- Ventilators are Class C (moderate-to-high risk)medical devices under India's MDR 2017 — CDSCO registration is mandatory before any import, manufacture, or sale in India.
- Importers must file Form MD-14 on the SUGAM portal through an authorized Indian agent; CDSCO then issues Form MD-15— the legal import license valid perpetually with a retention fee due every 5 years.
- Indian manufacturers apply via Form MD-9 to CDSCO supported by ISO 13485:2016 certification, Plant Master File, and a complete technical dossier — license issued as Form MD-10.
- BIS certification under IS 13450 (Part 2/Sec 12): 2023is separately mandatory for all ventilators sold in India — compliance deadline under BIS Scheme X extended to 1 September 2026.
Introduction: Why CDSCO Registration for Ventilators Cannot Be Skipped
If you are a manufacturer or an overseas company planning to import ventilators into India, CDSCO registration for ventilators is the single most important regulatory step before your product touches the Indian market. Without the correct CDSCO license, your ventilator cannot legally be manufactured, imported, distributed, or sold anywhere in India — not to a private hospital, not to a government facility, not through any channel.


India's medical device market is one of the fastest-growing in Asia and is projected to reach USD 50 billion by 2030. Ventilators, in particular, came under intense regulatory scrutiny post the COVID-19 pandemic and have remained one of the most closely monitored life-support devices in the country.
The Central Drugs Standard Control Organization (CDSCO), operating under the Ministry of Health and Family Welfare (MoHFW), Government of India, regulates the entire lifecycle of medical devices — from classification and licensing to post-market surveillance. This guide covers everything — classification, forms, documents, BIS standards, fees, timelines, and the most important 2025–2026 updates — so you enter the CDSCO registration process prepared, not surprised.
What Is CDSCO and Why Does It Regulate Ventilators?
CDSCO stands for the Central Drugs Standard Control Organization, which functions under the Drugs and Cosmetics Act, 1940 and is the primary national regulator for pharmaceuticals and medical devices in India. The Drug Controller General of India (DCGI) heads CDSCO and serves as the Central Licensing Authority (CLA) for high-risk medical devices including ventilators.
Ventilators are life-critical devices. A malfunction, incorrect labeling, or substandard performance can directly result in patient death. This is why the Indian government has classified them as Class C devices under the Medical Devices Rules (MDR), 2017 — requiring mandatory CDSCO approval before they can enter the Indian market.
All applications, fee payments, and document submissions happen digitally through the SUGAM portal (https://sugam.gov.in). Paper applications to CDSCO are no longer accepted as of 2022.
How Are Ventilators Classified Under CDSCO and MDR 2017?
Before you file a single form or pay a rupee in government fees, get the device classification right. A wrong classification can lead to application rejection, legal penalties, and costly delays. Under MDR 2017, medical devices are divided into four risk-based categories:
| Class | Risk Level | Examples | Licensing Authority |
| Class A | Low Risk | Surgical dressings, bandages, tongue depressors | State Licensing Authority (SLA) |
| Class B | Low-Moderate Risk | Pulse oximeters, syringes, cardiac monitors | State Licensing Authority (SLA) |
| Class C | Moderate-High Risk | ICU lung ventilators, anaesthesia machines, high-frequency ventilators | Central Licensing Authority (CDSCO/DCGI) |
| Class D | Highest Risk | Heart valves, implantable defibrillators, heart-lung machines | Central Licensing Authority (CDSCO/DCGI) |
| Key Point: Standard ICU ventilators and high-frequency ventilators are classified as Class C. This means your application goes directly to CDSCO (the Central Licensing Authority), not the State FDA. Applying to the wrong authority is one of the most common beginner mistakes. |
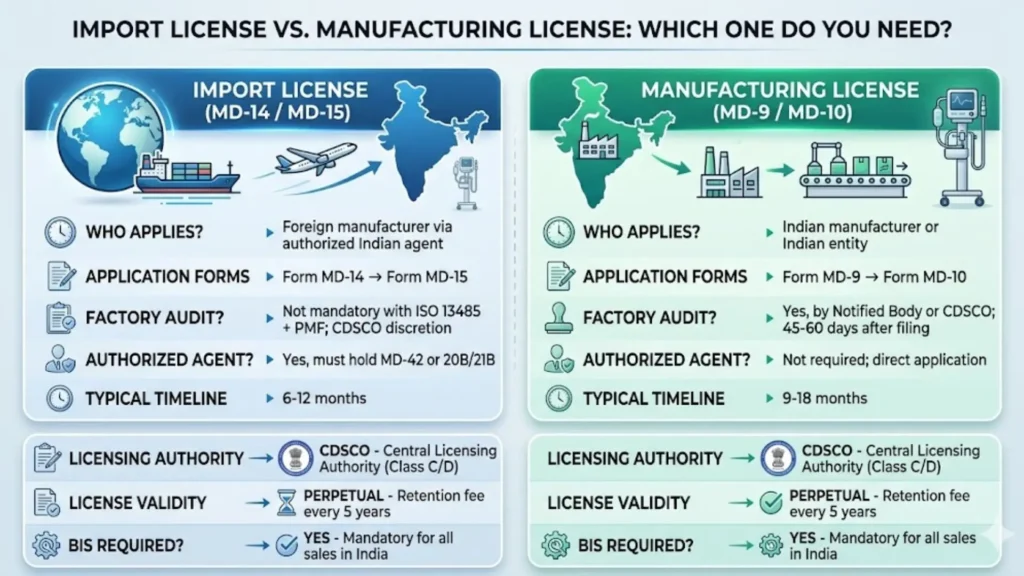
Import License vs. Manufacturing License — Which One Do You Need?
Your path depends entirely on where your ventilator is manufactured. Foreign manufacturers importing into India take a different route from Indian manufacturers making ventilators domestically. Here is a clear comparison:
| Factor | Import License (MD-14 / MD-15) | Manufacturing License (MD-9 / MD-10) |
| Who Applies? | Foreign manufacturer via authorized Indian agent | Indian manufacturer or Indian entity |
| Application Form | Form MD-14 (application) → Form MD-15 (license) | Form MD-9 (application) → Form MD-10 (license) |
| Licensing Authority | Central Licensing Authority — CDSCO | Central Licensing Authority — CDSCO (Class C/D) |
| Factory Audit? | Not mandatory if ISO 13485 + PMF submitted; CDSCO may audit at discretion | Yes — Notified Body or CDSCO auditor inspects plant (45–60 days after filing) |
| Authorized Agent? | Yes — must hold MD-42 or Form 20B/21B license | Not required; manufacturer applies directly |
| Typical Timeline | 6–12 months | 9–18 months (due to inspection cycle) |
| License Validity | Perpetual — retention fee every 5 years | Perpetual — retention fee every 5 years |
| BIS Required? | Yes — mandatory for all products sold in India | Yes — mandatory for all domestic sales |

| CDSCO FAQ Addendum-01 (April 2024) confirmed that each manufacturing site requires a separate application and separate government fees — even if multiple sites belong to the same company. |
Form MD-14 — The Import Application for Foreign Manufacturers
Form MD-14 is the gateway for any foreign ventilator manufacturer wanting to legally sell in India. It is submitted exclusively through the SUGAM portal. Paper submissions are not accepted under any circumstances.
Who Can Submit Form MD-14?
The application must be filed by an authorized Indian agent (also called an Authorized Indian Representative or IAA) on behalf of the foreign manufacturer. The agent must hold one of the following:
- A valid wholesale license in Form MD-42, OR
- A wholesale drug license in Form 20B and Form 21B, OR
- A manufacturing license in Form MD-5 or MD-9
The foreign manufacturer must issue a Power of Attorney (PoA) authorizing the Indian agent. The PoA must be apostilled by the Indian Embassy in the country of origin, authenticated by a First-Class Magistrate in India, or notarized by an equivalent authority.
Key Information Required in Form MD-14
| Field | Details Required |
| Device Name | Exact name as on the Free Sale Certificate / CE / FDA approval |
| Model Number | Must match FSC, device labeling, and all technical documents exactly — zero tolerance for mismatch |
| Intended Use | Clinical purpose — ICU ventilation, neonatal ventilation, transport ventilation, etc. |
| Manufacturing Site | Name and address of the actual manufacturing plant (not company headquarters) |
| Brand Name | Must appear on the Free Sale Certificate; otherwise state 'Not Applicable' |
| Reference Approval | CE, FDA, TGA, Health Canada, PMDA (Japan) — GHTF country approvals strengthen the dossier |
| Classification | Class A / B / C / D with MDR 2017 justification |
| Critical: All data in Form MD-14 — model number, brand name, manufacturer name — must match 100% with the Free Sale Certificate. Any mismatch will trigger a deficiency letter from CDSCO and can add 3–6 months to your timeline. |
Form MD-15 — The CDSCO Import License
Form MD-15 is the actual import license issued by CDSCO after it reviews and approves your Form MD-14 application. This is the document your customs team, buyers, and regulatory inspectors will ask for. Without it, your shipment can be seized at the port.
What Does an MD-15 License Contain?
- Name and address of the authorized Indian agent
- Name of the foreign manufacturer and the manufacturing site address
- Device name and exact model number
- Classification category (Class C for standard ventilators)
- Conditions of import — labeling requirements, storage, etc.
- Date of issue
Validity and Retention Fee
As per the Medical Devices (Amendment) Rules, 2020, Form MD-15 is now valid in perpetuity — it does not expire after 5 years as it did under older rules. However, you must pay a retention fee every 5 years to keep the license active. Missing the payment triggers automatic suspension.
| Entity Type | Retention Fee — Per Device | Retention Fee — Per Site |
| Indian Manufacturer (Class A/B) | Rs. 500 per device | Rs. 5,000 per site |
| Indian Manufacturer (Class C/D) | Rs. 1,000 per device | Rs. 50,000 per site |
| Foreign Importer (Class A) | USD 50 per device | USD 1,000 per site |
| Foreign Importer (Class B) | USD 1,000 per device | USD 2,000 per site |
| Foreign Importer (Class C/D) | USD 1,500 per device | USD 3,000 per site |
| Late payment attracts a penalty of 2% of the retention fee per month for up to 180 days. After 180 days without payment, the license is automatically cancelled and you must file a fresh application. |
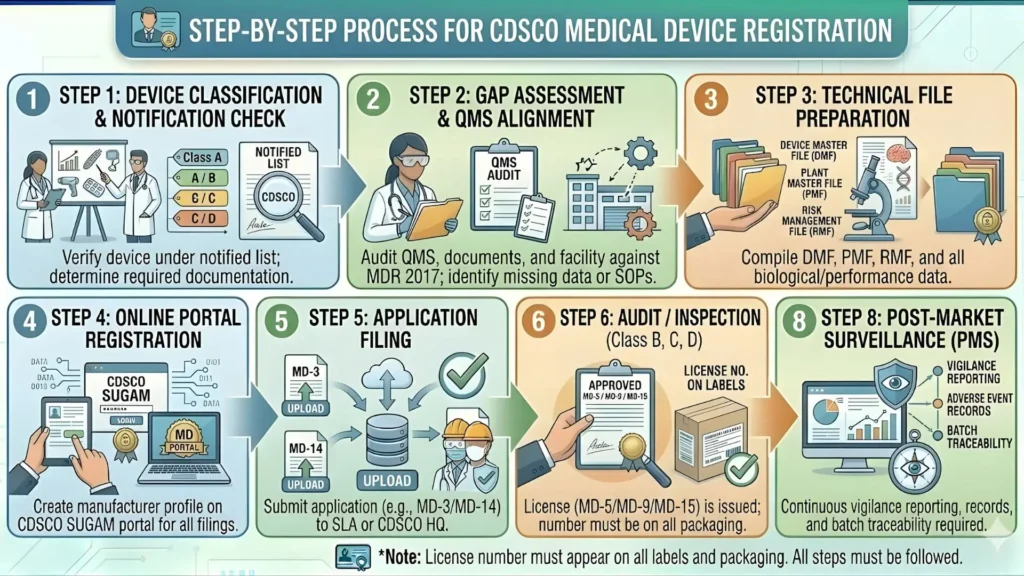
Step-by-Step CDSCO Registration Process for Ventilators (2026)
For Importers — Foreign Manufacturers
- Confirm Device Classification — Verify that your ventilator falls under Class C in CDSCO's notified medical device list. Engage a qualified regulatory consultant if you are unsure.
- Appoint an Authorized Indian Agent (AIA) — Select an Indian entity holding a valid MD-42 wholesale license or equivalent. Execute a notarized/apostilled Power of Attorney. Important: Do not appoint your commercial distributor as your regulatory agent — if you change distributors, you lose the CDSCO license.
- Prepare Your Technical Dossier — This is the most time-intensive step. Your dossier must be in the Indian Device Master File (DMF) format. Having CE or FDA clearance significantly accelerates CDSCO's technical review.
- Register on the SUGAM Portal — Your authorized Indian agent creates or logs into an account on https://sugam.gov.in. All Form MD-14 applications, fee payments, and query responses happen here.
- Pay the Government Fee — For Class C devices: USD 1,500 per manufacturing site + USD 1,000 per device. Payment via Bank of Baroda TR-6 challan or Electronic Clearance System (ECS) for international payments.
- Submit Form MD-14 Online — The authorized agent files the complete application with all technical documents and the payment receipt on the SUGAM portal.
- CDSCO Technical Review and Query Resolution — CDSCO reviews the dossier. Queries are posted on the portal. You typically get 30–90 days to respond. Late or incomplete responses restart the clock.
- Receive Form MD-15 — Once satisfied, CDSCO issues the import license. Your authorized agent can now legally import, stock, and sell your ventilators in India.
For Indian Manufacturers
- Confirm Classification and Regulatory Strategy — Class C ventilators require CDSCO (CLA) approval. Do not approach the State FDA for Class C devices.
- Establish ISO 13485:2016 Certified QMS — Your quality management system must be ISO 13485 certified from an accredited body. This is non-negotiable for Class C devices.
- Prepare Plant Master File (PMF) and Device Master File (DMF) — Cover all Schedule 3 compliance requirements under MDR 2017 including risk management per ISO 14971.
- Register on SUGAM Portal and Pay Fee — File Form MD-9 with the applicable government fee in Indian Rupees.
- Notified Body Audit or CDSCO Inspection — A CDSCO-empanelled Notified Body visits your manufacturing plant within 45–60 days. All non-conformities must be resolved before the license is granted.
- Query Resolution and License Grant — After audit clearance, respond to any CDSCO queries on SUGAM. Once satisfied, CDSCO issues the manufacturing license in Form MD-10.
- Obtain BIS Certification — Separately apply for BIS certification under IS 13450 (Part 2/Sec 12): 2023 before commercial distribution begins.

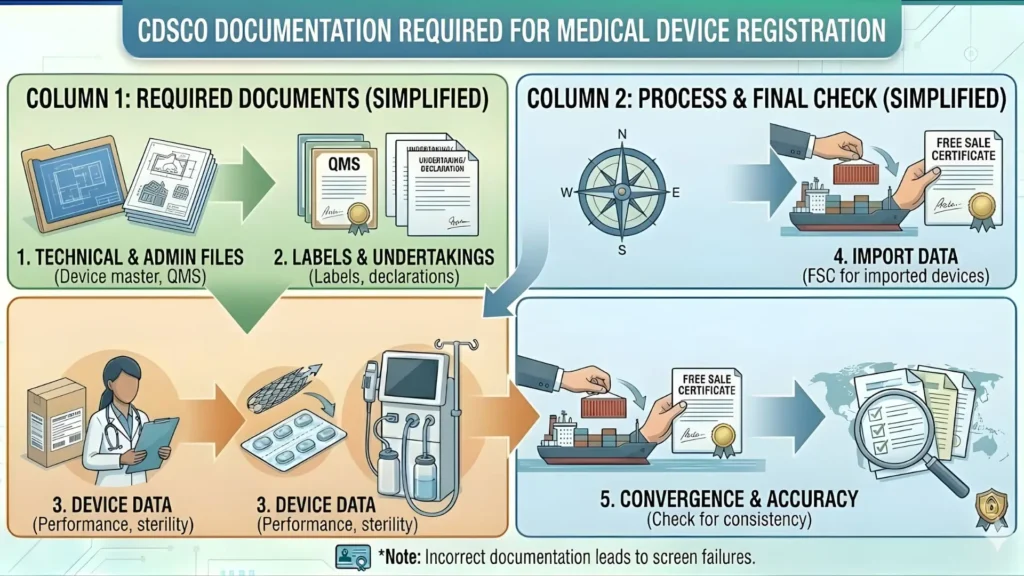
Documents Required for CDSCO Ventilator Registration
A complete, accurate dossier submitted the first time is the single most powerful thing you can do to speed up approval. Missing or mismatched documents are the leading cause of CDSCO query letters and multi-month delays.
Documents for Importers — Form MD-14 Application
| S.No. | Document | Notes |
| 1 | Cover Letter | On official letterhead of the authorized Indian agent, addressed to the Central Licensing Authority |
| 2 | Form MD-14 (SUGAM portal) | Fully completed online application — paper copies not accepted |
| 3 | Power of Attorney (PoA) | Notarized/apostilled; must be between foreign manufacturer and Indian agent specifically |
| 4 | Undertaking from Authorized Agent | Agent declares responsibility for regulatory compliance in India |
| 5 | ISO 13485:2016 Certificate | Valid from accredited certification body; must cover the actual manufacturing site, not just HQ |
| 6 | Plant Master File (PMF) | Manufacturing site details, infrastructure, QMS overview, process descriptions |
| 7 | Device Master File (DMF) | Design, materials, intended use, instructions for use, testing data — Indian DMF format |
| 8 | Free Sale Certificate (FSC) | Issued by regulatory authority in country of origin; must list exact device brand name and model |
| 9 | CE / FDA / TGA / Health Canada Approval | GHTF country approval significantly accelerates technical review — attach if available |
| 10 | Product Catalogue / Brochure | Must include model number, technical specifications, images, and performance data |
| 11 | Instructions for Use (IFU) | In English; must describe intended use, contraindications, warnings, and user guidance |
| 12 | Biocompatibility Reports | Required for materials that come in contact with the patient (per ISO 10993 series) |
| 13 | Electrical Safety Test Report | Compliance with IEC 60601-1 (General) and IEC 60601-1-2 (EMC) |
| 14 | Performance and Safety Test Report | Alarm testing, tidal volume accuracy, pressure limits, FiO2 accuracy |
| 15 | Software Validation Documents | Required for ventilators with embedded software or AI-assisted features (IEC 62304) |
| 16 | Labeling Samples | Must comply with Schedule 7 of MDR 2017 — include all required fields |
| 17 | Fee Payment Receipt (Challan) | Proof of government fee payment via Bank of Baroda or ECS |
| 18 | Market Standing / Non-Conviction Certificate | Now auto-generated digitally via SUGAM portal as per 2025 update — no manual affidavit needed |
Documents for Indian Manufacturers — Form MD-9 Application
- Form MD-9 application submitted on SUGAM portal
- ISO 13485:2016 certification from an accredited body covering all relevant manufacturing processes
- Plant Master File — complete facility layout, equipment list, utilities, manufacturing area details
- Device Master File — design history, materials, risk analysis, intended use, IFU
- Risk Management File as per ISO 14971:2019
- Essential Principles of Safety and Performance Checklist (Schedule 3, MDR 2017)
- Electrical safety test reports per IEC 60601-1, IEC 60601-1-2 (EMC), IEC 60601-1-6 (usability)
- Performance test reports — alarm functions, tidal volume accuracy, pressure controls, FiO2 accuracy
- Software validation documentation as per IEC 62304 (if applicable)
- Clinical evaluation data or clinical investigation report
- Labeling in English with all Schedule 7 MDR 2017 fields completed
- Site map, manufacturing area layout, and SOPs for critical manufacturing steps
- Government fee payment receipt

BIS Certification for Ventilators in India — A Separate Mandatory Requirement
CDSCO registration alone does not make your ventilator legally sellable in India. You must also obtain BIS (Bureau of Indian Standards) certification. This is a separate certification from a separate authority — but both are equally mandatory for legal sale in the Indian market.


Why Is BIS Certification Required for Ventilators?
Ventilators are classified as electro-medical equipment and are subject to electrical safety and performance standards enforced by BIS. The Government of India has progressively expanded mandatory BIS certification to medical and electrical equipment through Quality Control Orders (QCOs) and the Machinery and Electrical Equipment Safety (Omnibus Technical Regulation) Order, 2024.
Relevant BIS Standards for Ventilators (2026)
| IS Number | Scope | Equivalent International Standard |
| IS 13450 (Part 2/Sec 12): 2023 | Particular requirements for basic safety and essential performance of critical care ventilators — this is the primary BIS standard for ICU ventilators in India as of 2023 | ISO 80601-2-12:2020 + IEC 60601-1 family |
| IS 13450 (IEC 60601-1) | General requirements for basic safety and essential performance of electrical medical equipment | IEC 60601-1:2005 + Amendment 1:2012 |
| IS 13450 Part 1-2 / IEC 60601-1-2 | Electromagnetic compatibility (EMC) requirements for electro-medical equipment | IEC 60601-1-2:2014 (4th edition) |
| IS 13450 Part 1-6 | General requirements — safety in home healthcare environment (applies to home/portable ventilators) | IEC 60601-1-6 (Usability) |
| IS 7661 (Series) | Electro-medical equipment performance and safety requirements for lung ventilators | Referenced in BIS notifications for ventilators |
| IS 13252 / IEC 60601-1-2 | EMC requirements — electromagnetic emissions and immunity | IEC 60601-1-2:2014 |
| ISO 80601-2-12:2020 | Particular requirements — critical care ventilators (directly referenced in IS 13450 Part 2/Sec 12) | ISO 80601-2-12:2020 |
| Important Update (2025–2026): IS 13450 (Part 2/Sec 12): 2023 is the current primary BIS standard for critical care ventilators in India, aligning with ISO 80601-2-12:2020. Manufacturers must test against this standard and obtain certification before commercial sale. Always verify the latest applicable BIS notification before filing, as standards are updated periodically. |
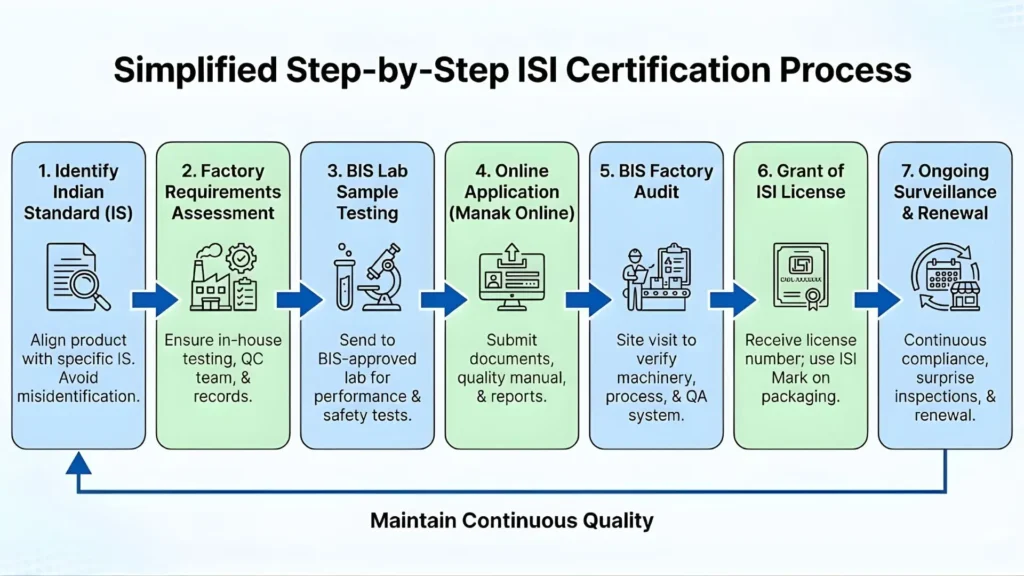
BIS Certification Process for Ventilators
- Identify the applicable IS standard — IS 13450 (Part 2/Sec 12): 2023 for critical care ICU ventilators.
- Submit application on BIS Manak Online Portal — Foreign manufacturers apply under the Foreign Manufacturers Certification Scheme (FMCS); Indian manufacturers apply under Scheme I (ISI Mark) or Scheme X (Certificate of Conformity).
- Product testing at a BIS-recognized laboratory — Testing against the applicable IS standard (IS 13450 Part 2/Sec 12: 2023 and IEC 60601-1 aligned norms). Labs can be in India or abroad if recognized by BIS.
- Factory audit by BIS officer or empanelled auditor — Verifies production processes and quality control systems meet IS standard requirements.
- BIS grants ISI Mark License (Scheme I) or Certificate of Conformity (Scheme X) — upon successful testing and audit clearance.
- Affix BIS Standard Mark on the ventilator or its packaging before sale in India — failure to display the mark constitutes an offense under the BIS Act, 2016.

Government Fees for CDSCO Ventilator Registration (2026)
Fees under MDR 2017 are set in the Sixth Schedule and differ for domestic manufacturers and foreign importers. Note that each manufacturing site requires separate fees.
Fees for Importers — Class C Ventilator
| Fee Head | Amount |
| Manufacturing Site Registration | USD 1,500 per site |
| Single Device Registration | USD 1,000 per device |
| Additional Device under Same Application | USD 1,000 per additional device |
| CDSCO Site Inspection (if triggered by CDSCO) | USD 5,000 per site — additional |
| Retention Fee (every 5 years) — Per Device | USD 1,500 |
| Retention Fee (every 5 years) — Per Site | USD 3,000 |
| Late Fee for Delayed Retention Payment | 2% per month of the retention fee amount (up to 180 days) |
Fees for Indian Manufacturers — Class C Ventilator
| Fee Head | Amount (INR) |
| Manufacturing License Application (Class C) | Rs. 50,000 per site (base fee as per Sixth Schedule — verify current amount on SUGAM) |
| Government Fee per Device (Class C) | Rs. 5,000 per device (approximate) |
| Notified Body Audit Charges | Rs. 50,000 – Rs. 1,50,000 (as per Notified Body's schedule) |
| Retention Fee per Device (every 5 years) | Rs. 1,000 |
| Retention Fee per Site (every 5 years) | Rs. 50,000 |
| Late Fee for Delayed Retention Payment | 2% per month (up to 180 days) |
| Disclaimer: Fee amounts are subject to periodic revision. Always verify the current fee schedule on the official CDSCO SUGAM portal (https://sugam.gov.in) or consult a qualified regulatory professional before making payments. |
CDSCO Ventilator Registration Timeline — How Long Does It Take?
| Stage | Importer (MD-14 / MD-15) | Indian Manufacturer (MD-9 / MD-10) |
| Document Preparation | 2–4 months (if CE/FDA already available) | 3–6 months (DMF, PMF, test reports) |
| Portal Registration and Fee Payment | 1–2 weeks | 1–2 weeks |
| CDSCO Initial Review | 4–8 weeks | 4–8 weeks |
| Factory Audit / Inspection | Usually waived if ISO 13485 + PMF submitted; CDSCO may audit at discretion | 45–60 days after application (Notified Body audit mandatory) |
| Query Resolution (if raised) | 30–90 days depending on complexity | 30–90 days depending on findings |
| License Issuance | 1–3 months after query closure | 1–3 months after audit clearance |
| Total Estimated Time | 6–12 months (typical for complete dossier) | 9–18 months (typical including audit cycle) |
| Pro Tip: The most common cause of delayed CDSCO approval is an incomplete dossier on first submission. Investing 4–6 extra weeks upfront to get documentation right easily saves 3–6 months of query-resolution cycles. |
Validity, Renewal, and Retention of Your CDSCO Ventilator License
Under the Medical Devices (Amendment) Rules, 2020, both the import license (MD-15) and manufacturing license (MD-10) are valid in perpetuity. They do not automatically expire after 5 years. However, the license holder must:
- Pay the retention fee every 5 years from the date of license issuance
- Begin the retention fee process at least 6 months before the 5-year deadline
- Keep all product details, site details, and agent details updated on the SUGAM portal
- Report any changes to the device, manufacturing site, or authorized agent to CDSCO before implementation
Important: If you have added multiple ventilator models to the same base MD-15 license at different points in time, all of them become due for retention together at the same time as the original base license. Plan your retention fee payment calendar accordingly.
Transfer of license: There is currently no provision under MDR 2017 to transfer a CDSCO import license from one authorized Indian agent to another. If you change your agent, you must apply for a completely fresh license — same documents, same fees, same timeline.
Post-Market Compliance — What Happens After You Get the License?
Getting your CDSCO license is not the end — it is the starting point of your ongoing regulatory obligations in India. Here is what you must actively manage after approval:
| Compliance Activity | Frequency | Regulatory Reference |
| Quarterly progress report (for manufacturing sites) | Every quarter | MDR 2017, Rule 76 |
| Periodic Safety Update Report (PSUR) | Annually | MDR 2017, Rule 76 |
| Adverse Event Reporting (Vigilance) | Within 10/15/30 days depending on severity | MDR 2017, Rule 73 |
| Field Safety Corrective Action (FSCA) / Recall | As required — immediately upon identification | MDR 2017, Rule 74 |
| Post-Market Clinical Follow-Up (PMCF) | Ongoing — part of PMS plan | MDR 2017 + CDSCO guidance |
| Labeling Compliance Check | Ongoing and before any label change | MDR 2017, Schedule 7 |
| Retention Fee Payment | Every 5 years from license issuance date | MDR 2017, Sixth Schedule |
| Change Notification (device, site, or agent changes) | Before any implementation | MDR 2017, Rule 59 |
| BIS Surveillance Audit (ISI Mark) | Annually (for Scheme I licensees) | BIS Act, 2016 |
Key 2025–2026 Regulatory Updates for Ventilator Manufacturers and Importers
The Indian medical device regulatory environment has seen significant changes in 2024–2026. Here are the most important updates that directly affect ventilator registrations:
- CDSCO FAQ Addendum-01 (April 2024): Clarified that each manufacturing site requires a separate MD-14 application and separate fees. Also provided clarity on biocompatibility data requirements and post-market surveillance reporting formats.
- CDSCO FAQ Addendum-02 (July 2024): Additional guidance on multilingual labeling for devices sold across Indian states, re-import approval procedures, and Software as a Medical Device (SaMD) classification — directly relevant for AI-assisted ventilators and software-driven ventilation modes.
- CDSCO FAQ Addendum-03 (November 2024 / 2025): Updates on general licensing, post-approval change procedures, and import/export compliance. Introduced auto-generated Market Standing and Non-Conviction Certificates on the SUGAM portal — eliminating the need for manual affidavits in many cases.
- BIS Scheme X Deadline Extension (June 2025): The Ministry of Heavy Industries extended the mandatory compliance deadline for BIS Scheme X certification — covering electrical and electro-medical equipment including ventilators — to 1 September 2026. This gives manufacturers additional time to meet IS 13450 (Part 2/Sec 12): 2023 testing and documentation requirements.
- IS 13450 (Part 2/Sec 12): 2023 Notified: BIS formally adopted IS 13450 (Part 2/Sec 12): 2023 as the specific standard for critical care ventilators, aligning with ISO 80601-2-12:2020. This is now the primary standard that ventilator manufacturers must certify against for BIS compliance.
- Neutral Export Code (2025): CDSCO introduced a system-generated Neutral Code for manufacturers exporting medical devices from India, simplifying export compliance documentation for Indian ventilator manufacturers selling abroad.
- Auto-Generated Compliance Certificates (2025): The SUGAM portal now auto-generates Market Standing Certificates and Non-Conviction Certificates digitally for all licensed entities — significantly reducing documentation burden for renewal and endorsement applications.
Common Mistakes That Delay CDSCO Ventilator Registration — And How to Avoid Them
- Mismatch between model numbers on Form MD-14 and the Free Sale Certificate — leads to an automatic CDSCO deficiency letter and multi-month delays.
- Appointing your commercial distributor as your authorized Indian agent — creates regulatory lock-in. If you change distributors for any commercial reason, you lose the CDSCO license and must start fresh.
- Submitting an incomplete Device Master File — CDSCO expects a complete dossier on first submission. Missing sections trigger mandatory queries. Every query cycle adds 30–90 days.
- Forgetting BIS certification — many companies focus only on CDSCO and neglect BIS, then face distribution disruptions when BIS compliance is demanded by buyers or inspectors.
- Not testing against IS 13450 (Part 2/Sec 12): 2023 — using older test reports against superseded standards is a common cause of BIS application rejection.
- Late retention fee payment — even a few days late triggers penalty fees. After 180 days, the license is automatically cancelled.
- Not notifying CDSCO before implementing design or labeling changes — post-approval changes must be reported and approved before implementation per MDR 2017, Rule 59.
- Assuming State FDA manages Class C ventilators — Class C devices are exclusively under CDSCO (Central Licensing Authority). Going to the State FDA wastes time and will result in rejection.
- Using a single MD-15 for ventilators manufactured at two different plants — each manufacturing site requires its own separate CDSCO license. Confirmed by CDSCO FAQ Addendum-01, 2024.
Labeling Requirements for Ventilators Under MDR 2017 — Schedule 7
Every ventilator sold in India must carry a label in English with the following mandatory information under Schedule 7 of MDR 2017:
- Name and model number of the device
- Name, address, and CDSCO license number of the manufacturer
- Name and address of the authorized Indian agent (for imported ventilators)
- Batch or lot number
- Date of manufacture
- Intended use or intended purpose
- Warnings and precautions — including electrical safety warnings
- Storage conditions (if applicable)
- The CDSCO registration/license number
- BIS Standard Mark (once BIS certification is obtained)
| 2024 Update: CDSCO FAQ Addendum-02 clarified that for ventilators distributed across multiple Indian states, multilingual labeling may be required for certain fields. Verify state-specific requirements with your regulatory consultant. |
Conclusion — Your Roadmap to CDSCO Ventilator Registration in India (2026)
Registering a ventilator with CDSCO in India is a multi-layer process — classification, technical documentation, authorized agent appointment, SUGAM portal filing, government fees, BIS certification under IS 13450 (Part 2/Sec 12): 2023, and ongoing post-market compliance. None of these steps can be skipped or shortcut without legal consequences.
Once you understand the structure — Class C classification, Form MD-14 for import, Form MD-15 as your license, Form MD-9 for Indian manufacturing, BIS as a parallel mandatory track — the path is manageable. The companies that get CDSCO registration right the first time are those that invest in complete documentation upfront, appoint the right regulatory agent, and treat BIS certification as an equally critical parallel process rather than an afterthought.
India's medical device market is projected to reach USD 50 billion by 2030. Ventilators remain among the most tightly regulated products in this market, and that regulatory discipline creates real barriers for non-compliant players. Getting your regulatory house in order now means you are positioned to serve government tenders, private hospitals, and export markets with full legal standing.
Frequently Asked Questions
Is CDSCO registration mandatory for all types of ventilators in India?
Yes. Ventilators are notified medical devices under MDR 2017 and classified as Class C. Both importers and Indian manufacturers require a valid CDSCO license before legally selling or supplying any ventilator in India. There is no exemption — not for trial units, not for donated equipment, and not for samples unless covered by a separate testing or import exemption provision.
What is the difference between Form MD-14 and Form MD-15?
Form MD-14 is the application form submitted on SUGAM to request CDSCO approval to import a specific medical device into India. Form MD-15 is the actual import license that CDSCO issues after reviewing and approving the MD-14 application. You submit MD-14 — CDSCO gives you MD-15.
Do I need a separate CDSCO license for each ventilator model and manufacturing site?
Yes. Each device model and each manufacturing site requires a separate Form MD-14 application with its own government fee. If you have three ventilator models manufactured at two plants, you need six separate applications. CDSCO FAQ Addendum-01 (April 2024) explicitly confirmed this site-specific requirement.
What is the typical approval timeline for CDSCO Class C ventilator registration?
For importers with a well-prepared dossier and existing CE or FDA clearance: 6–12 months. For Indian manufacturers requiring a factory audit: 9–18 months. Incomplete documentation or unresolved queries significantly extend this timeline. There is no official fast-track program for Class C devices as of 2026.
What BIS standard applies to ventilators in India in 2026?
The primary BIS standard for critical care ICU ventilators is IS 13450 (Part 2/Sec 12): 2023, which aligns with ISO 80601-2-12:2020 and the IEC 60601-1 family. General electrical safety requirements are covered under IS 13450 (IEC 60601-1) and EMC compliance under IS 13450 Part 1-2 / IEC 60601-1-2. Both CDSCO registration and BIS certification are separately mandatory for legal sale in India.
Can the CDSCO import license (Form MD-15) be transferred to a new authorized agent?
No. There is currently no provision under MDR 2017 to transfer a CDSCO import license from one authorized Indian agent to another. If you change your authorized agent for any reason — commercial dispute, restructuring, etc. — you must file a completely fresh application, pay full government fees, and restart the entire process from scratch.
What happens if I miss the retention fee payment deadline?
A penalty of 2% of the retention fee amount per month applies for up to 180 days after the due date. Beyond 180 days without payment, the MD-15 license is automatically cancelled by CDSCO. You will need to file a fresh application to resume sales — there is no reinstatement option after automatic cancellation.
Can a foreign manufacturer without CE or FDA approval still get CDSCO registration?
Yes, it is possible. However, CE, FDA, TGA, or Health Canada approval significantly strengthens your technical dossier and is the most reliable way to accelerate CDSCO's technical review. Without such approvals, CDSCO typically requests additional clinical evidence or performance data, which can extend the review timeline by 6–12 additional months.
Does CDSCO conduct a factory inspection for imported ventilators?
Not routinely. For Class C imported ventilators, CDSCO generally accepts a valid ISO 13485 certificate covering the actual manufacturing site along with a complete Plant Master File in lieu of a physical audit. However, CDSCO reserves the right to conduct a site inspection at the manufacturer's cost if it has concerns about the dossier. As of 2025, such inspections are rare for well-documented applications with GHTF country approvals.
What is the SUGAM portal and how does it work for ventilator registration?
SUGAM (https://sugam.gov.in) is CDSCO's official digital portal for all medical device regulatory applications in India. All Form MD-14 and Form MD-9 applications must be submitted here. The authorized Indian agent (for imports) or the company (for manufacturing) creates an account, uploads all technical documents, pays government fees online, responds to CDSCO queries, and tracks application status — all through SUGAM. Paper submissions are no longer accepted.





































