person who imports or manufactures packaged goods for sale must ensure the package carries certain statutory declarations. The LMPC registration certificate is proof that the importer or packer is authorized to bring packaged goods into the Indian market.
Who Needs LMPC for Stethoscopes?
- Any importer who imports pre-packaged stethoscopes for resale in India
- Any Indian manufacturer who packages stethoscopes in retail units for sale
- Distributors who repack or relabel stethoscopes under their own brand (private labelling)
Mandatory Label Declarations for Stethoscope Packages Under LMPC
| Declaration Required | Notes |
| Name and address of the manufacturer / importer / packer | Full postal address with PIN code |
| Country of origin | Mandatory for imported stethoscopes |
| Generic name of the product | 'Stethoscope' — not just the brand name |
| Net quantity | By number (e.g., 1 unit) |
| Month and year of manufacture / import | MM/YYYY format |
| Maximum Retail Price (MRP) | INR inclusive of all taxes |
| Customer care helpline number | Mandatory since 2022 |
| CDSCO Licence / Registration Number | Print the MD-14 or MD-5 number on the label |
LMPC Certificate Renewal
The LMPC certificate is typically valid for one year. Renewal must be done before expiry — do not wait for the last moment. Most state legal metrology departments now accept online renewal applications through their state portal. The renewal fee is nominal (approximately INR 300 to INR 1,000 depending on the state).
2026 Regulatory Updates — What Has Changed for Stethoscope Registration
Keeping up with CDSCO notifications is critical because the rules do evolve. Here are the most important updates relevant to stethoscope registration and compliance in 2026:
Update 1: Mandatory UDI (Unique Device Identifier) for Class A Devices
CDSCO issued a notification in 2025 indicating that UDI implementation would be progressively extended to Class A medical devices. By 2026, manufacturers and importers of stethoscopes are expected to apply for and display a UDI on their products and packaging. This aligns India with global practices (US FDA and EU MDR already mandate UDI). The UDI database is being built on the SUGAM portal, and applicants will need to register their devices in the UDI database as part of the registration process going forward.
Update 2: Strengthened Labelling Requirements
A 2025 CDSCO circular reinforced the requirement for medical device labels to carry the CDSCO license or registration number directly on the primary packaging. For importers of stethoscopes, this means you can no longer rely on a sticker over an original foreign label — the India-specific label must be a permanent printed label or a well-affixed, non-removable label with all mandatory information.
Update 3: Online Renewal and Digitization
CDSCO has been pushing all registration and licensing processes fully online through the SUGAM portal. Physical document submissions are being phased out. All applications for stethoscope registration in 2026 are expected to be submitted and tracked entirely online.
Update 4: Increased Focus on Post-Market Surveillance
CDSCO's enhanced vigilance programme is now conducting random market surveillance checks on medical devices including diagnostic instruments. Importers found selling unregistered stethoscopes are facing show-cause notices and product seizures at a higher rate than before. If you have been delaying your registration, 2026 is the year to get it done.
Update 5: State-Level LMPC Portals Going Live
Several states including Maharashtra, Karnataka, Tamil Nadu, and Delhi have launched or upgraded their online Legal Metrology portals, making LMPC certificate applications and renewals fully digital. This significantly reduces the turnaround time for LMPC certificates in these states.
Common Mistakes to Avoid in Stethoscope Registration
These are the most frequent errors that lead to delays, rejections, or compliance issues:
| Mistake | Why It Causes Problems | How to Avoid It |
| Incorrect product classification | Applying under wrong class leads to rejection | Verify classification on CDSCO notified list before applying |
| FSC not apostilled / authenticated | CDSCO rejects unauthenticated FSCs | Get FSC authenticated by the relevant authority in country of origin |
| Mismatch in company name across documents | Causes deficiency notices | Ensure all documents use the exact same legal entity name |
| Incorrect labelling format | Label deficiencies are very common | Follow MDR 2017 Schedule III and LMPC Rules checklist carefully |
| Not renewing LMPC on time | Selling with expired LMPC is a violation | Set calendar reminders 60 days before expiry |
| Ignoring post-market obligations | Can lead to licence suspension | Maintain distribution records and report adverse events |
Conclusion
Medical device registration for a stethoscope in India is genuinely manageable when you understand the system and approach it methodically. The regulatory framework exists to protect patients and ensure that devices sold in India meet basic safety and quality standards — and your compliance is a direct contribution to that goal.
The most important things to take away from this guide are: (1) register your stethoscope before you start selling, not after; (2) maintain both your CDSCO licence/registration and your LMPC certificate; (3) stay updated with CDSCO notifications because the regulatory environment is actively evolving in 2026; and (4) invest in proper regulatory counsel if you are doing this for the first time — the cost of professional help is far smaller than the cost of rejection, seizure, or penalty.
If you are ready to begin the registration process or have questions about a specific product or scenario, consult a CDSCO-experienced regulatory affairs professional Silvereye Certifications who can review your exact documents and advise accordingly.
Frequently Asked Questions
Is a stethoscope a medical device in India?
Yes. Under Schedule I of the Medical Devices Rules, 2017, stethoscopes are notified medical devices in India. They are classified as Class A (low risk) devices.
Can I sell stethoscopes online in India without registration?
Yes. Under Schedule I of the Medical Devices Rules, 2017, stethoscopes are notified medical devices in India. They are classified as Class A (low risk) device
Do I need a separate license for each model of stethoscope?
Generally, yes. Each distinct product (model) with different specifications or intended use needs its own registration entry. However, product families with very similar specifications can sometimes be covered under a single application with an attachment listing all variants — consult a regulatory expert for the optimal approach.
What is the difference between CDSCO registration and LMPC certificate for stethoscopes?
CDSCO registration (Form MD-14 or MD-5) is about confirming that your device meets medical device safety requirements and authorizes you to legally sell a medical device in India. The LMPC certificate is about packaged commodity compliance — it ensures your retail packaging meets the mandatory labelling requirements under the Legal Metrology Act. Both are required for most commercial stethoscope imports and sales.
How long does CDSCO stethoscope registration take in 2026?
With complete and accurate documentation, expect 60 to 90 days for Class A devices. If there are deficiencies in your documents, each round of back-and-forth can add another 30 to 45 days.
What happens if my LMPC certificate expires?
Selling stethoscopes with an expired LMPC certificate makes you liable for penalties under the Legal Metrology Act. The penalty can range from a fine to prosecution in serious cases. Renew your LMPC certificate well before the expiry date.
Can a foreign company directly register a stethoscope with CDSCO?
A foreign manufacturer without an Indian entity must appoint an Authorised Indian Representative (AIR) — an Indian company or individual — who applies for and holds the CDSCO import licence on their behalf. The foreign company cannot directly hold the Indian registration.






































