- Step-by-step process to obtain CDSCO import license for anesthesia machines using Form MD-14 (new license) and MD-15 (loan/exhibition license).
- Full document checklist, eligibility criteria, official government fees, and realistic timelines — based on the Medical Devices Rules, 2017.
- Renewal and post-approval compliance requirements every importer must follow to stay legally compliant in 2026.• Answers to the 10 most common questions asked by importers, hospitals, and distributors navigating CDSCO's medical device licensing system.
Introduction
If you are planning to import anesthesia machines into India, getting your CDSCO import license is not optional — it is a legal mandate. Under the Medical Devices Rules, 2017, anesthesia machines are classified as Class C or Class D medical devices, and no shipment can legally enter India without a valid license issued by the Central Drugs Standard Control Organization (CDSCO).
This guide breaks down everything you need to know — from understanding the difference between MD-14 and MD-15 forms, to eligibility, document requirements, fees, timelines, and what happens at renewal. Whether you are a first-time importer or a distributor looking to streamline your compliance, you will find clear, actionable answers here.
What Is the CDSCO Import License for Anesthesia Machines?
The CDSCO import license is an official authorization issued by the Central Drugs Standard Control Organization under the Ministry of Health and Family Welfare, Government of India. It permits an individual or company to legally import medical devices — including anesthesia machines — into India for sale or distribution.


Anesthesia machines fall under the regulated category of medical devices as per Schedule III of the Medical Devices Rules, 2017. These are life-support critical devices used in surgeries and intensive care settings, which is why CDSCO maintains strict regulatory oversight over their import.
Without this license, any attempt to import, sell, stock, exhibit, or distribute anesthesia equipment in India is considered a legal violation and can attract penalties, seizure of goods, or criminal action.
| Why This Matters in 2026:India's medical device import regulations have been progressively tightened since 2017. CDSCO has increased enforcement activity, and platforms like the Sugam Portal now make tracking non-compliant importers easier. Getting licensed before importing is not just a regulatory requirement — it protects your business investment. |
MD-14 vs MD-15: Understanding the Two Forms
One of the most common points of confusion for importers is the difference between Form MD-14 and Form MD-15. Here is a clear breakdown:
| Parameter | Form MD-14 | Form MD-15 |
| Purpose | Import license for sale and distribution | License for loan or exhibition purposes only |
| Use Case | Commercial import, stocking, selling | Import for demo, trial, or medical exhibition |
| Validity | 3 years (renewable) | Limited period as specified in the license |
| Applicable To | All Class A, B, C, D medical devices | Same device categories, restricted use only |
| Fee Structure | Varies by device class and quantity | Lower fee; purpose-restricted |
| Who Applies | Importers, distributors, authorized agents | Hospitals, manufacturers, event organizers |
| Post-License Requirement | Full compliance including labeling, storage, recalls | Device must not be sold commercially |
In simple terms: if you plan to import anesthesia machines to sell them in the Indian market, you need Form MD-14. If you are bringing in a machine temporarily for a medical exhibition, a hospital trial, or a loan arrangement, Form MD-15 is the route to go.
Most commercial importers and authorized Indian representatives of foreign manufacturers will work with Form MD-14. Form MD-15 is more relevant for companies exploring the Indian market before committing to full-scale distribution.
Who Is Eligible to Apply for a CDSCO Import License?
CDSCO has specific eligibility conditions for applicants. Meeting these before you apply saves time and avoids rejection.
Eligibility Criteria for Form MD-14 (Commercial Import License)
- The applicant must be a registered company or firm in India — a proprietorship, partnership, LLP, or private/public limited company.
- The applicant should have a valid business address in India from which the imported device will be distributed or stored.
- The applicant must appoint a technically qualified person (TQP) — typically someone with a science or pharmacy degree — who is responsible for regulatory compliance.
- The foreign manufacturer must hold a valid manufacturing license or equivalent regulatory approval in their country of origin (e.g., FDA 510(k) clearance in the USA, CE Mark in Europe, or PMDA approval in Japan).
- The device must be registered or notified with CDSCO as per the Medical Devices Rules, 2017 (registration is separate from the import license).
- The applicant must not have any prior license cancellations or criminal convictions related to drug or medical device violations in India.
Eligibility Criteria for Form MD-15 (Loan/Exhibition License)
- Any registered Indian entity, hospital, medical college, or event organizer can apply.
- The purpose must be clearly stated — exhibition, demonstration, trial, or loan — and cannot include commercial sale.
- The device must return or be disposed of as per CDSCO conditions after the authorized period ends.
Document Checklist for CDSCO Import License Application
Getting your documents right the first time is the biggest factor in avoiding delays. CDSCO applications that are incomplete are returned, and resubmission restarts processing timelines. Here is the complete document list for an anesthesia machine import license:
Documents from the Indian Applicant (Importer)
| S.No. | Document Required | Details / Notes |
| 1 | Covering Letter | On company letterhead, addressed to the Central Licensing Authority (CLA), CDSCO |
| 2 | Completed Form MD-14 or MD-15 | Filled online via Sugam Portal (sugamweb.in); physical copy signed by authorized signatory |
| 3 | Certificate of Incorporation | Issued by MCA (Ministry of Corporate Affairs); for firms, Partnership Deed or LLP Agreement |
| 4 | GST Registration Certificate | Valid GST number mandatory for all commercial importers |
| 5 | Technically Qualified Person (TQP) Details | Name, qualification, appointment letter, and signature specimen |
| 6 | Site Master File / Premises Details | Address proof of warehouse or business premises + NOC from property owner if rented |
| 7 | Power of Attorney / Board Resolution | Authorizing the signatory to apply on behalf of the company |
| 8 | Demand Draft / Online Fee Payment Proof | As per CDSCO fee schedule; drawn in favor of 'Drugs Controller General (India)' |
Documents from the Foreign Manufacturer
| S.No. | Document Required | Details / Notes |
| 1 | Free Sale Certificate (FSC) | Issued by the competent authority in the country of origin; apostilled or notarized |
| 2 | Manufacturing License | Valid license to manufacture the specific device in the country of origin |
| 3 | ISO 13485 Certificate | Quality management system certification for medical device manufacturers |
| 4 | CE Mark / FDA Clearance / PMDA Approval | Applicable regulatory approval in the country of origin or major market |
| 5 | Authorization Letter | Authorizing the Indian applicant as the exclusive importer or authorized representative |
| 6 | Technical Documentation / Dossier | Device description, specifications, intended use, labeling, IFU (Instructions for Use) |
| 7 | Test Reports / Performance Data | Clinical and performance data; bio-compatibility data as per IS/ISO standards |
| 8 | List of Countries Where Device Is Marketed | Helps establish device's global regulatory standing |

| Pro Tip — Apostille Requirement: All documents from foreign manufacturers must be apostilled or notarized by a competent authority in the originating country. CDSCO does not accept plain photocopies or unattested translations. This step alone adds 2-4 weeks if not planned in advance. |
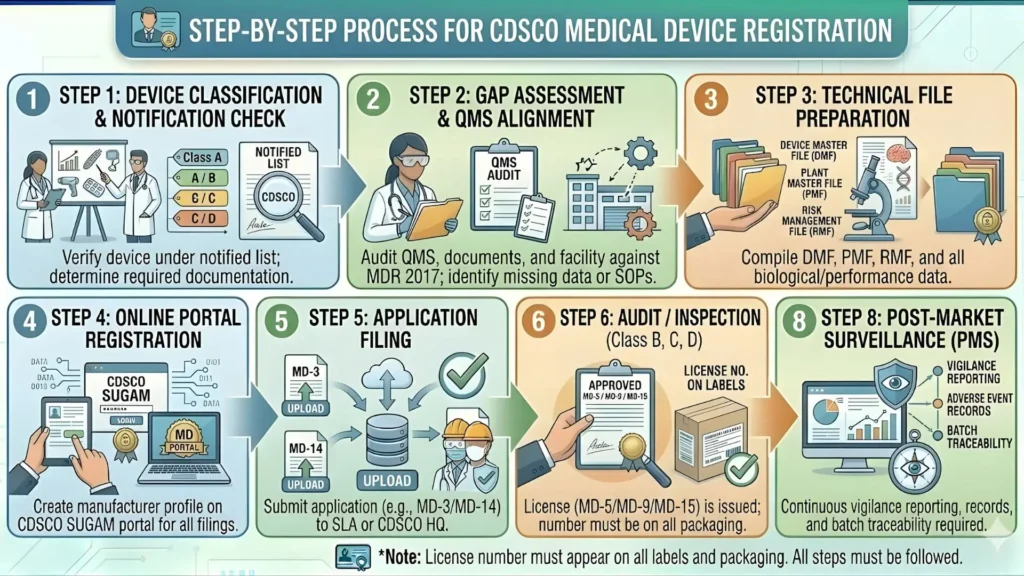
Step-by-Step Application Process on the CDSCO Sugam Portal
Since 2020, CDSCO has moved the import license application process almost entirely online through its Sugam Portal (sugamweb.in). Here is how the process works in 2026:
Step 1 — Register on the Sugam Portal
Visit sugamweb.in and create a new account as an 'Importer.' You will need your company PAN, GST, and a valid email address. Verification takes 1-2 working days.
Step 2 — Select the Correct Form
Navigate to 'Medical Devices' > 'Import License' > Choose Form MD-14 (for commercial import) or Form MD-15 (for loan/exhibition). Do not confuse these — submitting the wrong form causes rejection.
Step 3 — Fill the Online Application Form
Enter all mandatory fields: applicant details, device details, manufacturer details, intended use, and TQP information. The portal auto-validates certain fields, so errors are flagged before submission.
Step 4 — Upload Supporting Documents
Upload all required documents in PDF format (max 5 MB per file). Ensure scans are legible. Blurry or incomplete uploads are a common cause of rejection.
Step 5 — Pay the Application Fee
Pay online via the Sugam Portal's payment gateway (net banking, UPI, or credit/debit card). Keep the payment receipt. Alternatively, you can submit a Demand Draft at the CDSCO office.
Step 6 — Application Review by CDSCO Officer
A designated CDSCO officer reviews the application. If additional information or clarification is needed, a query is raised on the portal. You typically have 30 days to respond to queries before the application is closed.
Step 7 — Inspection (If Required)
For first-time applicants or those without prior regulatory history, CDSCO may schedule a premises inspection. Ensure your warehouse or storage facility meets the Good Distribution Practice (GDP) requirements.
Step 8 — License Issuance
Once approved, the import license (Form MD-14 or MD-15) is issued digitally through the Sugam Portal. Download and store it securely. A physical copy is usually generated as well.

CDSCO Import License Fees for Anesthesia Machines (2026)
Fees for medical device import licenses are governed by the Second Schedule of the Medical Devices Rules, 2017. Note: fee structures are periodically revised by the government. Always verify the current fee on the official Sugam portal before payment.
| Application Type | Device Class | Government Fee (Approx.) | Notes |
| Form MD-14 — New License | Class C (e.g., standard anesthesia machines) | INR 50,000 per device | Per device/model basis |
| Form MD-14 — New License | Class D (e.g., advanced critical care anesthesia systems) | INR 50,000 per device | Subject to revision by MoHFW |
| Form MD-14 — Amendment | Class C / Class D | INR 25,000 | For changes to existing license |
| Form MD-15 — Loan/Exhibition | Class C / Class D | INR 1,000 – 5,000 | Lower fee; for temporary import |
| Form MD-14 — Renewal | Class C / Class D | 50% of original fee | Applied before expiry |
| Duplicate License | Any class | INR 1,000 | For lost or damaged license |
| Important Note on Fees: The fees mentioned above are government fees only (as per Second Schedule, Medical Devices Rules, 2017). If you are engaging a regulatory consultant or CRO (Contract Research Organization) for the application, their professional fees are separate and additional. Always verify exact current fee amounts on sugamweb.in as government fee schedules are amended from time to time. |
Processing Timeline: How Long Does It Take?
This is the question every importer asks first. The honest answer: it depends on your application quality and device classification. Here is a realistic breakdown:
| Stage | Estimated Time | Key Dependency |
| Document preparation and notarization | 4–8 weeks | Foreign manufacturer response speed; apostille process abroad |
| Sugam Portal registration and application filing | 1–3 days | Complete documents ready before filing |
| CDSCO primary review (scrutiny) | 30–45 working days | Application completeness; officer workload |
| Query resolution (if raised) | 15–30 days | Applicant's response time to CDSCO queries |
| Premises inspection (if required) | 2–6 weeks | CDSCO scheduling; first-time applicants |
| Final approval and license issuance | 10–15 working days after inspection | No pending queries or issues |
| Total (realistic estimate — first-time) | 4 to 6 months | Varies; experienced importers may complete in 3 months |
| Total (renewal — existing licensee) | 45–90 days | Renewal filed 3 months before expiry is recommended |
CDSCO does not have a publicly stated statutory timeline for license issuance. The timelines above reflect actual industry experience. Delays are common for first-time applicants, for devices with incomplete manufacturer dossiers, or during periods of high application volumes.
Validity and Renewal of the CDSCO Import License
License Validity
- Form MD-14 import licenses for anesthesia machines are valid for 3 years from the date of issuance.
- Form MD-15 (loan/exhibition) licenses are valid for the specific period mentioned in the license — typically days to a few months.
- The license is device-specific and manufacturer-specific. If you add a new anesthesia machine model or change the manufacturer, a fresh or amended license is required.
Renewal Process
Renewal is not automatic. You must proactively initiate it. Here is what to keep in mind:
- File the renewal application at least 3 months before the license expires. Late renewal may require you to stop importing until a new license is issued.
- The renewal application follows the same process as the original — through the Sugam Portal, with updated documents and fee payment.
- Updated Free Sale Certificates, revised ISO certificates (if expired), and updated manufacturer authorization letters are typically required at renewal.
- If you miss the renewal window and the license expires, you must apply for a fresh license — not a renewal — which restarts the entire timeline.
When Does a License Become Invalid?
- Expiry of the 3-year term without renewal.
- Cancellation by CDSCO due to non-compliance, adverse inspection findings, or consumer safety violations.
- Voluntary surrender by the licensee.
- Change in device classification requiring a new license category.
Post-License Compliance Requirements
Getting the license is just step one. Maintaining compliance is an ongoing obligation. CDSCO expects importers of anesthesia machines to adhere to the following:
| Compliance Area | Requirement | Frequency |
| Labeling | All imported anesthesia machines must carry labels in English with device name, model, manufacturer details, batch number, manufacturing date, expiry (if applicable), and importer name/address | At time of import |
| Storage Conditions | Maintain good distribution practices (GDP); appropriate storage for the device type; temperature, humidity as applicable | Ongoing |
| Record Keeping | Maintain import records, distribution records, and customer details for at least 5 years | Ongoing; audit-ready |
| Adverse Event Reporting | Report any serious adverse events (device malfunction, patient harm) to CDSCO within 7–30 days depending on severity | As applicable |
| Recall Management | Have a written recall procedure; execute manufacturer-initiated or CDSCO-directed recalls promptly | As directed |
| Import Inspector Cooperation | Allow CDSCO or state drug controller inspections of premises and records without obstruction | On demand |
| Device Registration Linkage | Ensure the imported device is registered under its own CDSCO registration (separate from import license) | Before import |
| Annual Updates to CDSCO | Inform CDSCO of any change in address, TQP, or manufacturer authorization within 30 days | As applicable |
Common Reasons for CDSCO Import License Rejection or Delay
Understanding why applications fail helps you avoid the same pitfalls. Here are the most frequently observed reasons:
Incomplete document package:
Missing apostille, outdated Free Sale Certificate, or absence of ISO 13485 certificate are top reasons for scrutiny failure.
Wrong device classification:
Anesthesia machines fall under Class C or D. Applying under an incorrect class leads to rejection and fee non-refundability.
Authorization letter issues:
If the foreign manufacturer's authorization letter is generic, undated, or not notarized, CDSCO typically raises a query or rejects the application.
TQP qualification mismatch:
The Technically Qualified Person must hold a recognized qualification in science, pharmacy, or engineering. An unqualified TQP is a common rejection ground.
Delayed query response:
Failing to respond to CDSCO's query within 30 days results in application closure. Monitor your Sugam Portal account regularly.
Device not registered with CDSCO:
The import license and device registration are separate processes. Importing without device registration invites penalties regardless of license status.
Benefits of Obtaining a CDSCO Import License for Anesthesia Machines
Beyond regulatory compliance, there are tangible business benefits to being a licensed anesthesia machine importer in India:
- Legal authority to import, stock, sell, and distribute — without the fear of CDSCO enforcement action or customs detention.
- Credibility with hospitals, government tenders, and institutional buyers who require licensed vendor status before placing purchase orders.
- Access to the government procurement ecosystem — GeM (Government e-Marketplace) and CPWD tenders typically require CDSCO import license proof.
- Brand protection — a CDSCO license signals to healthcare providers that the equipment meets India's quality and safety standards.
- Foundation for building an authorized distribution network — you can legally appoint sub-distributors and dealer networks across India.
- Reduced customs clearance friction — licensed importers face fewer detentions at ports of entry compared to entities importing without proper documentation.
Conclusion
Obtaining a CDSCO import license for anesthesia machines is a non-negotiable step for any company looking to legally participate in India's growing medical device market. The process involves careful preparation — assembling the right documents from both the Indian importer and the foreign manufacturer, understanding the difference between Form MD-14 and MD-15, and navigating the Sugam Portal systematically.
The regulatory landscape in 2026 is more enforcement-focused than ever. CDSCO has strengthened port-level checks, increased online surveillance of unlicensed medical device trade, and has also simplified the application process through digital tools. This means the barrier to entry for compliant importers is lower, but the consequences for non-compliance are sharper.
Start your application with complete documents, assign a qualified TQP, ensure your foreign manufacturer is cooperative and responsive, and file at least six months before you need to start importing. If this is your first application, working with a CDSCO-experienced regulatory consultant can significantly reduce the timeline and rejection risk.
Frequently Asked Questions
Is a CDSCO import license mandatory for all anesthesia machines?
Yes. As per the Medical Devices Rules, 2017, anesthesia machines are regulated medical devices. Any import for sale, distribution, or stocking in India requires a valid CDSCO import license (Form MD-14). Import for trial or exhibition requires Form MD-15. There is no exemption for hospitals importing for self-use in commercial quantities.
How is an anesthesia machine classified under CDSCO — Class C or Class D?
Anesthesia machines are generally classified as Class C (moderate risk) under the Medical Devices Rules, 2017. However, certain advanced anesthesia workstations with integrated monitoring, life-support functionality, or intended for use in critical care settings may be classified as Class D (high risk).
Can a foreign manufacturer apply for the CDSCO import license directly?
No. The CDSCO import license must be applied for by an Indian entity. The foreign manufacturer must authorize an Indian importer, agent, or subsidiary as their representative. The license is issued in the name of the Indian applicant (importer), not the foreign manufacturer.
What is the difference between CDSCO device registration and the import license?
These are two separate authorizations. Device registration (under Form MD-14 for the device itself, or previously under Form 40) establishes that the device meets Indian regulatory requirements.
What happens if I import anesthesia machines without a CDSCO license?
Importing regulated medical devices without a valid CDSCO import license is a punishable offence under the Drugs and Cosmetics Act, 1940 (as applicable to medical devices) and the Medical Devices Rules, 2017.
Does the CDSCO import license cover all models from the same manufacturer?
No. The import license is model-specific. If you import three different models of anesthesia machines from the same manufacturer, you may need separate licenses for each model, or a license amendment listing all models.
Is premises inspection mandatory for all applicants?
Not always. Premises inspection is typically conducted for first-time applicants or when CDSCO has specific concerns about storage and handling capabilities. Repeat applicants with a clean compliance history may not face routine inspections.
Can I transfer or sell my CDSCO import license to another company?
No. CDSCO import licenses are non-transferable. They are issued specifically to the applicant entity for the specified device. If your company is acquired, merged, or restructured, you need to apply for a fresh license under the new entity's name.
What is the Sugam Portal and how does it work for import license applications?
Sugam (sugamweb.in) is CDSCO's official online portal for regulatory submissions, including medical device import license applications. All new applications, renewals, amendments, and fee payments for import licenses are processed through this portal.
How do I renew my CDSCO import license before it expires?
Log into your Sugam Portal account and navigate to your existing license. Select the 'Renewal' option, update any changed information (TQP details, manufacturer authorization, updated certificates), upload fresh documents where required, and pay the renewal fee (typically 50% of the original application fee).






































