- Medical Device For Skin Patch Registration in India are regulated exclusively under the Medical Devices Rules (MDR) 2017 — separate from the pharmaceutical or cosmetics pathway.
- Risk classification from Class A to Class D determines your documentation burden, timeline, and whether a notified body assessment is mandatory.
- All overseas manufacturers must appoint an Indian Authorized Representative before a single SUGAM portal application can be filed.
- From standard wound dressings to TENS patches, diagnostic patches, and biosensor-integrated systems — this guide covers exactly what CDSCO requires in 2026.
Introduction
India is one of the fastest-growing medical device markets in the world. The wound care segment is expanding at a significant pace, and advanced skin patch technologies — from electrotherapeutic systems to biosensor-integrated patches — are seeing strong demand across hospitals, clinics, and home care settings. But for any overseas manufacturer or importer planning to bring a medical device skin patch into India, one framework governs every step of your market entry: the Medical Devices Rules, 2017.


Understanding MDR 2017 in the context of skin patch products specifically — what it requires, how your product will be classified, what documents you must prepare, and how the SUGAM portal process actually works — is not just useful background knowledge. It is the difference between a predictable, well-managed approval process and a series of costly delays that push your India launch back by a year or more.
This guide is written for manufacturers, importers, and regulatory teams who are serious about getting their medical device skin patch registration in India right the first time in 2026.
What is Medical Device For Skin Patch Registration?
Medical device registration in India is the formal process by which a manufacturer or importer obtains legal authorization from the Central Drugs Standard Control Organization (CDSCO) to market and sell a medical device in the Indian territory. Under the Medical Devices Rules (MDR) 2017, no medical device — including any skin patch product regulated as a medical device — may be imported, stocked, distributed, or sold in India without a valid Registration Certificate issued by CDSCO.
Registration is not a one-time administrative formality. It is a structured regulatory review process in which CDSCO evaluates whether the medical device meets the Essential Principles of Safety and Performance prescribed under MDR 2017, whether the manufacturer operates a compliant quality management system, and whether the product's intended purpose is adequately supported by technical and clinical evidence.
For skin patch products specifically, registration establishes that the device is safe for application to human skin, performs as the manufacturer claims, and will be marketed in India under appropriate labelling and post-market controls. The registration pathway a skin patch follows — and the documentary evidence required — depends entirely on its assigned risk class under MDR 2017.
Why Registration is Mandatory for Skin Patches in India
India's medical device regulatory framework underwent a fundamental transformation with the notification of MDR 2017, which brought medical devices — including skin patch products — under a dedicated, structured licensing regime for the first time. Before MDR 2017, many device categories operated in a regulatory grey zone. Today, registration is a legal prerequisite without exception.
The requirement applies across all categories of skin patch products regulated as medical devices, including:
- Wound dressings and wound care patches of any complexity
- Electrotherapeutic patches such as TENS and neuromuscular stimulation systems
- Diagnostic and continuous monitoring patches
- Biosensor-integrated wearable patches
- Heat therapy and physical energy delivery patches
Failure to register before placing a skin patch product on the Indian market is a direct violation of MDR 2017 and can result in product seizure, significant financial penalties, and in serious cases, criminal prosecution of the responsible parties.
The Legal Basis: MDR 2017 and CDSCO Authority
Medical device registration in India is governed by the Medical Devices Rules, 2017, notified under the Drugs and Cosmetics Act, 1940. CDSCO — functioning under the Directorate General of Health Services, Ministry of Health and Family Welfare — is the national regulatory authority responsible for granting, maintaining, and where necessary suspending or cancelling registration certificates for medical devices.
The MDR 2017 framework establishes the following core elements that apply to all skin patch registrations:
| Legal Element | What It Means for Skin Patch Registration |
| Registration Certificate | The legal document issued by CDSCO that authorises a specific skin patch product to be imported and marketed in India |
| Indian Authorised Representative (IAR) | A mandatory India-based legal entity that holds the registration certificate on behalf of the overseas manufacturer and bears legal responsibility for the product in India |
| Essential Principles of Safety & Performance | The MDR 2017 benchmark against which every skin patch's safety and performance evidence is assessed during CDSCO review |
| Risk Classification (Class A–D) | The risk tier assigned to the skin patch that determines the documentation burden, notified body requirements, and overall registration timeline |
| SUGAM Portal | The online government portal through which all medical device registration applications in India are submitted, tracked, and managed |
| Post-Market Obligations | The ongoing surveillance, vigilance reporting, and change notification duties that continue throughout the product's commercial life in India |
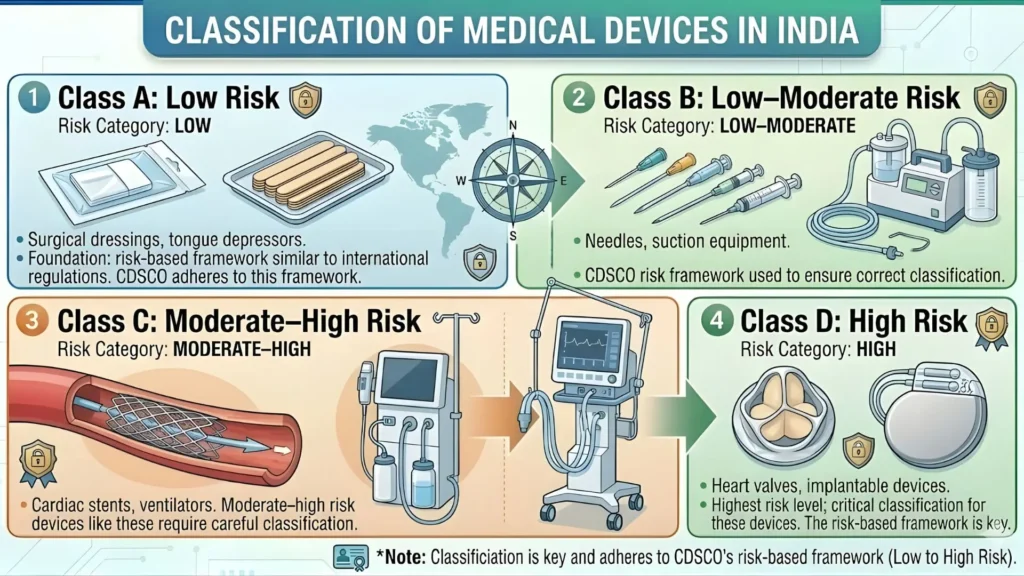
Risk Classification Under MDR 2017: Understanding Your Class
The Medical Devices Rules 2017 divide medical devices into four risk classes — Class A, Class B, Class C, and Class D — in ascending order of risk. The class your skin patch is assigned to determine the documentation you need, whether a third-party notified body assessment is required, and how long the overall CDSCO registration process will take.
Classification is based on criteria set out in the Third Schedule of MDR 2017, which considers whether the device is invasive or non-invasive, whether it is active (powered) or passive, the intended duration of use, and the potential consequences of device failure.
Class A — Low Risk
Standard wound dressings, non-medicated adhesive patches, and simple thermal comfort patches with no active energy delivery typically fall into Class A. These are non-invasive, passive devices that pose minimal risk in normal use. Class A registration is the most streamlined pathway — clinical evidence is not required, and the review timeline is significantly shorter than for higher-risk classes.
Class B — Low to Moderate Risk
Advanced wound care patches — including hydrocolloid dressings, foam dressings, alginate patches, and antimicrobial patches incorporating silver or similar agents — typically sit at Class B. The documentation burden is moderate, and while clinical performance evidence is expected, full clinical trial data is not always required for products with a well-established equivalence to predicate devices already on the Indian market.
Class C — Moderate to High Risk
Active electrotherapeutic devices, including TENS patches and neuromuscular stimulation patches, almost always classify as Class C. Diagnostic patches measuring internal physiological parameters and wound management systems with active electronic components also fall here. A critical requirement at Class C is assessment by a Notified Body recognized under MDR 2017. Detailed technical documentation, a complete risk management file, and clinical performance data are all required.
Class D — High Risk
Class D is the highest risk classification and applies to patches interfacing with implantable components or used in critical care contexts with direct life-support implications. Very few standalone skin patch products reach Class D, but where they do, a full technical dossier, clinical investigation data, and a notified body assessment are all mandatory requirements without exception.
MDR 2017 Risk Classification Table for Medical Device Skin Patches
| Class | Risk Level | Skin Patch Examples | Notified Body | Clinical Evidence | Timeline |
| Class A | Low | Basic wound dressings, simple adhesive patches | Not Required | Not Required | 4–8 months |
| Class B | Low–Moderate | Advanced wound care, hydrocolloid, antimicrobial silver patches | Not Required | Recommended | 6–12 months |
| Class C | Moderate–High | TENS patches, active electrotherapy, diagnostic patches | Mandatory | Required | 12–20 months |
| Class D | High | Biosensor patches, critical care, implantable interface | Mandatory | Full Clinical Data | 18–30 months |

Note: Timelines above reflect well-prepared submissions with complete documentation. Deficiency letters from CDSCO can extend these ranges by three to twelve months depending on the nature of the query.
Mandatory Indian Authorized Representative: A Non-Negotiable Legal Requirement
Before any overseas manufacturer can register a medical device skin patch in India, one legal requirement must be satisfied without exception: the appointment of an Indian Authorized Representative. This is not an administrative detail — it is a hard legal prerequisite under MDR 2017, and no application will be accepted without it.
The AIR is a company or individual physically based in India who takes on formal legal responsibility for the medical device in the Indian market. CDSCO treats the AIR as the primary point of contact for all regulatory matters relating to the product.
What the AIR is legally responsible for:
- Holding the registration certificate on behalf of the overseas manufacturer
- Receiving and responding to all official communications from CDSCO
- Managing post-market surveillance obligations in India
- Coordinating recalls, field safety corrective actions, and vigilance reporting
- Ensuring the product marketed in India conforms to the approved specifications at all times
The AIR appointment must be evidenced by a formal authorization letter from the overseas manufacturer specifying the scope of authorization, the products covered, the territory (India), and the duration of the appointment. This document is a mandatory part of the registration dossier — missing elements in the authorization letter are among the most commonly cited deficiencies in CDSCO reviews of medical device applications.
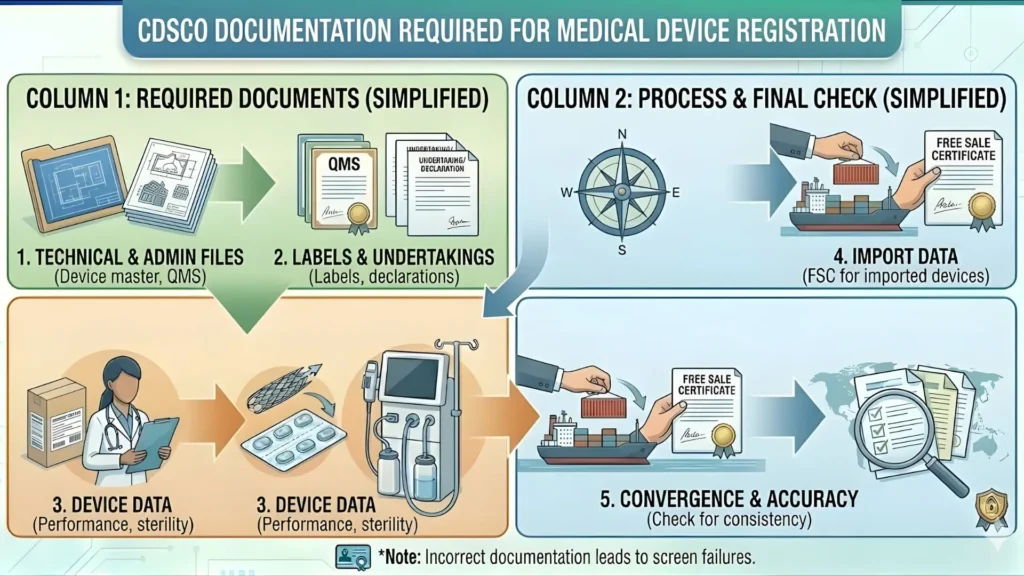
Technical Documentation File: What CDSCO Requires
The technical file for a medical device skin patch registration in India must demonstrate that the device is safe, performs as intended, and meets the applicable Essential Principles of Safety and Performance under MDR 2017. The depth of documentation required scales directly with the risk class of the device.
Core Technical Documents Required Across All Classes
| Document | What It Must Include |
| Device Description | Complete description of the patch — materials, dimensions, construction, intended use, and all components |
| Intended Purpose Statement | Clear, precise statement of the device's intended function and the target patient population |
| Risk Management File | Full hazard identification, risk control measures, and residual risk assessment aligned with ISO 14971:2019 |
| ISO 13485 Certificate | Valid QMS certification for the manufacturing facility — expired or out-of-scope certificates are rejected |
| Labelling (Schedule V) | All labels and instructions for use meeting MDR 2017 Schedule V requirements including IAR details |
| Performance Evidence | Bench testing, in vitro data, or clinical performance evidence scaled to risk class |
| Post-Market Surveillance Plan | Active monitoring plan covering complaints, adverse events, and periodic safety review obligations |
| Declaration of Conformity | Manufacturer declaration confirming conformity with all applicable Essential Principles |
| Notified Body Certificate | Required for Class C and Class D devices — must be from a CDSCO-recognised notified body |

Labelling Requirements Under MDR 2017 Schedule V
Indian labelling for medical device skin patches must comply with Schedule V of MDR 2017. Every mandatory element must be present on the label in English, clearly legible, and permanently marked. Claims on the label must reflect only what is covered by the approved intended purpose — overstating device capabilities or adding therapeutic claims not supported by the technical file is a serious compliance risk.
| Mandatory Label Element | Details Under MDR 2017 Schedule V |
| Device Name & Intended Purpose | Clearly stated on the primary label — must match the approved intended purpose statement |
| Manufacturer Name & Address | Full legal name and registered address of the overseas manufacturer |
| Indian Authorised Representative | Full name and Indian address — mandatory for all imported medical device skin patches |
| Batch / Lot Number | Unique batch identifier allowing full traceability through the supply chain |
| Date of Manufacture | Must be present in DD/MM/YYYY or MM/YYYY format |
| Expiry / Use-By Date | Clearly displayed — patch products with defined shelf life must state it explicitly |
| Storage Conditions | Temperature, humidity, and light conditions required for safe storage |
| Single-Use Indication | Where applicable, single-use symbol or written statement required |
| Warnings & Precautions | All relevant safety warnings including contraindications and patient population restrictions |
| Registration Number | CDSCO-issued registration number — added to label after approval is granted |
Additional Requirements for Class C and Class D Patches
Class C and Class D medical device skin patches require the following documents in addition to the core technical file:
- Notified Body assessment certificate from a CDSCO-recognized notified body
- Clinical performance evaluation report demonstrating clinical safety and effectiveness
- Summary of Safety and Clinical Performance (SSCP)
- Full clinical investigation data (mandatory for Class D)
- Enhanced post-market surveillance plan with defined periodic review cycles
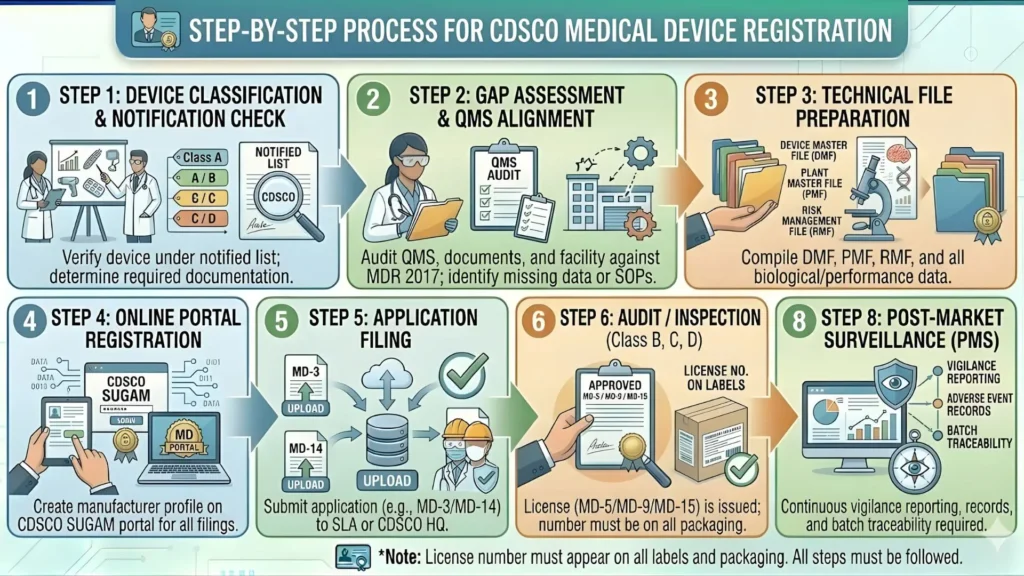
The SUGAM Portal: Step-by-Step Submission Process for 2026
All medical device skin patch registrations in India are processed through the SUGAM online portal managed by CDSCO. The portal is the single point of entry for all application types, from Class A wound dressings to Class D biosensor patches. Navigating SUGAM correctly from the very first step is critical — errors in form selection, document formatting, or data entry are among the most common causes of early-stage delays.
| Step | Action | Key Details for 2026 |
| 1 | Registration on SUGAM | The Indian Authorised Representative registers and creates a verified account on the SUGAM portal before any application work begins |
| 2 | Select Application Form MD-14 | MD-14 is the correct form for import registration of medical devices — selecting the wrong form results in rejection without substantive review |
| 3 | Upload Technical Documentation | All files in PDF format meeting CDSCO naming and size requirements — document structure must follow the SUGAM system sequence |
| 4 | Pay Prescribed Application Fee | Fees are per the First Schedule of MDR 2017, paid through the SUGAM payment gateway — fee receipt must be uploaded as part of the submission |
| 5 | Administrative Scrutiny | CDSCO checks for completeness and formatting — incomplete submissions are returned for correction before entering technical review |
| 6 | Technical Review | Substantive review of device description, classification, risk management, and performance evidence — most deficiency letters are issued at this stage |
| 7 | Deficiency Letter Response | IAR submits a formal, precise response addressing every query raised — quality of response directly determines how quickly review resumes |
| 8 | Registration Certificate Issued | CDSCO issues the Registration Certificate — the product can legally be imported and sold in India only after this certificate is in hand |

Practical points for SUGAM submissions in 2026:
- All uploaded PDFs must meet CDSCO's file size limits and follow specific naming conventions — files that do not conform are flagged during administrative scrutiny
- Application status on SUGAM is not always updated in real time — proactive follow-up by the IAR is often necessary to monitor progress accurately
- CDSCO query responses submitted through SUGAM must be precise and complete — partial responses result in further queries and additional timeline delays
- A single deficiency letter, if not responded to promptly and comprehensively, can add three to six months to the total registration timeline
Common Reasons Medical Device Skin Patch Applications Are Delayed
Even well-resourced international manufacturers regularly encounter delays in the Indian MDR 2017 registration process. The issues below come up consistently in CDSCO reviews and are entirely avoidable with proper preparation.
| Common Mistake | Why It Causes Delay or Rejection |
| Incorrect Risk Classification | A Class C electrotherapeutic patch submitted as Class B will lack the required notified body certificate and clinical evidence — guaranteed deficiency letter |
| Expired ISO 13485 Certificate | Quality management system certification must be valid at the time of submission and throughout the review period — expired certificates are rejected outright |
| Missing or Incomplete IAR Appointment Letter | The authorisation letter must specify the scope of products, the territory (India), and the duration — missing any element makes it non-compliant |
| Labelling Not Meeting Schedule V | Absent IAR details, missing batch information, or non-compliant claim language are among the most cited deficiencies in CDSCO reviews |
| Risk Management File Below ISO 14971 Standard | Incomplete hazard identification, missing risk control justifications, or absent residual risk statements consistently attract queries |
| No Notified Body Certificate for Class C/D | This is a hard requirement for Class C and Class D devices — submitting without it wastes the review cycle entirely |
| Overly Broad Intended Purpose Statement | Vague or expanded intended purpose claims that are not supported by the technical file create review complications and scope-of-use queries |
| Post-Market Surveillance Plan Absent | Many first-time applicants overlook this document — it is a mandatory component of the MDR 2017 technical file across all device classes |
Post-Registration Compliance: Obligations That Continue After Approval
Obtaining the Registration Certificate from CDSCO is the beginning of ongoing compliance under MDR 2017 — not the end of the regulatory journey. Manufacturers and IARs must have compliance management systems in place from day one of market entry.
| Compliance Obligation | What Is Required Under MDR 2017 |
| Licence Renewal | Registration Certificates have a prescribed validity period and must be renewed before expiry — lapsed certificates make the product non-compliant for import and sale |
| Post-Market Surveillance (PMS) | Active, documented monitoring of device performance in the Indian market including complaint management and trend analysis |
| Vigilance Reporting | Serious incidents and field safety corrective actions must be reported to CDSCO within defined timelines — failure to report is a regulatory offence |
| Change Notifications | Significant changes to device design, materials, manufacturing site, or intended purpose require prior CDSCO approval before implementation |
| Labelling Compliance Reviews | Label changes — including addition of the registration number and any content updates — must be reviewed against current Schedule V requirements |
| Periodic Safety Updates | For higher-class devices, periodic safety update reporting may be required to confirm ongoing benefit-risk profile |
Failure to meet post-approval obligations can result in suspension or cancellation of the Registration Certificate. In serious cases — particularly around failure to report serious incidents — enforcement action is a real risk.
Timelines for Medical Device Skin Patch Registration in India
Building your India market entry plan around realistic timelines — rather than best-case scenarios — is essential for commercial planning. The figures below are based on well-prepared, complete submissions. A single deficiency letter, which is common, can add three to twelve months depending on complexity.
| Device Class | Patch Type | Preparation Time | CDSCO Review | Total Range |
| Class A | Basic wound dressings | 1–2 months | 3–6 months | 4–8 months |
| Class B | Advanced wound care, antimicrobial | 2–4 months | 4–8 months | 6–12 months |
| Class C | TENS, electrotherapy, diagnostic | 4–6 months | 8–14 months | 12–20 months |
| Class D | Biosensor, critical care | 6–10 months | 12–20 months | 18–30 months |
Key factors that affect the total timeline:
- Completeness of the technical file at first submission — the single biggest variable in overall timeline
- Whether a notified body assessment is required — Class C and D patches must factor in notified body lead times before filing
- ISO 13485 certification status — an expiring or recently renewed certificate can cause delays if it lapses during the review period
- Quality of the deficiency letter response — comprehensive, precise responses keep the review moving; vague answers restart the clock
- CDSCO review workload — application volumes and internal review timelines at CDSCO can vary across the calendar year
Pre-Submission Checklist: Medical Device Skin Patch Registration India 2026
Before filing your application on the SUGAM portal, confirm each of the following is in place:
- Product confirmed as medical device (physical/mechanical mechanism — not pharmacological)
- Risk class determined — Class A, B, C, or D under MDR 2017 Third Schedule
- Indian Authorized Representative appointed with compliant authorization letter
- ISO 13485 certificate valid, in-scope, and not expiring during the review period
- Risk management file complete and aligned with ISO 14971:2019
- Notified body assessment obtained (mandatory for Class C and Class D)
- Labelling reviewed and compliant with MDR 2017 Schedule V requirements
- Intended purpose statement precise, clear, and fully supported by the technical file
- Post-market surveillance plan documented and included in the technical file
- SUGAM portal application form MD-14 selected — not a pharmaceutical or cosmetics form
- All documents in PDF format meeting CDSCO file naming and size requirements
- Commercial launch timeline accounts for realistic review period plus deficiency letter buffer
Getting Medical Device Skin Patch Registration Right in India
India's medical device market represents a genuine and growing opportunity for skin patch manufacturers across wound care, electrotherapy, and diagnostic monitoring. The MDR 2017 framework is well-structured and navigable — but it rewards precision, complete documentation, and a practical understanding of how CDSCO's review process works in practice.
The most expensive mistakes in India medical device registration are not the ones made during SUGAM submission — they are the ones made weeks or months earlier, during product classification and dossier preparation. A product submitted under the wrong class, with an incomplete risk management file, or without a compliant AIR appointment will not succeed regardless of how well the SUGAM portal process is managed.
For manufacturers approaching India for the first time in 2026, the best starting point is a thorough classification review and documentation gap assessment before any application work begins. Getting these fundamentals right creates a submission that moves through CDSCO review with minimal friction — and brings your product to market within a timeline your commercial team can plan around.
Frequently Asked Questions
Does a CE mark or US FDA clearance speed up the Indian registration process?
A CE mark or US FDA clearance is treated as supporting evidence by CDSCO and can strengthen the credibility of your technical file — particularly in demonstrating that the product has undergone rigorous conformity assessment. However, it does not replace the Indian registration process. All MDR 2017 documentation requirements apply independently of international approvals.
Can a single Indian Authorized Representative cover multiple products?
Yes. A single IAR can represent multiple products from the same overseas manufacturer, provided the formal authorization letter clearly identifies all products within its scope. The IAR must be capable of fulfilling their legal obligations for each product they represent — taking on an IAR role without the operational capacity to manage post-market surveillance and vigilance reporting is a compliance risk.
Is ISO 13485 mandatory for all classes of medical device skin patch?
Yes. ISO 13485 — the international standard for quality management systems in medical device manufacturing — is a baseline requirement under MDR 2017 for all device classes. An expired, suspended, or out-of-scope ISO 13485 certificate will result in a deficiency. Manufacturers should check certificate validity well in advance of filing and ensure renewal is completed before the submission is made.
My wound dressing contains silver — is it automatically Class B?
Antimicrobial wound dressings incorporating silver are typically classified as Class B under MDR 2017 because the silver acts through a physical antimicrobial mechanism. However, if the silver component is considered to act pharmacologically in the specific formulation, the product may be reviewed as a drug-device combination, which can trigger a different classification pathway. This is a nuanced area where specialist regulatory classification advice is strongly recommended before the application is prepared.
What happens if a medical device skin patch is sold in India without registration?
Selling an unregistered medical device in India is a direct violation of MDR 2017 and can result in seizure of the product, significant financial penalties, prohibition orders against the importer, and in serious cases, criminal prosecution. The risk falls on both the IAR and the overseas manufacturer. Market access through informal or unregistered channels is not sustainable and carries disproportionate legal exposure.
How does CDSCO view the intended purpose statement — can it be broad?
No. The intended purpose statement must be precise and directly supported by the technical file. Broad or expansive intended purpose claims that are not backed by performance evidence will attract queries. CDSCO's entire technical assessment is anchored to the stated intended purpose — getting this statement right, with appropriate specificity, is one of the most important decisions in preparing the registration dossier.





































