- Every hair oil brand entering India must hold a valid CDSCO Hair Oil Import Registration Certificate — no exceptions, no shortcuts.
- The COS-1 application, filed through CDSCO's SUGAM portal, covers up to 50 products and multiple variants in a single submission.
- Standard processing takes approximately 180 working days; errors in documents or labels can add months to your timeline.
- Appointing an Authorized Indian Representative (AIR) is the fastest, most compliant route for foreign manufacturers in 2026.
Introduction
If you are a foreign hair oil brand thinking about entering the Indian beauty market, the first thing you need to understand is this: India does not allow a single bottle of imported hair oil to clear customs without a valid Import Registration Certificate issued by the Central Drugs Standard Control Organization (CDSCO). This is not paperwork for the sake of paperwork. It is the legal gateway to one of the world's fastest-growing personal care markets — and it is non-negotiable.


India's hair care segment is projected to cross USD 6 billion by 2027, with international brands driving a significant share of the premium end. Argan oil blends from Morocco, scalp serums from South Korea, Ayurvedic oil formulations from Europe — consumers want them. Retailers want to stock them. But none of that movement is possible without CDSCO hair oil import registration in India.
Under Rule 21 of the Drugs and Cosmetics Act, 1940, and the Cosmetics Rules, 2020, all cosmetic products imported into India — including every variant, every SKU, every pack size of your hair oil — must hold a valid Import Registration Certificate before a single shipment enters the country. Fail to comply, and your consignment gets held at customs, returned to the country of origin, or confiscated. In some cases, penalties apply.
What Is CDSCO Hair Oil Import Registration and Why Does It Regulate Cosmetic Imports?
CDSCO — the Central Drugs Standard Control Organisation — is India's national regulatory authority for pharmaceuticals, medical devices, and cosmetics. It operates under the Ministry of Health and Family Welfare and functions as the apex body for ensuring product safety standards in the Indian market.
When it comes to cosmetics, CDSCO's mandate is clear: no imported cosmetic product can be sold in India unless it has been registered and approved by the authority. Hair oils fall squarely under the definition of cosmetics in Indian law — regardless of whether they are marketed as Ayurvedic, natural, clinical, or treatment-focused.
The SUGAM portal (https://sugam.cdsco.gov.in) is CDSCO's official online platform where all import registration applications for cosmetics are submitted and processed. There is no offline route. All document uploads, fee payments, query responses, and certificate downloads happen through this portal.
What Is the Difference Between COS-1 and COS-2?
Two forms sit at the heart of the CDSCO cosmetic import registration process. Understanding them is the first step to getting your application right.
| Form | Purpose & What It Means for You |
| Form COS-1 | The application form you submit to CDSCO requesting an Import Registration Certificate. Filed online through the SUGAM portal. One COS-1 can cover up to 50 cosmetic products, multiple variants, and multiple manufacturing sites. |
| Form COS-2 | The Import Registration Certificate itself — issued by CDSCO after successful review of your COS-1 application. This is your legal authorization to import and sell the registered hair oil products in India. Valid for 5 years from the date of issue. |
| Retention Fee | To keep your COS-2 active after 5 years, you must pay a retention fee before the expiry date. Missing this deadline means your registration lapses and imports must stop. |
In practical terms: COS-1 is what you file, and COS-2 is what you get. The period between the two — the CDSCO review and query cycle — is where most delays happen if the application is not prepared correctly.
Who Is Eligible to File a COS-1 Application with CDSCO?
CDSCO allows the COS-1 application to be filed by one of three parties:
- The foreign manufacturer directly, if they choose to register on the SUGAM portal
- An Authorized Indian Representative (AIR) — an Indian agent appointed by the foreign manufacturer through a valid Power of Attorney
- An Indian subsidiary or wholly owned entity of the foreign manufacturer incorporated in India
For most international brands — particularly those based in Europe, Southeast Asia, the Middle East, or East Asia — the Authorized Indian Representative route is the cleanest and most practical. The AIR manages the SUGAM portal account, handles all document coordination, responds to CDSCO queries in real time, and stays accountable for the registration lifecycle.
Choosing the right AIR for your CDSCO cosmetic import registration in India is not a minor decision. Delays caused by an inexperienced agent can cost months and result in shipments being turned back at the Indian border — even after you've already invested in manufacturing, packaging, and logistics.
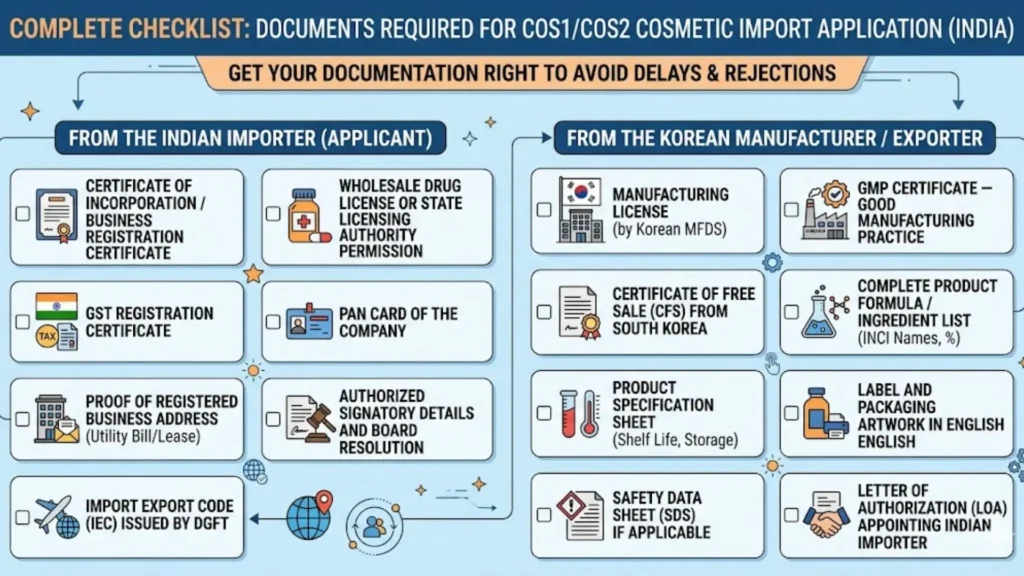
Complete Document Checklist for CDSCO Hair Oil Import Registration
Document completeness is the single most important factor in avoiding CDSCO queries and keeping your timeline on track. Here is the full list of documents required for a hair oil COS-1 application in 2026:
Documents Required from the Foreign Manufacturer
- Manufacturing License — issued by the competent regulatory authority in the country of origin (e.g., FDA, TGA, MFDS, ANSM)
- Free Sale Certificate (FSC) — confirming the hair oil is freely marketed in the home country without restrictions
- Power of Attorney — authorizing the Indian agent or representative to act on behalf of the manufacturer for CDSCO purposes
- Complete Ingredient List — in INCI (International Nomenclature of Cosmetic Ingredients) format, with percentage concentrations for each ingredient
- Product Formulation & Composition Details — showing the full formula with manufacturing process notes
- Manufacturing Site Information — full name, address, contact details, and capacity
- Company Profile — brief overview of the manufacturer's history, capabilities, and market presence
Product-Specific Documents
- Proposed label artwork for the Indian market — compliant with Indian labeling norms (discussed in detail below)
- Product category classification — for hair oils, this is typically 'Hair and Scalp Care — Hair Oil'
- Safety Data and Stability Testing Reports — demonstrating that the product is safe for consumer use under Indian climatic conditions
- Method of Analysis — detailing how the product's quality is tested
- Specification Sheets — for each product and each variant included in the application
- Shelf life data with supporting stability study reports
Documents from the Indian Applicant or Authorized Representative
- Business Registration Certificate of the Indian importer or agent
- GST registration number and PAN details
- Correspondence address and contact information in India
- Authorization Letter — signed by the foreign manufacturer, designating the Indian entity as the authorized representative
One common question we receive: 'Does every variant need a separate set of documents?' The answer is no — but every variant must be listed in the COS-1 application, and any variant-specific data (such as different ingredients or fragrance compositions) must be documented clearly. The base manufacturer documents can be shared across all products from the same manufacturing site.

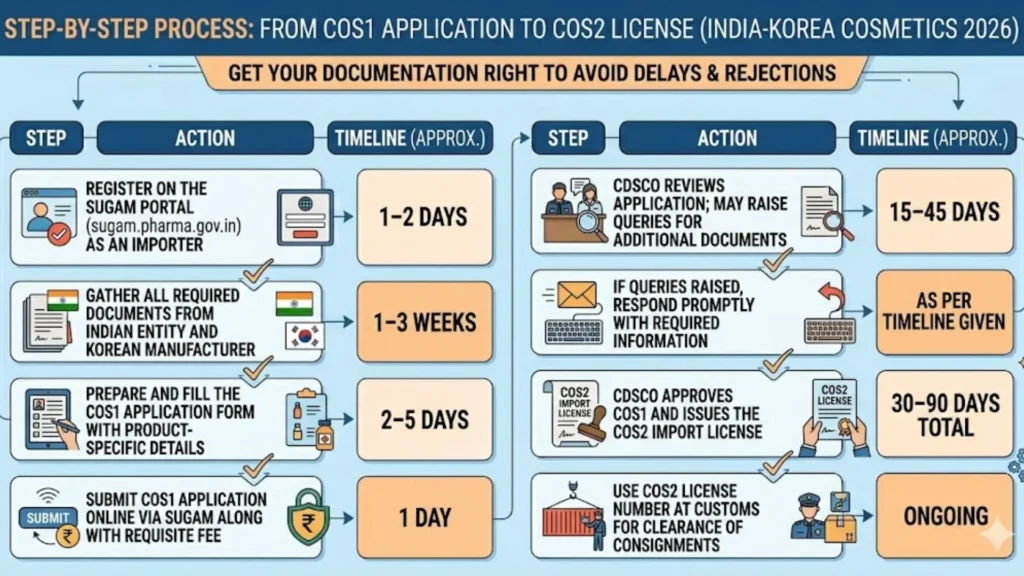
How to Register Hair Oil with CDSCO: Step-by-Step Process via SUGAM Portal
Here is exactly how the CDSCO hair oil import registration process works in 2026, from first contact with the portal to receiving your COS-2 certificate:
Step 1 — SUGAM Portal Registration
Your Authorized Indian Representative registers on CDSCO's SUGAM portal. This involves providing company details, address proof, and contact information. Verification can take a few days before the account is active.
Step 2 — COS-1 Application Filing
Once the account is live, the AIR begins filling out the COS-1 application form. Every product to be imported must be entered here — product name, brand name, category, variants, pack sizes, manufacturing site, and applicant details. Accuracy at this stage directly determines whether CDSCO raises queries later.
Step 3 — Document Upload
All required documents are uploaded in PDF format through the portal. CDSCO is specific about file naming, format, and organization. This is where most first-time applicants make errors — uploading the wrong version of a document, an expired Free Sale Certificate, or a label that does not meet Indian labeling norms. Errors here are the most common cause of rejection and delay.
Step 4 — Government Fee Payment
Fee payment is made online through the SUGAM portal after the application is submitted. The current government fee structure is approximately USD 1,000 per product category and USD 500 per additional variant within the same category. These fees are payable to CDSCO and are separate from any professional or agency fees.
Step 5 — Technical Review by CDSCO
CDSCO's technical reviewers assess the application. They examine ingredients against the BIS IS 4707 permitted list, verify label compliance, review safety data, and check document authenticity. If they have questions — about a specific ingredient, a label claim, or a missing document — they raise a query on the portal. Your agent must respond accurately and promptly. Multiple query rounds can each add weeks to the timeline.
Step 6 — COS-2 Certificate Issued
Once CDSCO is satisfied with the application and all queries are resolved, the Import Registration Certificate (Form COS-2) is issued on the SUGAM portal. You can now legally import and sell the registered hair oil products in India. The certificate is valid for five years.

CDSCO Cosmetic Registration Timeline
| Stage | Typical Duration | What Can Cause Delays |
| SUGAM account setup | 3–7 working days | Incomplete company details |
| COS-1 application preparation | 1–3 weeks | Missing documents, non-compliant labels |
| CDSCO technical review (no queries) | ~180 working days | High application volume |
| CDSCO query response cycle | +30–90 days per query round | Incorrect or incomplete responses |
| COS-2 certificate issuance | 7–14 days after final approval | Portal processing time |
The 180 working days figure is CDSCO's internal processing guideline for a complete, clean application. In practice, applications with missing documents, expired certificates, non-compliant label artwork, or unsupported claims can take considerably longer — sometimes a year or more from first submission to certificate issuance.
The single most effective way to reduce your CDSCO registration timeline in India is to submit a complete, accurate application with fully compliant label artwork on day one. No shortcuts here pay off — they all cost time at the other end.
CDSCO Hair Oil Import Registration Fees
| Fee Type | Amount (Approx.) | Notes |
| COS-1 Application Fee (first product category) | USD 1,000 | Payable online via SUGAM portal |
| Additional variants within same category | USD 500 per variant | Applies to different sizes, fragrances, formulations |
| COS-2 Retention Fee (renewal after 5 years) | Similar to initial fee | Must be paid before certificate expiry |
| New Cosmetic (COS-3) Approval Fee | As applicable | Required only for products with novel/unlisted ingredients |
These are government-charged fees payable directly to CDSCO through the portal. Professional fees charged by an Authorized Indian Representative for managing the full registration process are separate and vary by agency, scope, and complexity.
Hair Oil Label Requirements for Indian Market: What Must Appear on Your Label
Labeling is one of the most frequently raised query areas in CDSCO cosmetic import registration applications. Your hair oil's Indian market label must be reviewed and approved as part of the COS-1 process. Here is what CDSCO requires on the label:
- Brand name and product name (exactly as registered)
- Complete ingredient list in INCI format, listed in descending order of concentration
- Net contents — volume (ml) or weight (g) in metric units
- Name and full address of the Indian importer or authorized representative
- Country of origin — e.g., 'Made in France', 'Made in South Korea', 'Made in Japan'
- Manufacturing date and expiry date or 'best before' date
- Batch number or lot number for traceability
- Applicable cautions — e.g., 'For external use only. Keep away from eyes. In case of contact, rinse thoroughly.'
- Import Registration Certificate number (COS-2 number) — added to the label after the certificate is issued
What About Label Claims? This Is Where Brands Often Go Wrong.
This is a critical area that catches many international brands off-guard when applying for CDSCO hair oil import registration in India. Claims on your label must fall within CDSCO's permitted cosmetic claim guidelines. Here's the key distinction:
| Acceptable Cosmetic Claims (Generally Permitted) | Problematic Claims (Trigger Re-classification Risk) |
| 'Nourishes and conditions hair' | 'Clinically proven to cure hair fall' |
| 'Leaves hair soft and smooth' | 'Reverses alopecia or baldness' |
| 'Adds shine and manageability' | 'Stimulates dormant follicles' |
| 'Strengthens hair strands' | 'Promotes hair regrowth' |
| 'Moisturises scalp' | 'Treats dandruff or scalp conditions' |
| 'Reduces visible breakage' | 'Restores damaged hair follicles' |
Claims that cross into therapeutic or drug-like territory — 'treats,' 'cures,' 'reverses,' 'restores' — can trigger CDSCO to re-classify your hair oil as a drug rather than a cosmetic. This means your product would need a drug license, not an Import Registration Certificate, which is an entirely different and far more complex process. Review your label claims carefully before filing.
What Is a 'New Cosmetic' Under CDSCO Rules — And Does Your Hair Oil Qualify?
Most hair oil products will follow the standard COS-1 route. But there is a scenario you need to be aware of, particularly if your formulation includes cutting-edge actives, novel botanical extracts, or ingredients that are not recognized in standard international scientific literature.
If your hair oil contains an ingredient that has never been used in cosmetics anywhere in the world — not listed in any national or international cosmetic ingredient database — CDSCO classifies the product as a 'New Cosmetic.' Before you can file a COS-1 application, you must first obtain New Cosmetic Approval through Form COS-3 from the Central Licensing and Standardization Authority (CLSA).
This COS-3 process requires submission of full safety evaluation data as per BIS IS 4011:2018, including toxicological studies, dermal sensitization data, and full safety assessment reports. It is a significantly more rigorous process and adds considerable time to your market entry timeline.
How do you know if your ingredient qualifies as 'new'? If it appears in the INCI dictionary, in any EU/US/ASEAN cosmetic ingredient list, or in any published scientific literature, it is likely not a 'new' ingredient. If you are using a proprietary botanical extract patented by your lab with no prior cosmetic use history, speak to your regulatory consultant before filing.
Post-Registration Compliance: What Happens After You Get Your COS-2 Certificate
Receiving your Import Registration Certificate is the beginning of your compliance obligations in India, not the end. As a registered importer of hair oil in India, here is what you are legally required to maintain:
- Every shipment must use only the products, variants, formulations, and manufacturing sites listed in your COS-2 certificate. Importing an unregistered variant — even a different pack size — is non-compliant.
- Any change to your product's label, formulation, ingredients, variant range, manufacturer, or manufacturing site must be notified to and approved by CDSCO before that change affects an import shipment.
- The COS-2 certificate is valid for five years. You must pay the retention fee and file for renewal before the expiry date. A lapsed certificate means all imports must stop.
- Maintain complete import records for each shipment — including invoice, packing list, bill of lading, and certificate copies — for audit and compliance inspection purposes.
- Ensure ongoing compliance with BIS IS 4707 ingredient standards. If any ingredient in your formulation is removed from the permitted list in future, you must update your formulation or discontinue the product.
Common Mistakes That Delay CDSCO Hair Oil Import Registration
- Submitting an expired Free Sale Certificate — CDSCO requires certificates that are current at the time of application
- Using a label with drug-like claims — triggers re-classification review and significant delays
- Uploading ingredient lists without percentage concentrations — CDSCO requires percentages in INCI format
- Incorrect product category classification — placing a hair oil in the wrong category creates query cycles
- Power of Attorney not notarized or apostilled — CDSCO may reject documents without proper authentication
- Stability test reports from unrecognized labs — reports must meet CDSCO's quality and methodology standards
- Missing the retention fee deadline — results in certificate lapse and forced suspension of all imports
Why Work with an Experienced CDSCO Registration Consultant for India?
The CDSCO cosmetic import registration process is thorough, detail-intensive, and unforgiving of errors. For foreign brands without a deep understanding of Indian regulatory language, portal mechanics, and document standards, navigating it alone is a significant risk.
- An experienced Authorized Indian Representative — one who has handled dozens of successful CDSCO hair oil import registrations — brings several concrete advantages:
- Label review before submission, not after rejection — ensuring your Indian label meets CDSCO norms before it becomes a query
- Document preparation to the highest standard — reducing query-raising significantly and keeping your timeline predictable
- Real-time SUGAM portal management — so no query sits unanswered and no deadline is missed
- Post-registration support — for variant additions, label changes, manufacturer updates, and certificate renewals
- A single point of accountability in India — removing the coordination burden from your overseas team entirely
Silvereye Certifications acts as an Authorized Indian Representative for cosmetic brands from across Asia, Europe, the Middle East, and the Americas. With a track record of successful CDSCO registrations across hair care, skin care, and personal care categories, Silvereye manages the full import registration lifecycle — from first document review to COS-2 issuance and beyond.
Conclusion: CDSCO Registration Is Your Entry Ticket to India's Hair Care Market
India's hair care and beauty market in 2026 is not slowing down. Demand for international hair oil brands — whether infused with Moroccan argan, Korean fermented actives, French botanical blends, or Ayurvedic herbs — is growing faster than most markets globally. But there is only one legal door into this market for imported products: CDSCO Import Registration.
The process is methodical, documented, and entirely navigable with the right preparation. A complete COS-1 application with fully compliant label artwork, accurate INCI ingredient lists, current certificates, and properly authenticated documents will move through CDSCO's review process as efficiently as the system allows. The brands that get stuck — for months, sometimes years — are the ones who cut corners on document preparation, used non-compliant label claims, or worked with agents who did not understand CDSCO's specific expectations.
If you are a foreign hair oil brand planning to enter the Indian market in 2026 or beyond, start your CDSCO registration process as early as possible. The 180-working-day timeline means that decisions made today determine when your product reaches Indian shelves tomorrow.
Frequently Asked Questions
Can I start selling my hair oil in India while my CDSCO registration is pending?
No. Indian customs will not clear cosmetic imports without a valid Import Registration Certificate. Attempting to import before your COS-2 is issued can result in confiscation and penalties.
How many products can I include in a single COS-1 application?
One COS-1 application can cover up to 50 cosmetic products, with multiple variants and multiple manufacturing sites included. This makes it highly efficient for brands with a full hair care range.
Do I need a separate CDSCO registration for each hair oil variant?
Not a separate application — but each variant must be listed and documented within the COS-1. The fee structure does apply per variant (USD 500 per additional variant within the same category).
What happens if CDSCO raises a query on my application?
Your Authorized Indian Representative receives the query notification on the SUGAM portal and must respond with the requested information, corrected documents, or additional data. Each query round typically adds 30 to 90 days to the timeline, depending on the complexity of the query and how promptly it is addressed.
Is CDSCO registration the same as BIS certification for cosmetics?
No. CDSCO Import Registration (COS-1/COS-2) is the import authorization process. BIS IS 4707 is the Indian standard for permitted cosmetic ingredients that your product must comply with. Both are relevant — CDSCO registration is the certificate you need, and BIS IS 4707 compliance is the ingredient standard your formulation must meet to obtain that certificate.
How often do I need to renew my CDSCO cosmetic import registration?
The COS-2 certificate is valid for 5 years. You must pay a retention fee before the expiry date to keep it active. Unlike the initial registration, renewal is generally faster as long as your product, formulation, and labels remain unchanged.
What is the cost of CDSCO hair oil import registration in India?
Government fees are approximately USD 1,000 for the first product category and USD 500 for each additional variant. Professional fees for an Authorized Indian Representative are charged separately and vary by provider.
Can I register hair oil products from multiple countries in one application?
Products from different countries of manufacture can be included in one COS-1 application, but each manufacturing site must be separately documented with its own Manufacturing License, Free Sale Certificate, and site details.





































