- CDSCO Registration For Pulse oximeters are Class B medical devices under MDR 2017 — mandatory CDSCO registration applies to both importers and manufacturers.
- Without a valid CDSCO import license or manufacturing license, selling or distributing oximeters in India is illegal and can attract heavy penalties.
- The entire registration process — from application to approval — typically takes 45 to 90 working days when documents are in order.
- This guide covers every step: device classification, documents required, government fees, UDI compliance, online selling rules, and common mistakes to avoid.
What Is a CDSCO Registration for Pulse Oximeter?
A pulse oximeter is a non-invasive medical device that measures the oxygen saturation (SpO2) level in a person's blood, along with the pulse rate. It works by emitting light through the skin and detecting how much light is absorbed by oxygenated versus deoxygenated blood.


Sounds simple, right? But here is the thing — these devices are used to make clinical decisions. A low SpO2 reading can indicate conditions like pneumonia, heart disease, or COVID-19 complications. If a device gives a wrong reading, it can cost a life. That is why CDSCO regulates pulse oximeters strictly under the Medical Devices Rules, 2017.
There are two primary types of pulse oximeters sold in India:
• Fingertip/handheld oximeters – Portable consumer-grade devices commonly sold online and in pharmacies.
• Tabletop/patient monitoring oximeters – Hospital-grade devices used in ICUs, operation theatres, and critical care settings.
Both types fall under CDSCO's regulatory purview and require registration before they can be legally sold in India.
Device Classification of Pulse Oximeters Under MDR 2017
Under the Medical Devices Rules, 2017, medical devices in India are classified into four risk categories:
| Class | Risk Level | Examples |
| Class A | Low Risk | Tongue depressors, bandages, surgical gloves |
| Class B | Low to Moderate Risk | Pulse oximeters, stethoscopes, blood pressure monitors |
| Class C | Moderate to High Risk | Dialysis machines, ventilators, bone screws |
| Class D | High Risk | Pacemakers, heart valves, HIV test kits |
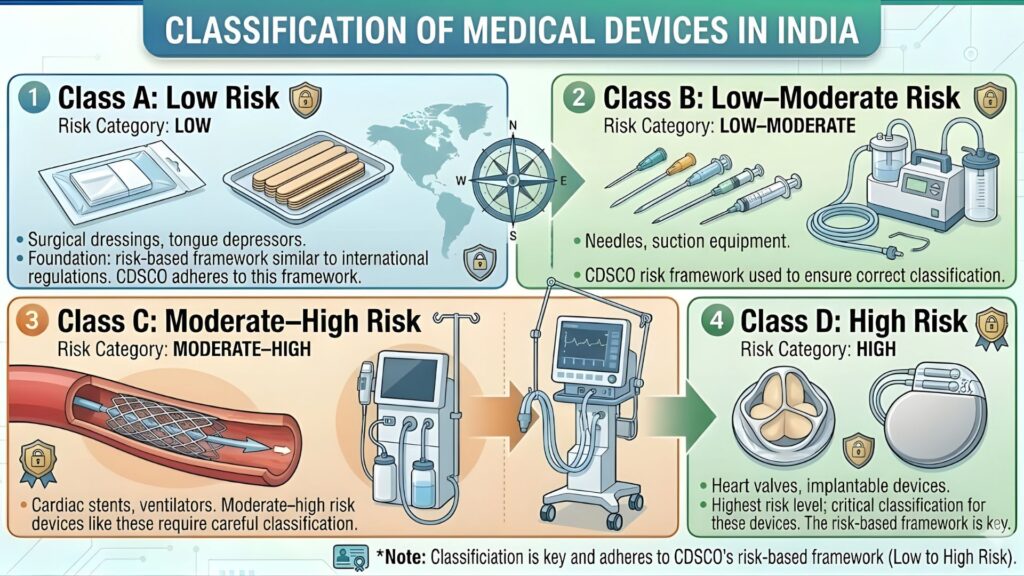

Pulse oximeters are classified as Class B medical devices. This is important because the registration requirements, fees, and timelines differ based on class. Class B devices have a streamlined process compared to Class C and D, but the requirements are still substantive and must be followed carefully.
| Important Note for 2026:As of the latest CDSCO notification, fingertip pulse oximeters used for clinical purposes continue to be regulated as Class B devices. Consumer wellness-only devices may have different compliance pathways, but any device making SpO2 claims is treated as a medical device. |
Who Needs CDSCO Registration for Oximeters?
A very common question we hear is: "Does everyone selling oximeters need a CDSCO license?" The short answer is yes — but let us break it down.
- Importers: Any company or individual importing pulse oximeters from abroad (China, USA, Europe, etc.) into India must hold a valid Import License (Form MD-14) issued by CDSCO. The importer must be registered as an Indian Authorized Agent or a company with a presence in India.
- Manufacturers: Any company manufacturing pulse oximeters in India must hold a Manufacturing License (Form MD-5/MD-9) issued by the State Licensing Authority (SLA) in coordination with CDSCO.
- Repacked/relabelled products: If you are repacking or relabelling an imported oximeter under your own brand, you are treated as a manufacturer and need the appropriate license.
- Online sellers: E-commerce platforms and direct-to-consumer brands selling oximeters online must ensure the underlying product is CDSCO-registered. Selling without registration can result in platform removal and legal penalties.
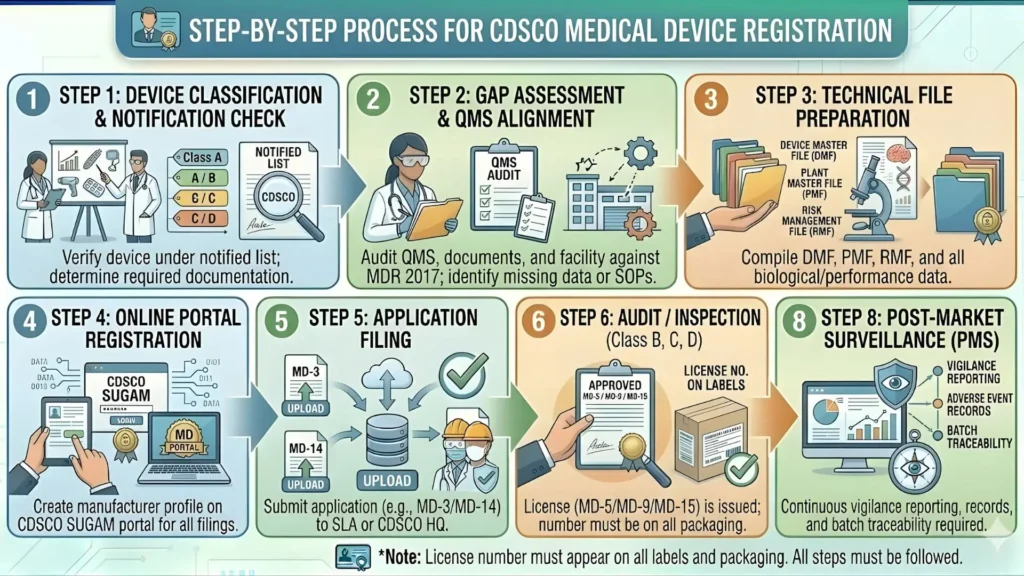
How to Get CDSCO Import License for Pulse Oximeters – Step by Step
Getting an import registration for pulse oximeters involves a clearly defined process under MDR 2017. Here is exactly how it works in 2026:
Step 1: Appoint an Indian Authorized Agent (IAA)
If the manufacturer is based outside India, they must appoint an Indian Authorized Agent (IAA) who will represent them before CDSCO. The IAA must be a registered Indian entity and must sign a formal agreement with the foreign manufacturer, taking responsibility for post-market activities.
Step 2: Register on the SUGAM Portal
All CDSCO applications are processed through the online SUGAM portal (sugamapplication.cdscoonline.gov.in). The importer or IAA must create an account, complete the company profile, and upload all required documents.
Step 3: Submit Application in Form MD-14
The import registration application for Class B devices is filed in Form MD-14. This form captures device details, manufacturer information, intended use, and technical specifications.
Step 4: Technical Evaluation by CDSCO
Once the application is submitted, CDSCO's technical division reviews the documents. For Class B devices like oximeters, CDSCO evaluates safety and performance data, test reports, and conformity certifications. They may raise queries (deficiency letters) if documents are incomplete.
Step 5: Approval and Issuance of Import License
If the application is approved, CDSCO issues an Import License in Form MD-15. This license is device-specific and tied to the foreign manufacturer's name. A fresh license is needed for each new model or manufacturer.
| Pro Tip:Never submit incomplete applications. One missing document can trigger a deficiency notice that delays your approval by 30–60 days. Use a regulatory consultant familiar with CDSCO's current checklist to avoid this. |

CDSCO Manufacturing License for Pulse Oximeters in India
If you are manufacturing pulse oximeters in India, the registration pathway is slightly different. Manufacturing licenses for medical devices are issued by the State Licensing Authority (SLA) of the state where your factory is located, based on the recommendation of CDSCO.
Registration Process for Manufacturers
- Step 1 – Apply to the State Licensing Authority (SLA) in Form MD-3 (for Class A and B devices).
- Step 2 – SLA inspects your manufacturing premises to verify GMP (Good Manufacturing Practice) compliance as per Schedule M.
- Step 3 – SLA forwards the application to CDSCO for technical review of Class B device dossier.
- Step 4 – CDSCO evaluates the technical documentation and issues a recommendation.
- Step 5 – SLA issues the Manufacturing License in Form MD-5, allowing you to manufacture and sell in India.
| Requirement | Details |
| Premises | Dedicated manufacturing space meeting Schedule M GMP requirements |
| Quality System | ISO 13485 certification strongly recommended; internal QMS mandatory |
| Technical Staff | Qualified technical person (QTP) must be appointed |
| Testing | In-house testing or third-party NABL-accredited lab for device testing |
| Device Master File | Complete technical file with design history, risk analysis, test reports |
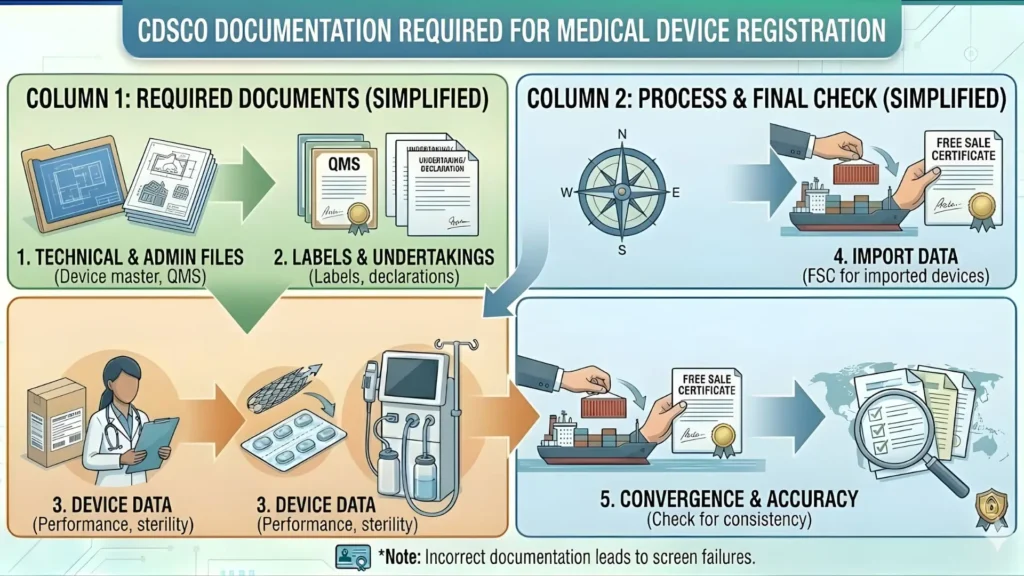
Documents Required for Pulse Oximeter CDSCO Registration
This is where most applicants get tripped up. The document checklist for Class B oximeter registration is specific, and any gap can delay your approval significantly. Here is a complete list:
For Import Registration (Form MD-14)
• Certificate of Free Sale (CFS) or CE Certificate from the country of origin
• ISO 13485 certificate of the foreign manufacturer
• IEC 60601-1 test report (electrical safety) from accredited lab
• ISO 9919 test report (performance standard specific to pulse oximeters)
• Device description and intended use statement
• Labeling and Instructions for Use (IFU) in English
• Manufacturing site details and plant master file (PMF)
• Authorization letter from foreign manufacturer to Indian agent
• Undertaking by the Indian Authorized Agent
• Form MD-14 application (filled and signed on SUGAM portal)
• Proof of payment of government fee
• Declaration of conformity from the manufacturer
For Manufacturing Registration (Form MD-3)
• Site Master File (SMF) of the manufacturing premises
• ISO 13485 Quality Management System certificate
• Design and Development records (DHF – Device History File)
• Risk management file as per ISO 14971
• Biocompatibility data (if applicable) as per ISO 10993
• Software documentation (if software is a component) as per IEC 62304
• Clinical evaluation report or clinical investigation data
• Performance test reports from NABL-accredited lab
• Labeling artwork with all required CDSCO/MDR 2017 labeling elements
• Qualified Technical Person (QTP) appointment proof
• Proof of site ownership or lease agreement

| Document Tip: For Chinese-origin imports (which make up a large volume of oximeters in India), CDSCO often specifically scrutinizes the ISO 9919 test report and the CE certificate issuing notified body. Ensure your reports come from accredited, recognized bodies — not self-declarations. |
Government Fees and Registration Timeline for Oximeter CDSCO Registration
One of the most frequently asked questions is: 'How much does CDSCO registration for a pulse oximeter cost?' Here is a clear breakdown as applicable in 2026:
Government Fee Structure
| Application Type | Device Class | Fee (INR) | Fee (USD Approx.) |
| Import License (MD-14) | Class B | Rs. 1,00,000 per device | ~USD 1,200 |
| Manufacturing License (MD-3) | Class B | Rs. 50,000 per device | ~USD 600 |
| Amendment/Variation | Class B | Rs. 25,000 | ~USD 300 |
| License Renewal | Class B | Rs. 50,000 | ~USD 600 |
Note: The above fees are government/statutory fees only. Professional fees for regulatory consultants, document preparation, lab testing, and translations are additional costs that vary widely.
Approval Timeline
| Stage | Approximate Time |
| Document preparation and IAA appointment | 2–4 weeks |
| SUGAM portal submission | 1–3 days |
| CDSCO technical review (Class B) | 30–45 working days |
| Deficiency resolution (if raised) | Additional 15–30 days |
| Import License issuance | Total: 45–90 working days from submission |
| Manufacturing License (including SLA inspection) | 60–120 working days |
Labeling Requirements for Pulse Oximeters in India (MDR 2017 Compliance)
Labeling compliance is non-negotiable. Non-compliant labels are one of the top reasons for market seizures and penalties. MDR 2017 Schedule IV specifies mandatory labeling elements for medical devices sold in India.
Mandatory Label Information for Oximeters
• Name and address of the manufacturer
• Name and address of the Indian Authorized Agent or importer
• Brand name and device name (e.g., 'Pulse Oximeter')
• Model number / catalogue number
• Lot number or batch number
• Date of manufacture and expiry (if applicable)
• Intended purpose / indication for use
• Storage conditions and precautions
• CDSCO Import License number or Manufacturing License number
• Unique Device Identification (UDI) number (mandatory from June 2026 for Class B)
• CE marking information (if applicable, for imported devices)
• Warning statements and contraindications
• Language: English (with Hindi for domestic sales — strongly recommended)
| 2026 UDI Update:CDSCO has been progressively rolling out the Unique Device Identification (UDI) system in India. Class B devices, including pulse oximeters, are expected to comply with UDI requirements for labeling and registration on the SUGAM UDI database. Ensure your UDI strategy is in place before applying for or renewing your license. |
UDI Compliance for Pulse Oximeters in India – What You Must Know
The Unique Device Identification (UDI) system is a global initiative to improve traceability of medical devices in the supply chain. CDSCO is aligned with this system and has been implementing UDI requirements under MDR 2017.
What Is UDI?
A UDI is a unique numeric or alphanumeric code assigned to a medical device. It consists of two parts: the Device Identifier (DI), which identifies the device model, and the Production Identifier (PI), which identifies the specific unit (batch, serial number, expiry date).
UDI Requirements for Oximeters in 2026
• Class B devices including pulse oximeters must have UDI codes on the device label.
• The UDI must be registered in the CDSCO UDI database through the SUGAM portal.
• UDI must be in a machine-readable format (barcode or QR code) on the packaging.
• The UDI must match the device information registered with CDSCO.
If you are importing from a foreign manufacturer who already has a GS1 UDI assigned, you can use that identifier. However, it must still be registered in India's UDI database.
Online Selling Compliance for Pulse Oximeters in India
With platforms like Amazon India, Flipkart, 1mg, and Netmeds driving significant medical device sales, online compliance has become a major focus for CDSCO enforcement. Here is what you need to know if you are selling oximeters online:
- CDSCO License is mandatory: E-commerce platforms now require sellers to submit their CDSCO registration/import license before listing medical devices. Amazon India and Flipkart have specific medical device compliance programs.
- License number on listing: The product listing should ideally display the CDSCO license number and the importer or manufacturer name.
- No false claims: Marketing claims must match the device's intended use as approved by CDSCO. Claims like 'medical-grade', 'hospital-grade', or 'clinical accuracy' must be substantiated by test data.
- Consumer Devices vs Clinical Devices: Some sellers try to sell clinical oximeters as 'wellness devices' to avoid CDSCO oversight. This is a risky strategy and has been flagged by CDSCO in enforcement actions.
- GST and invoicing: Proper GST-compliant invoices must mention the device name, HSN code, and CDSCO license number.
| E-commerce Seller Alert: CDSCO and state drug authorities have intensified online surveillance since 2023. Non-compliant sellers — especially those on B2C platforms — have faced seizures and show-cause notices. Do not risk your business by selling without proper registration. |
Validity and Renewal of CDSCO Registration for Pulse Oximeters
Many importers and manufacturers overlook renewal timelines and end up selling with an expired license — which is treated the same as selling without a license. Here is what you need to know:
| License Type | Initial Validity | Renewal Period | Renewal Timeline |
| Import License (MD-15) | As long as compliant / 5 years (as per CDSCO) | Every 5 years | Apply 3 months before expiry |
| Manufacturing License (MD-5) | As long as GMP compliance maintained | Annual/Periodic inspection | Apply before expiry |
| UDI Registration | Ongoing – update with any label change | N/A (update on change) | Immediate on label change |
Renewal applications must be filed through the SUGAM portal. Updated test reports, label changes, and any manufacturing site changes must be reflected in the renewal application.
Common Mistakes to Avoid in CDSCO Oximeter Registration
After working with numerous importers and manufacturers, these are the most frequent errors that delay registration or lead to rejection:
- Submitting expired test reports: IEC 60601-1 and ISO 9919 test reports must be current. CDSCO does not accept reports older than 5 years for new applications.
- Wrong authorized agent format: The IAA agreement must be in CDSCO's prescribed format and notarized. A simple letter of authorization is not sufficient.
- Missing IFU in English: Instructions for Use must be in English for CDSCO review. Providing only Chinese or other language IFU is a common deficiency.
- Applying for wrong device classification: Some applicants try to classify their oximeter as a Class A device. CDSCO's classification database is clear — pulse oximeters are Class B.
- Not updating labels before submission: Submitting labels that do not include the CDSCO license number (which is not yet available at the time of application) is normal — but the final approved label must include it. Have a label revision plan ready.
- Ignoring state-level requirements: For manufacturers, SLA requirements vary by state. What works in Maharashtra may have slight differences in Karnataka or Tamil Nadu.
- Not tracking the SUGAM portal: CDSCO communicates deficiency notices and queries through the SUGAM portal. If you are not checking your dashboard regularly, you may miss deadlines.
Who Is Eligible to Apply for CDSCO Oximeter Registration?
Eligibility requirements differ for importers and manufacturers:
For Import License Applicants
• Must be an Indian company, LLP, or sole proprietorship registered in India.
• Must be authorized in writing by the foreign manufacturer.
• Must have a registered office in India with a valid GST registration.
• Must have a qualified person responsible for regulatory compliance.
For Manufacturing License Applicants
• Must own or lease a manufacturing facility that meets Schedule M GMP requirements.
• Must employ a Qualified Technical Person (QTP) as per MDR 2017 requirements.
• Must have a quality management system in place (ISO 13485 certification preferred).
• The facility must be inspectable by SLA and CDSCO officers.
Benefits of Getting CDSCO Registration for Pulse Oximeters
Beyond legal compliance, CDSCO registration delivers real business benefits that are often underestimated:
- Legal right to sell: Only CDSCO-registered devices can be legally imported, manufactured, and sold in India. Registration protects you from enforcement actions.
- Market access: Large hospital networks, government tenders, and institutional buyers only purchase from CDSCO-registered suppliers. Without it, you are locked out of a massive B2B market.
- E-commerce listing: Major platforms require CDSCO registration for medical device listings. Registration opens up the INR 3,000+ crore online medical device market.
- Trust and brand credibility: Consumers and healthcare professionals increasingly look for CDSCO registration as a mark of quality and safety. It differentiates your product in a crowded market.
- Export opportunities: Some international markets accept CDSCO registration as a reference standard, easing reciprocal market access.
- Protection from counterfeits: Registered devices are listed on CDSCO's public database, giving consumers a way to verify authenticity.
Conclusion
Getting CDSCO registration for a pulse oximeter in India is not as complicated as it might seem — but it does demand attention to detail, the right documentation, and a clear understanding of the process. The regulatory environment under MDR 2017 has matured significantly, and CDSCO enforcement has become more systematic in recent years.
Whether you are importing from China, setting up a manufacturing unit in India, or launching a branded oximeter product for the e-commerce market, the fundamentals are the same: classify your device correctly, prepare a complete technical dossier, appoint a qualified Indian Authorized Agent, and submit through the SUGAM portal with all mandatory documents.
The investment in proper registration pays off — not just in legal protection, but in market credibility, access to institutional buyers, and long-term business sustainability in India's growing medical device sector.
If you are not sure where to start, the safest first step is a regulatory gap analysis with a CDSCO-experienced consultant. Most delays and rejections are entirely avoidable with the right guidance.
Frequently Asked Questions
Is CDSCO registration mandatory for all pulse oximeters sold in India?
Yes. Under the Medical Devices Rules, 2017, all pulse oximeters — whether imported or manufactured in India — require CDSCO registration before they can be legally sold, stocked, or distributed. There are no exemptions for consumer-grade or lower-priced devices if they make SpO2 measurement claims.
How long does it take to get CDSCO approval for an oximeter?
For import registration (Form MD-14/MD-15), the typical timeline is 45 to 90 working days from the date of complete application submission. Manufacturing licenses can take 60 to 120 working days, including site inspection by the State Licensing Authority. Having all documents in order from the start significantly reduces this timeline.
Can a foreign manufacturer apply directly for CDSCO registration?
No. Foreign manufacturers cannot apply directly. They must appoint an Indian Authorized Agent (IAA) — an Indian company that acts as their local representative. All correspondence, applications, and post-market obligations are managed through the IAA. The IAA must sign a formal agreement with the foreign manufacturer.
What is the cost of CDSCO registration for a pulse oximeter?
The government fee for a Class B import license is Rs. 1,00,000 per device (approximately USD 1,200). Manufacturing license fees for Class B are Rs. 50,000 per device. These are statutory fees only. Professional and consulting fees, lab testing, and document translation are additional costs.
Do I need a separate license for each oximeter model?
Yes. In most cases, each distinct device model requires a separate CDSCO registration. However, if devices are in the same product family with similar design, intended use, and manufacturing process, CDSCO may allow them under a single application as device variants — this must be discussed with a regulatory consultant or CDSCO directly during pre-submission.
What is the validity of a CDSCO import license for oximeters?
CDSCO import licenses for medical devices are valid as long as the device and its regulatory compliance remain current, typically reviewed every five years. You must apply for renewal before the expiry date. Any change in manufacturer, model, or labeling may require an amendment or fresh application.
What happens if I sell oximeters without CDSCO registration?
Selling, importing, manufacturing, or stocking medical devices without proper CDSCO registration is a punishable offense under the Drugs and Cosmetics Act, 1940 (as amended to include medical devices) and MDR 2017. Penalties include seizure of goods, cancellation of business licenses, fines, and in serious cases, imprisonment. Online sellers risk permanent platform bans.
Is CE certification sufficient for selling oximeters in India?
No. CE certification (European conformity) is a prerequisite document for the CDSCO import registration application, but it does not replace CDSCO registration. You must still obtain a valid Import License from CDSCO before the device can enter or be sold in the Indian market.
Do I need ISO 13485 to register a pulse oximeter with CDSCO?
For import registration, the foreign manufacturer must hold ISO 13485 certification, which is a mandatory document in the application checklist. For Indian manufacturers applying for a manufacturing license, ISO 13485 is strongly recommended and may be required by the SLA in many states. It also substantially strengthens your technical dossier during CDSCO review.
Where can I check if an oximeter is CDSCO registered?
CDSCO maintains a public online database of registered medical devices at cdsco.gov.in. You can search by device name, manufacturer, or import license number to verify the registration status of any pulse oximeter. This database is also used by enforcement authorities, hospitals, and e-commerce compliance teams to check for valid registration.






































