- Medical Device Registration For Anesthesia machines are classified as Class C (high-risk) medical devices under CDSCO — mandatory registration is required before import or sale in India.
- Import registration uses Form MD-14/MD-15; domestic manufacturing uses Form MD-7/MD-9 — both routes require technical dossier, ISO 13485, and FSC.
- The entire CDSCO approval process for an anesthesia machine typically takes 9 to 18 months, with registration fees ranging from INR 50,000 to INR 3,00,000.
- Registration is valid for 5 years and must be renewed 6 months before expiry — non-compliance attracts penalties under the Medical Devices Rules, 2017.
Introduction
If you are planning to import or manufacture anesthesia machines in India, the single most important step you need to get right is CDSCO registration. The Central Drugs Standard Control Organization (CDSCO) is the national regulatory authority under the Ministry of Health & Family Welfare, and no anesthesia machine — whether imported or made in India — can legally enter the market without its approval.


This guide covers everything you need to know about anesthesia machine registration India in 2026: from understanding what an anesthesia machine is and how CDSCO classifies it, to the exact documents, timelines, fees, common rejection reasons, and practical tips that experienced regulatory consultants actually use.
Whether you are a hospital procurement manager, a medical device importer, an OEM manufacturer, or a startup entering the Indian medtech space, this pillar page is your complete reference.
What Is an Anesthesia Machine?
An anesthesia machine — also called an anesthesia workstation or anesthesia delivery system — is a complex medical device used to deliver a controlled mixture of medical gases and inhalational anesthetic agents to a patient during surgical procedures. It maintains the unconscious state of the patient while simultaneously monitoring vital parameters.
Modern anesthesia workstations are integrated systems that typically combine the following components:
- Gas delivery system (oxygen, nitrous oxide, medical air)
- Vaporizers for volatile anesthetic agents (isoflurane, sevoflurane, desflurane)
- Breathing circuit with rebreathing bag and CO2 absorber
- Mechanical ventilator
- Patient monitoring modules (SpO2, capnography, ECG, NIBP)
- Alarm systems and data recording features
- Scavenging system for waste anesthetic gas disposal
Anesthesia machines are used in operating rooms, ICUs, procedural suites, and increasingly in remote or ambulatory surgical settings. Given their direct role in maintaining patient life during surgery, they are subject to the strictest regulatory scrutiny.
Applicable Standards:
| Standard | Applicability |
| ISO 80601-2-13:2022 | Particular requirements for anaesthetic workstations |
| IEC 60601-1 | General safety and essential performance for medical electrical equipment |
| IEC 60601-1-2 | Electromagnetic compatibility (EMC) |
| IEC 60601-1-6 | Usability engineering |
| IS/IEC 60601-1-8 | Alarm systems |
| ISO 13485:2016 | Quality management system for medical device manufacturers |
Risk Classification Under CDSCO: Why Anesthesia Machines Are Class C
Under the Medical Devices Rules (MDR), 2017 — as amended up to 2023 — medical devices are classified into four risk classes:
| Class | Risk Level | Examples |
| Class A | Low Risk | Tongue depressors, bandages, examination gloves |
| Class B | Low-Medium Risk | Hypodermic syringes, thermometers, blood pressure cuffs |
| Class C | Medium-High Risk | Anesthesia machines, ventilators, infusion pumps, dialysis machines |
| Class D | High Risk | Implantable pacemakers, heart valves, HIV test kits |
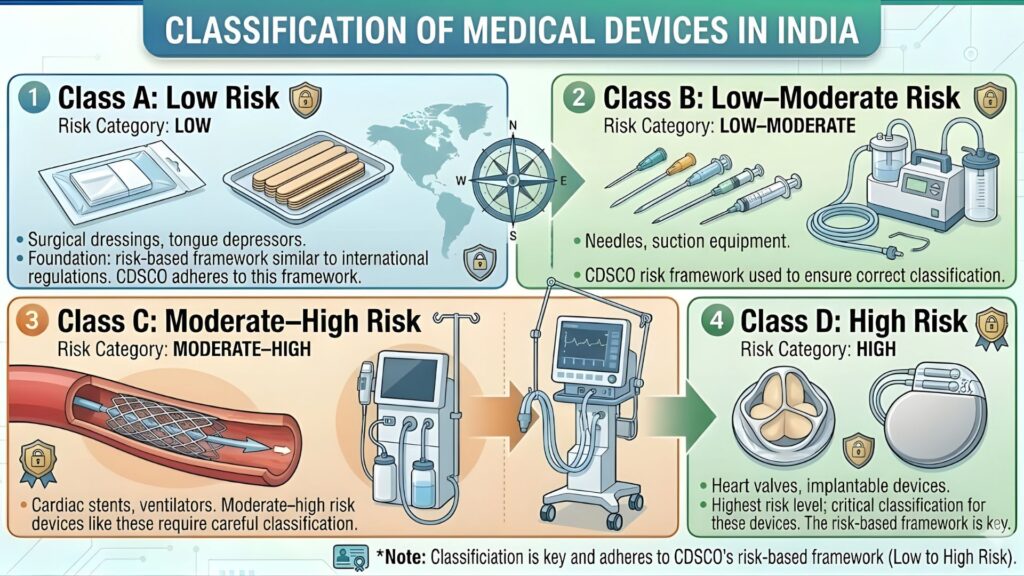

Anesthesia machines fall squarely under Class C because:
- They are directly connected to the patient's airway and life-sustaining functions
- A malfunction can cause immediate, irreversible patient harm or death
- They involve complex interactions with multiple drugs and gases
- They require specialized operator training to function safely
This Class C classification means CDSCO mandates a full technical dossier review and often requires performance testing data, clinical evidence, and post-market surveillance commitments. There is no shortcut — regulators will check every document.
Registration Pathways: Import vs. Manufacturing
There are two primary registration pathways for anesthesia machine registration India:
Import Registration (Form MD-14 / MD-15)
If you are an Indian company or authorized agent importing anesthesia machines from a foreign manufacturer, you need to apply for an import license under the CDSCO framework. The process uses two key forms:
| Form | Purpose | Who Files |
| MD-14 | Application for import license for medical devices (new device) | Indian importer / authorized agent |
| MD-15 | Grant of import license for medical devices | Issued by CDSCO on approval |
Key points about the import route:
- The Indian entity (importer) must appoint a single authorized representative for each foreign manufacturer
- The foreign manufacturer must have a valid manufacturing license or equivalent in their home country
- A Free Sale Certificate (FSC) from the country of origin is mandatory for Class C devices
- All devices must comply with IS/IEC 60601-1 or equivalent and ISO 80601-2-13
- Registration is product-specific and model-specific — each unique model/SKU needs separate registration
Domestic Manufacturing Registration (Form MD-7 / MD-9)
If you are manufacturing anesthesia machines within India, the licensing path is through state licensing authority for the manufacturing license and CDSCO central for the market authorization:
| Form | Purpose | Issuing Authority |
| MD-3 | Application for manufacturing license (state-level) | State Licensing Authority (SLA) |
| MD-4 | Grant of manufacturing license | State Licensing Authority (SLA) |
| MD-7 | Application for market authorization (Class C/D devices) | CDSCO Central |
| MD-9 | Grant of market authorization | CDSCO Central |
The domestic manufacturer must first obtain a valid manufacturing license from the State Licensing Authority (SLA) of the state where the manufacturing facility is located. Only after this can the manufacturer apply to CDSCO for market authorization (Form MD-7).
Key requirements for domestic manufacturers:
- ISO 13485:2016 certification from an accredited certification body
- Approved manufacturing premises with adequate infrastructure per Schedule M of MDR 2017
- Qualified Person (QP) appointed as per regulatory requirements
- Complete Design History File (DHF) and Device Master Record (DMR)
- Performance testing conducted at NABL-accredited lab or notified body
Benefits of CDSCO Registration for Anesthesia Machines
Getting CDSCO registration for your anesthesia machine is not just about legal compliance. It opens up real commercial and strategic benefits:
| Benefit | Why It Matters |
| Legal market access | Only registered devices can be legally sold, imported, or distributed in India |
| Eligibility for government tenders | Central and state government hospital tenders (AIIMS, CGHS, state health departments) require CDSCO registration |
| Hospital procurement compliance | NABH-accredited hospitals are required to procure only CDSCO-registered devices |
| Investor & partner confidence | CDSCO registration signals regulatory maturity — essential for fundraising and partnerships |
| Insurance & liability protection | Selling unregistered devices voids manufacturer liability claims and insurance |
| Brand credibility | Registration demonstrates product safety and quality to healthcare professionals |
Step-by-Step Registration Process Medical Device Registration for Anesthesia Machines
For Import (MD-14 Application)
- Create SUGAM Account: Both the foreign manufacturer and Indian importer must register on the SUGAM portal (sugamapplication.cdsco.gov.in).
- Prepare Technical Dossier (Device Master File / DMF): Compile all technical, clinical, and quality documents (detailed in Section 5 below).
- Online Application Submission: File Form MD-14 on SUGAM portal with all documents uploaded in specified format.
- Fee Payment: Pay the prescribed fee online (see Section 8 for fee details).
- Scrutiny by CDSCO: Application is reviewed for completeness. Deficiencies are communicated via SUGAM portal — typically within 30 days.
- Technical Evaluation: CDSCO's Medical Device Technical Advisory Committee (MDTAC) evaluates the technical dossier. May call for additional data or lab testing.
- Site Inspection (if applicable): CDSCO may conduct a facility audit of the foreign manufacturer or the Indian importer's premises.
- Grant of Import License (MD-15): If the application is approved, CDSCO issues the import license (Form MD-15).
For Domestic Manufacturing (MD-7 Application)
- Obtain manufacturing license from State Licensing Authority (Form MD-3/MD-4).
- Prepare full technical dossier including Design Dossier, Risk Management File, and Clinical Evaluation Report.
- File Form MD-7 on SUGAM portal with all technical and quality documents.
- CDSCO reviews application. Queries may be raised and must be responded to within 30 days.
- Manufacturing facility inspection may be conducted by CDSCO or MDTAC-designated experts.
- Market authorization (Form MD-9) granted upon satisfactory completion of all requirements.
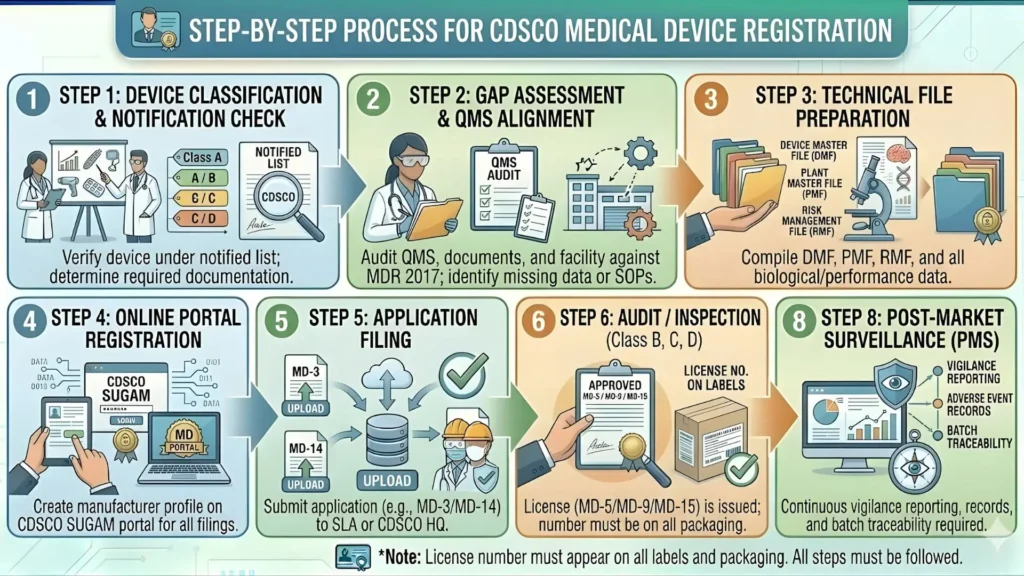

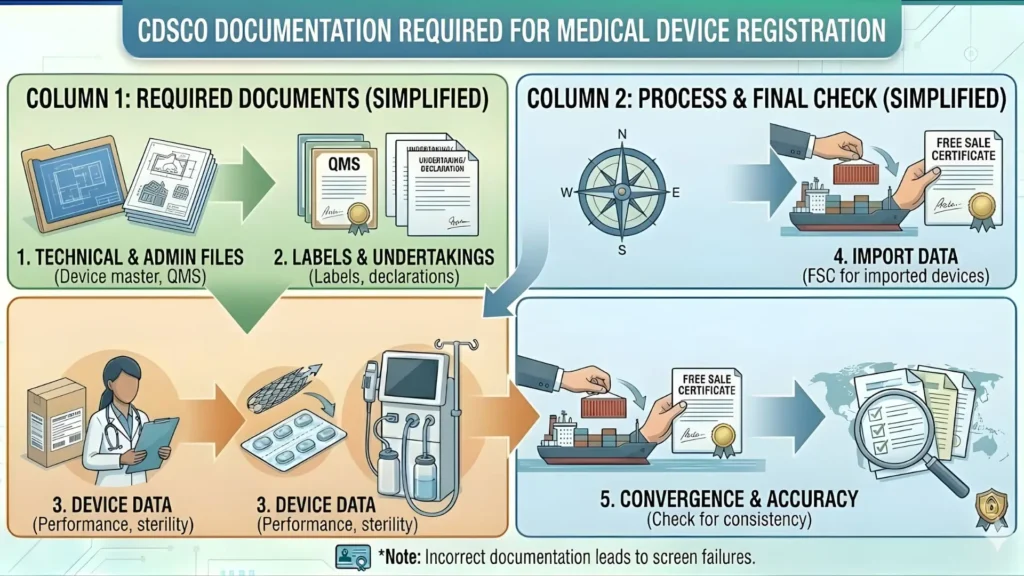
Documents Required for CDSCO Anesthesia Machine Registration
This is the most critical section. Many applications fail at the scrutiny stage because of incomplete or poorly structured documentation. Here is the complete checklist:
Device Master File (DMF) / Technical Dossier
| Document | Details Required |
| Device Description | Intended use, indications, model numbers, accessories, variants |
| Device Classification Justification | Why the device is Class C with regulatory basis |
| Design & Engineering Drawings | Exploded views, assembly drawings, dimensions, materials |
| Bill of Materials (BOM) | Complete component list with specifications |
| Functional Block Diagram | Electrical, pneumatic, and mechanical block diagrams |
| Labeling & IFU | Draft label, Instructions for Use in English (Hindi optional) |
| Shelf Life Data | If applicable — packaging validation, accelerated aging data |
| Sterilization Validation | Not typically required for anesthesia machines (non-sterile device) |
| Packaging Validation Report | ASTM D4169 or equivalent drop/vibration test reports |
Performance & Safety Testing Documents
| Test Document | Standard / Reference |
| Electrical Safety Test Report | IEC 60601-1 (tested at NABL-accredited lab) |
| EMC Test Report | IEC 60601-1-2 |
| Performance Test Report | ISO 80601-2-13 / relevant national standard |
| Alarm System Test Report | IEC 60601-1-8 |
| Usability Engineering File | IEC 60601-1-6 / IEC 62366-1 |
| Software Documentation (if applicable) | IEC 62304 — software lifecycle for medical device software |
| Biocompatibility Test Report | ISO 10993 for patient-contact parts |
Quality System Documents
- ISO 13485:2016 Certificate (valid, from accredited certification body)
- Quality Manual (brief overview of QMS structure)
- Declaration of Conformity (manufacturer's declaration)
- CE Mark Certificate or US FDA 510(k) / PMA (if available — strengthens application)
Import-Specific Documents
- Free Sale Certificate (FSC) — from regulatory authority of country of origin, apostilled or notarized
- Certificate of Pharmaceutical Product (CPP) equivalent — for devices where applicable
- Manufacturer's Authorization Letter — authorizing the Indian importer as exclusive agent
- Power of Attorney — for Indian importer/authorized agent
- Foreign manufacturer's valid manufacturing license / registration certificate
- Undertaking for post-market surveillance and adverse event reporting
- Undertaking to comply with MDR 2017 and all CDSCO directives
Clinical Evaluation Report (CER)
For Class C devices like anesthesia machines, CDSCO requires a Clinical Evaluation Report (CER) that demonstrates clinical safety and performance. The CER must include:
- Summary of clinical data from literature (published peer-reviewed studies)
- Equivalent device comparison (if claiming equivalence to a predicate device)
- Post-market clinical follow-up (PMCF) plan
- Risk-benefit analysis based on clinical evidence
Note: CDSCO does not always require clinical trials for Class C devices that have CE or FDA approval. However, the absence of such international approvals may trigger additional scrutiny or a request for Indian clinical trial data.
Risk Management File
A complete Risk Management File as per ISO 14971:2019 must be submitted. This includes:
- Risk Management Plan
- Hazard identification and risk estimation
- Risk control measures and residual risk evaluation
- Overall residual risk acceptability assessment

Labeling & UDI Requirements for Anesthesia Machines in India
Labeling compliance is non-negotiable. CDSCO has specific labeling requirements under Rule 24 of MDR 2017, and anesthesia machines must carry all mandatory information.
Mandatory Label Information
- Name and model number of the device
- Name and address of manufacturer (and Indian importer/authorized agent)
- Manufacturing date and batch/lot number
- CDSCO registration/license number once approved
- Intended use or indication (brief)
- Storage conditions and special handling instructions
- Caution/warning statements as per IEC 60601-1-8
- Country of origin
- IFU reference or QR code linking to electronic IFU
Unique Device Identification (UDI)
India's UDI framework for medical devices is being phased in. As of 2026, Class C medical devices are required to carry a UDI as per the CDSCO UDI guidelines published in 2023. The UDI consists of:
- Device Identifier (DI): Identifies the specific device model and version
- Production Identifier (PI): Includes lot/batch number, serial number, manufacturing date, expiry date
Manufacturers and importers must register their UDI on the SUGAM UDI portal and on the Global UDI Database (GUDID) if the device is also marketed internationally.
Registration Timeline: How Long Does CDSCO Approval Take?
One of the most common questions from importers and manufacturers is: how long does CDSCO anesthesia machine approval actually take? Here is a realistic breakdown based on current 2026 processing timelines:
| Stage | Estimated Time | Notes |
| Document preparation & compilation | 2 to 4 months | Depends on readiness of test reports, CER, FSC |
| SUGAM portal application & fee payment | 1 to 3 days | Portal is online — quick once documents are ready |
| Scrutiny by CDSCO | 30 to 60 days | CDSCO raises deficiency if docs are incomplete |
| Response to deficiency queries | 30 to 60 days | Your team must respond promptly within timeline |
| Technical evaluation by MDTAC | 3 to 6 months | Longest stage — depends on CDSCO workload |
| Facility inspection (if called) | 30 to 90 days | Not always required; adds significant time if called |
| Approval & license issuance (MD-15/MD-9) | 15 to 30 days | After technical clearance |
| TOTAL (typical range) | 9 to 18 months | Well-prepared files can achieve 9 months |
Pro Tip: Applications with CE Mark (EU) or US FDA clearance/approval are typically processed faster because CDSCO treats these as strong evidence of safety and performance. Always include your CE/FDA documentation if available.
Registration Fees for Anesthesia Machines
CDSCO registration fees are prescribed under the Second Schedule of Medical Devices Rules, 2017 (as amended). Here are the applicable fees for Class C medical devices:
| Fee Type | Entity Type | Fee (INR) |
| Import License Application (MD-14) | Large company (turnover > INR 250 crore) | INR 3,00,000 per device |
| Import License Application (MD-14) | Medium company (turnover INR 10-250 crore) | INR 1,50,000 per device |
| Import License Application (MD-14) | Small/micro company (turnover < INR 10 crore) | INR 50,000 per device |
| Manufacturing Market Authorization (MD-7) | Large company | INR 2,00,000 per device |
| Manufacturing Market Authorization (MD-7) | Medium company | INR 1,00,000 per device |
| Manufacturing Market Authorization (MD-7) | Small/micro company | INR 40,000 per device |
| Renewal Fee | All categories (proportionate) | 50% of original application fee |
| Amendment Fee | All categories | Varies — 25% to 50% of original fee |
Important: The above fees are for the CDSCO application only. You will also need to budget for:
- ISO 13485 certification cost: INR 1,50,000 to INR 5,00,000 (depending on certifying body)
- Lab testing (IEC 60601-1, EMC, performance): INR 3,00,000 to INR 8,00,000
- Regulatory consultant fees: INR 2,00,000 to INR 10,00,000
- Translation and notarization of foreign documents: INR 50,000 to INR 2,00,000
Validity & Renewal of CDSCO Registration
Understanding the validity period and renewal requirements is essential for maintaining compliance and uninterrupted market access.
| Parameter | Details |
| Initial Registration Validity | 5 years from date of grant (import license MD-15 and manufacturing market authorization MD-9) |
| Renewal Application Window | Apply for renewal 6 months before expiry (i.e., in the 5th year, not after expiry) |
| Renewal Form | Form MD-17 (for import license renewal) / Form MD-11 (manufacturing) |
| Renewal Timeline | Typically 3 to 6 months if all post-market compliance is in order |
| Post-Market Surveillance (PMS) | Annual PMS reports must be submitted; non-submission can cause renewal delays |
| Adverse Event Reporting | Mandatory under MDR 2017 — serious adverse events must be reported within 30 days |
| Changes During License Period | Any change to device, manufacturer, or label requires prior amendment approval from CDSCO |
What happens if the license expires? Selling or importing an anesthesia machine with an expired CDSCO license is a violation of MDR 2017 and the Drugs and Cosmetics Act. Penalties can include cancellation of license, seizure of products, and prosecution. Never let your license expire without renewing.
Common Rejection Reasons & How to Avoid Them
Based on real-world regulatory experience, these are the most frequent reasons CDSCO rejects or delays anesthesia machine registration applications:
| Rejection Reason | How to Avoid It |
| Incomplete DMF / missing test reports | Use a comprehensive checklist; get all IEC 60601-1 and ISO 80601-2-13 tests done before filing |
| FSC not from competent regulatory authority | Ensure FSC is issued by the FDA, EMA, TGA, PMDA, or equivalent — not just a trade body |
| FSC not apostilled or notarized | Always get FSC apostilled if from a Hague Convention country; notarized otherwise |
| Manufacturer authorization letter expired or vague | Use specific, dated, device-specific authorization letters — not generic letters |
| Outdated ISO 13485 certificate | Check certificate validity — must be valid at time of submission and throughout review |
| Clinical Evaluation Report (CER) insufficient | Engage a qualified clinical evaluator; use MEDDEV 2.7/1 Rev. 4 as reference methodology |
| Labeling non-compliant with Rule 24 MDR 2017 | Review Indian labeling requirements specifically — not just CE/FDA labels |
| Wrong device classification claimed | Confirm Class C with CDSCO guidance documents before filing |
| Delayed response to deficiency queries | Set up a monitoring system for SUGAM portal notifications; respond within 30 days |
| Risk Management File not per ISO 14971:2019 | Use the 2019 version (not 2007) — CDSCO has updated its requirements |
Eligibility: Who Can Apply for CDSCO Registration?
Not everyone can directly apply for CDSCO registration. Here is who is eligible for each registration pathway:
For Import Registration (MD-14)
- An Indian company registered under the Companies Act, 2013 or a partnership/LLP firm registered in India
- Must be appointed as the Authorized Agent (AA) by the foreign manufacturer
- The AA is solely responsible for all regulatory compliance, adverse event reporting, and post-market surveillance in India
- The AA cannot sub-license or transfer the authorization without CDSCO approval
For Manufacturing Registration (MD-7)
- Indian manufacturers with a valid manufacturing license (Form MD-4) from the State Licensing Authority
- The manufacturing facility must be specifically licensed for the class of medical device being manufactured
- Own-brand importers who wish to manufacture in India must obtain separate manufacturing licenses
Conclusion: Getting CDSCO Anesthesia Machine Registration
Anesthesia machine registration India is a complex, document-intensive process that requires careful planning, technical expertise, and regulatory knowledge. But it is absolutely achievable — thousands of devices get registered every year.
The key to a smooth CDSCO anesthesia machine approval process is preparation. Start collecting your documents — especially the FSC, ISO 13485 certificate, and test reports — well before you plan to file. Engage a qualified regulatory consultant if you are unfamiliar with CDSCO's requirements. And always monitor your SUGAM portal for queries and deficiency notices.
In 2026, the Indian medtech market is growing rapidly, and the regulatory environment is becoming more streamlined with SUGAM upgrades and clearer guidance documents. Companies that invest in proper registration today will have a significant competitive advantage in one of the world's fastest-growing healthcare markets.
If you need help with CDSCO registration for anesthesia machines or other Class C medical devices, consult a registered regulatory affairs professional with proven experience in MDR 2017 applications.
Frequently Asked Questions
Is CDSCO registration mandatory for all anesthesia machines sold in India?
Yes. Under the Medical Devices Rules, 2017, all medical devices listed in the notified schedules — including anesthesia machines — must be registered with CDSCO before they can be manufactured for sale, imported, distributed, or sold in India. There are no exemptions for Class C devices.
Can a foreign manufacturer apply directly to CDSCO without an Indian agent?
No. A foreign manufacturer must appoint an Indian Authorized Agent (AA) who submits the application on their behalf. The AA is legally responsible for all compliance obligations in India, including adverse event reporting, PMS submissions, and recall management.
Is a CE Mark or US FDA clearance sufficient for selling anesthesia machines in India?
No. CE Mark or US FDA approval is not accepted as a substitute for CDSCO registration. However, having CE or FDA approval significantly strengthens your application and can reduce CDSCO review time. You must still apply through the standard MD-14 process.
What is the Free Sale Certificate (FSC) and how do I get one?
An FSC is a certificate issued by the competent regulatory authority of the country of origin confirming that the device is legally marketed in that country.
How long is the CDSCO registration valid for anesthesia machines?
CDSCO registration (both import license MD-15 and manufacturing market authorization MD-9) is valid for 5 years from the date of grant. Renewal must be applied for using Form MD-17 (import) or Form MD-11 (manufacturing) at least 6 months before expiry.
Can one registration cover multiple models of anesthesia machines?
No. CDSCO registration is model-specific. Each distinct model or SKU with different technical specifications, intended use, or design requires a separate registration application. However, you can apply for multiple models under the same MD-14 application if they are minor variants of the same product family, with prior CDSCO guidance.
What is the CDSCO registration fee for an anesthesia machine in 2026?
For an import license (MD-14) for a Class C device, the fee ranges from INR 50,000 (small companies) to INR 3,00,000 (large companies) per device.
What happens if I sell an anesthesia machine in India without CDSCO registration?
Selling or importing an unregistered medical device in India is a criminal offence under the Drugs and Cosmetics Act, 1940 and the Medical Devices Rules, 2017. Penalties include imprisonment of up to 3 years, fines, seizure of goods, and cancellation of import/manufacturing licenses. The buyer (hospital/clinic) may also face liability.
Does CDSCO require clinical trials for anesthesia machines manufactured in India?
Not necessarily. CDSCO does not automatically require clinical trials for anesthesia machines that have adequate clinical evidence from published literature and post-market data from international markets
Can an anesthesia machine registration be transferred to a new Indian importer?
Yes, but it requires prior CDSCO approval. If you change your Indian authorized agent, the foreign manufacturer must submit a Change Notification (Form MD-16) to CDSCO, along with the new authorization letter and a no-objection from the outgoing agent (or a legal basis for the change). The transfer is not automatic and takes time.






































