- CE Marking For Medical Devices is the legal gateway to sell medical devices across the EU and EEA — without it, your product cannot enter the market.
- EU MDR 2017/745 fully governs CE marking in 2026, replacing the old MDD with far stricter clinical evidence and post-market standards.
- EUDAMED is now fully operational — mandatory device registration and UDI tracking are actively enforced by EU competent authorities.
- The EU AI Act has added a new compliance layer for AI-powered medical devices, with key obligations phasing in from August 2026.
Introduction
CE marking for medical devices is not just a regulatory tick box. It is the cornerstone of market access across 27 EU member states and the broader European Economic Area. In 2026, the standards are tighter, the enforcement is stronger, and the consequences of getting it wrong — delayed launches, forced withdrawals, regulatory penalties — are more serious than ever.


What CE marking is, who needs it, what the requirements are, who is eligible, what the benefits are, how long certification lasts, how to renew, and answers to the ten most common questions manufacturers ask. Whether you are building your first device or managing a complex MDR transition, this is your starting point.
What Is CE Marking for Medical Devices?
CE stands for Conformité Européenne — European Conformity. The CE mark on a medical device tells regulators, healthcare professionals, and patients that the manufacturer has completed a conformity assessment and confirmed the device meets all applicable EU safety, performance, and labelling requirements.
CE marking is mandatory for every medical device placed on the EU or EEA market, regardless of where the manufacturer is based. A device manufactured in India, the United States, South Korea, or anywhere else in the world must still achieve CE marking before it can legally be sold in Europe.
The CE mark itself is not issued by a government body. It is the manufacturer's declaration — backed by technical documentation, clinical evidence, and in most cases a third-party Notified Body audit — that their device complies with EU law.
Which Regulations Govern CE Marking in 2026?
| Regulation | Covers | Effective Since |
| EU MDR 2017/745 | All medical devices — instruments, implants, software, diagnostics | May 26, 2021 |
| EU IVDR 2017/746 | In vitro diagnostic devices — tests, reagents, analyzers | May 26, 2022 |
| EU AI Act 2024/1689 | AI-powered devices and Software as a Medical Device (SaMD) | Phasing in 2025–2027 |
| Regulation EU 2024/1860 | Extended transitional provisions for legacy MDD-certified devices | In force 2024 |
CE Marking Eligibility Criteria: Who Needs It and Who Qualifies?
Before investing in the CE marking process, manufacturers need to answer two fundamental questions: Does my product qualify as a medical device under EU MDR? And am I, as the manufacturer or economic operator, eligible to apply?
Does Your Product Qualify as a Medical Device?
Under EU MDR Article 2, a product qualifies as a medical device if it is intended by the manufacturer to be used for one or more of the following specific medical purposes for human beings:
- Diagnosis, prevention, monitoring, prediction, prognosis, treatment, or alleviation of disease
- Diagnosis, monitoring, treatment, alleviation of, or compensation for an injury or disability
- Investigation, replacement, or modification of the anatomy or a physiological or pathological process or state
- Providing information by means of in vitro examination of specimens derived from the human body
- Control or support of conception
It is the manufacturer's stated intended purpose — in labelling, instructions for use, and marketing materials — that determines whether a product is a medical device. A product with genuine medical capability that is labelled only for 'general wellness' may fall outside MDR scope, but EU competent authorities scrutinize such claims carefully.
Software qualifies as a medical device (SaMD) when it performs a medical function for individual patients — not just population-level analytics. MDCG guidance 2019-11 provides the framework for software qualification and classification.
Who Can Apply for CE Marking?
The legal manufacturer is responsible for CE marking. Under EU MDR, the legal manufacturer is the entity that places the device on the market under its own name or trademark, regardless of whether it actually manufactured the product.
| Who | Role | CE Marking Responsibility |
| Manufacturer (EU-based) | Designs, produces, and places the device on market | Full responsibility — QMS, technical documentation, Declaration of Conformity |
| Manufacturer (Non-EU based) | Based outside EEA — sells into EU market | Must appoint an EU Authorised Representative (EU AR) who shares legal responsibility |
| EU Authorised Representative | Acts as official EU contact for non-EU manufacturers | Jointly liable; must be registered in EUDAMED; can be targeted by regulators |
| Importer | Introduces device from outside EU into EU market | Must verify CE mark is in place; must register in EUDAMED; label duties apply |
| Distributor | Makes device available in EU without altering it | Must verify CE mark before supplying; storage and transport obligations |
| Private Label / Own Brand Manufacturer | Rebrands another manufacturer's device under own name | Treated as legal manufacturer — full CE marking obligations apply |
Eligibility Conditions to Proceed with CE Marking
Not every manufacturer is ready to begin the formal CE marking process immediately. These conditions must be in place first:
- Confirmed device classification: You must have a documented, justified classification per EU MDR Annex VIII rules before selecting a conformity assessment route.
- Legal manufacturer identity established: Your organisation must be clearly identified as the legal manufacturer, with a registered address and appointed PRRC (Person Responsible for Regulatory Compliance).
- EU Authorized Representative contracted (if non-EEA): Non-EEA manufacturers must have a signed EU AR mandate in place before approaching a Notified Body.
- EUDAMED economic operator registration initiated: You must have applied for EUDAMED registration and obtained or be in the process of obtaining your Single Registration Number (SRN).
- QMS framework initiated: A quality management system aligned to ISO 13485:2016 must be in place or actively being implemented.
- Notified Body availability confirmed (for Class IIa+): Given 9–15 month waiting lists in 2026, confirming Notified Body availability before starting documentation can save significant time.
CE Marking Requirements for Medical Devices Under EU MDR
EU MDR 2017/745 sets out a comprehensive framework of requirements that every manufacturer must satisfy before affixing the CE mark. These are not light-touch obligations — they represent a significant regulatory workload, particularly for medium and high-risk device classes.
1. Device Classification
Classification determines your entire compliance pathway. Get it wrong and the consequences range from rejected submissions to post-market enforcement. EU MDR Annex VIII sets out 22 classification rules based on four key criteria: intended purpose, duration of contact with the body, degree of invasiveness, and whether the device is active or implantable.
| Class | Risk Level | Examples | Conformity Route |
| Class I | Low | Bandages, walking frames, non-sterile gloves, Class I software | Self-certification (manufacturer) |
| Class I sterile / measuring / reusable surgical | Low-Medium | Sterile dressings, surgical measuring instruments | Notified Body for QMS/production aspects only |
| Class IIa | Medium | Hearing aids, dental fillings, short-term contact lenses, ultrasound | Notified Body — QMS + Technical Documentation |
| Class IIb | Medium-High | Ventilators, surgical lasers, infusion pumps, X-ray machines | Notified Body — Full QMS + Technical Documentation |
| Class III | High | Heart valves, coronary stents, pacemakers, neural implants | Notified Body — Full QMS + Technical Documentation + Expert Panel (many cases) |
2. Quality Management System (QMS)
A QMS compliant with EU MDR Article 10(9) is mandatory for all manufacturers. The accepted benchmark is ISO 13485:2016, which provides a structured framework for quality in design, production, and post-market activities. The QMS must be actively maintained — not filed away after certification.
Key QMS elements required under EU MDR:
• Documented quality policy, objectives, and management review processes
• Design and development controls with full traceability
• Supplier qualification and management procedures
• Complaint handling and non-conformance management
• Corrective and Preventive Action (CAPA) system
• Internal audit programme and management review
• Person Responsible for Regulatory Compliance (PRRC) appointed and qualified
3. Technical Documentation
Technical documentation is the complete evidence dossier for your device. It must be prepared per EU MDR Annex II and Annex III and kept up to date throughout the device's market lifetime. For Class III devices and implantable Class IIb devices, a publicly available Summary of Safety and Clinical Performance (SSCP) must also be uploaded to EUDAMED.
| Documentation Component | What It Must Contain |
| Device Description and Intended Use | Full product description, intended patient population, clinical indications, contraindications, and variants/accessories |
| Design and Manufacturing Information | Design drawings, BOM, manufacturing processes, sterilisation method, packaging details |
| GSPR Checklist | Systematic mapping of each General Safety and Performance Requirement in Annex I to the evidence demonstrating compliance |
| Risk Management File | Complete ISO 14971 risk management process — hazard identification, risk estimation, mitigation, residual risk, benefit-risk analysis |
| Clinical Evaluation Report (CER) | Systematic review of clinical evidence: literature, equivalent device data, clinical investigation results — demonstrates safety and clinical benefit |
| Labelling and IFU | Device labels and Instructions for Use in all required EU languages — must comply with MDR Annex I Chapter III |
| Post-Market Surveillance (PMS) Plan | Documented system for collecting and reviewing real-world performance data — feeds into PSUR and PMCF |
| SSCP (Class IIb implantable / Class III only) | Plain-language public document summarising safety and clinical performance — validated by Notified Body |
4. Clinical Evaluation
The Clinical Evaluation Report (CER) is one of the most demanding requirements under EU MDR. Unlike the old MDD, where clinical evaluation was sometimes superficial, EU MDR requires a rigorous, systematic review of all available clinical evidence — published literature, clinical investigations, equivalent device data, or a combination — that demonstrates the device's safety, clinical performance, and benefit-risk profile.
For Class IIb and Class III devices, clinical investigations (controlled trials or real-world studies) are frequently required. Relying solely on literature-based evidence is increasingly difficult to defend for high-risk devices. The CER is a living document — it must be updated throughout the device lifecycle as new post-market data becomes available.
5. Declaration of Conformity
The EU Declaration of Conformity (DoC) is the formal legal statement in which the manufacturer declares that the device complies with all applicable EU regulations. It must reference the correct regulations (EU MDR 2017/745 and any other relevant EU directives), the harmonized standards applied, the Notified Body identification number (if applicable), and be signed by the authorized representative of the manufacturer.
6. Post-Market Obligations
CE marking does not end at certification. EU MDR imposes significant ongoing obligations:
| Post-Market Obligation | Required For | Frequency |
| Post-Market Surveillance (PMS) System | All classes | Continuous — actively maintained |
| Periodic Safety Update Report (PSUR) | Class IIa, IIb, III | Class IIa: every 2 years | Class IIb and III: annually |
| Post-Market Clinical Follow-Up (PMCF) | Class IIb, III, and AIMDs | Ongoing — 10-year minimum for AIMDs |
| Vigilance Reporting | All classes | Serious incidents: 15 days | Trend reporting: 30 days |
| Summary of Safety and Clinical Performance (SSCP) | Class IIb implantable + Class III | Annual update and Notified Body validation |
| EUDAMED Updates | All classes | Continuous — device data, certificates, UDIs must be kept current |
Benefits of CE Marking for Medical Devices
Beyond regulatory compliance, CE marking delivers real, tangible value for manufacturers, distributors, healthcare providers, and patients. Here is why obtaining CE marking is one of the most strategically important steps a medical device company can take.
1. Legal Market Access Across 30+ Countries
CE marking provides a single approval pathway that unlocks access to 27 EU member states plus Norway, Iceland, and Liechtenstein — a combined market of over 450 million people. Rather than navigating separate national approval processes in each country, a single CE mark covers the entire EEA. This is arguably the most significant commercial benefit: one process, one mark, one market.
2. Global Regulatory Recognition
CE marking under EU MDR is widely recognized as one of the world's most rigorous medical device approval frameworks. Several non-EU countries — including Switzerland, Turkey, and others negotiating regulatory mutual recognition agreements — accept CE-marked devices through simplified or fast-tracked local approval processes. Building a CE marking-compliant technical file also significantly overlaps with documentation requirements for the FDA, Health Canada, Australia's TGA, and CDSCO, reducing the cost and effort of multi-market submissions.
3. Increased Buyer Confidence and Procurement Advantage
Hospitals, health systems, procurement committees, and distributors across Europe will not consider a device without CE marking. The mark signals that the product has been independently assessed and meets internationally recognised safety standards. For manufacturers competing for NHS, hospital group, or national tender contracts, CE marking is a baseline requirement — not a differentiator.
4. Improved Internal Quality and Risk Management
The process of achieving CE marking forces manufacturers to build rigorous systems — ISO 13485-certified QMS, structured risk management per ISO 14971, comprehensive clinical evidence review. These are not just compliance outputs. They are operational assets that reduce manufacturing defects, improve complaint handling, support faster product iterations, and reduce liability exposure. Manufacturers who take CE marking seriously typically build better products.
5. Competitive Moat for Market Incumbents
CE marking under EU MDR represents a significant barrier to entry, particularly for Class IIb and Class III devices where the certification process requires years, millions in investment, and deep regulatory expertise. Once achieved, a CE certificate and the underlying compliance infrastructure becomes a competitive advantage that is difficult for new entrants to replicate quickly.
6. Protection Against Regulatory Enforcement
Non-CE-marked devices face serious enforcement actions: market withdrawal orders, import bans, financial penalties, and in cases of patient harm, criminal liability. CE marking provides a structured legal defense — demonstrating to regulators that the manufacturer followed due process. Properly maintained CE certification also protects distributors and importers who are downstream in the supply chain.
7. Enhanced Patient Safety Outcomes
The clinical evidence requirements, post-market surveillance obligations, and vigilance reporting systems built into the EU MDR CE marking framework are specifically designed to detect problems early and protect patients. Manufacturers who maintain robust post-market systems identify device issues faster, take corrective action sooner, and build the kind of long-term safety record that sustains market reputation.
8. Access to New Markets Opening in 2026
The EMA's breakthrough device pilot programme launching Q2 2026 gives manufacturers of innovative, high-need devices access to priority regulatory support and scientific advice — a significant benefit for companies developing next-generation diagnostics, AI-powered tools, or novel implantable devices. Additionally, the UK's new framework expected in mid-2026 is set to introduce international reliance routes that will allow EU MDR-certified devices to access the GB market more efficiently.
CE Marking Validity and Renewal: How Long Does Certification Last?
CE marking is not a permanent authorization. Certificates have defined validity periods, ongoing surveillance obligations, and formal renewal processes. Understanding how certificate management works is critical to avoiding forced market withdrawal — a situation that more manufacturers faced than expected during the MDR transition.
How Long Is a CE Certificate Valid?
| Certificate Type | Typical Validity Period | Renewal Required By |
| CE Certificate of Conformity (QMS + Technical Documentation) — Class IIa, IIb, III | 3–5 years (set by Notified Body) | Before expiry — typically initiate 12–18 months prior |
| ISO 13485 QMS Certificate | 3 years (with annual surveillance) | Before expiry — annual surveillance audits maintain validity |
| Class I Self-Certified (Declaration of Conformity) | No fixed expiry — ongoing manufacturer responsibility | Updated whenever device, QMS, or applicable standards change significantly |
| Class III devices (annual PSUR required) | Certificate valid 3–5 years, but annual review by Notified Body | PSUR submission annually; any safety concern can trigger early review |
| Legacy MDD Certificates (transitional) | Expired or expiring per Regulation EU 2024/1860 timelines | Transition to EU MDR required — see MDR transition deadlines table |
What Maintains Certificate Validity Between Renewals?
A CE certificate does not stay valid automatically. Manufacturers must actively maintain validity throughout the certificate period by meeting ongoing Notified Body and regulatory requirements:
- Periodic surveillance audits: Notified Bodies conduct annual or biannual surveillance audits of the QMS and technical documentation. Failing a surveillance audit can lead to certificate suspension.
- Updated technical documentation: Technical documentation must be kept current. New post-market surveillance data, updated clinical evidence, changes in applicable standards, and any design or manufacturing changes must be reflected.
- Active PMS system: Post-market surveillance activities must be ongoing — not dormant until the next audit. Notified Bodies expect to see real data being collected, reviewed, and acted upon.
- Change notifications: Significant changes to the device design, intended use, manufacturing processes, or QMS elements must be notified to the Notified Body before implementation. Unreported significant changes can invalidate a certificate.
- EUDAMED currency: Device registrations, UDI data, certificate information, and economic operator details in EUDAMED must be kept accurate and up to date. Outdated EUDAMED entries trigger regulatory queries.
- PSUR submission (Class IIa+): Periodic Safety Update Reports must be submitted on schedule. Missing PSUR submissions are a common cause of Notified Body queries and formal warnings.
When Can a Certificate Be Suspended or Withdrawn?
Notified Bodies and national competent authorities have the power to suspend, restrict, or withdraw CE certificates. The most common triggers are:
- Serious patient safety incidents not reported in the required timeframe
- Failure to implement corrective actions following a surveillance audit finding
- Significant unreported changes to the device or manufacturing process
- Failure to maintain QMS compliance — lapsed ISO 13485, unresolved non-conformances
- Fraudulent or materially incorrect technical documentation discovered post-certification
- Failure to submit required post-market reports (PSUR, vigilance reports, PMCF data)
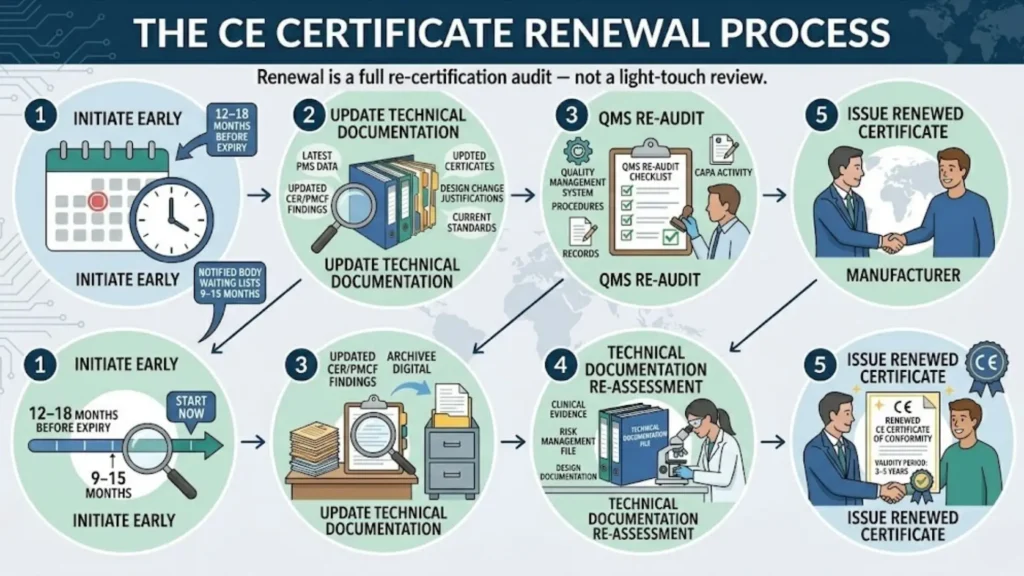
The CE Certificate Renewal Process
Renewal of a CE certificate requires a full re-certification audit — not a light-touch review. This involves:
- Step 1 — Initiate early: Start the renewal process 12–18 months before your certificate expires. Given Notified Body waiting lists of 9–15 months in 2026, waiting until 6 months before expiry creates serious risk.
- Step 2 — Update technical documentation: Refresh your technical documentation with the latest post-market surveillance data, updated CER/PMCF findings, any design change justifications, and current applicable standards.
- Step 3 — QMS re-audit: The Notified Body will conduct a full QMS re-audit, reviewing your quality management system, procedures, records, and any CAPA activity since the last audit.
- Step 4 — Technical documentation re-assessment: For Class IIb and Class III devices, the Notified Body will conduct a detailed re-assessment of the clinical evidence, risk management file, and design documentation.
- Step 5 — Issue renewed certificate: Upon successful completion, the Notified Body issues a renewed CE Certificate of Conformity with a new validity period, typically 3–5 years.

MDR Transition Deadlines Still Active in 2026–2028
| Device Category | MDR Transition Deadline | Status in April 2026 |
| Class III Custom-Made Implantable Devices | May 26, 2026 | PASSED — Full MDR compliance required NOW |
| Class III and Class IIb Implantable Devices | December 31, 2027 | Active — transition must be well underway |
| Class IIb (non-implantable), IIa, Class I Sterile/Measuring | December 31, 2028 | Active — MDR transition planning required |
| IVDR Class D (HIV, blood grouping, hepatitis) | In force now | PASSED — Full IVDR compliance required NOW |
| IVDR Class C | December 31, 2027 | Active |
| IVDR Class B and A (relabelled) | December 31, 2028 | Active |
Key 2026 Regulatory Updates That Affect CE Marking
Staying current with regulatory changes is part of your ongoing compliance obligation under EU MDR. Here are the most important developments affecting CE marking for medical devices as of April 2026:
| EUDAMED Full Enforcement | All mandatory EUDAMED functionalities are now operational and actively enforced. Non-registration of economic operators, devices, or UDIs now triggers regulatory action from EU competent authorities. There is no grace period remaining. |
| Legacy MDD Certificates Expired (Class III Custom-Made) | The May 26, 2026 deadline for Class III custom-made implantable devices has passed. Any device in this category still operating under MDD certification faces forced withdrawal. |
| EU AI Act Obligations Phase-In | AI literacy requirements under EU AI Act Article 4 apply from August 2, 2026. CE-marked SaMD with AI components subject to Notified Body assessment have until August 2, 2027 for full Article 6(1) compliance. |
| EMA Breakthrough Device Pilot | Q2 2026 launch of the EMA pilot programme giving manufacturers of breakthrough devices access to priority scientific advice and enhanced regulatory support from EU expert panels. |
| MDCG 2025-6 Guidance Published | New guidance clarifying the interplay between EU MDR and the EU AI Act for SaMD. Notified Bodies are expected to assess AI Act compliance as part of MDR QMS review for relevant devices. |
| UK New Framework Approaching | Key parts of the UK's new medical device legislation are expected to apply from mid-2026, introducing MDR-aligned classification rules and international reliance routes based on EU MDR, FDA, and Health Canada approvals. |
| Regulation EU 2024/1860 in Effect | Extended transitional provisions confirmed for legacy MDD-certified Class III and Class IIb devices to December 2027, and Class IIb non-implantable, IIa and Class I sterile to December 2028 — provided manufacturers meet conditions including having an active MDR transition plan with a designated Notified Body. |
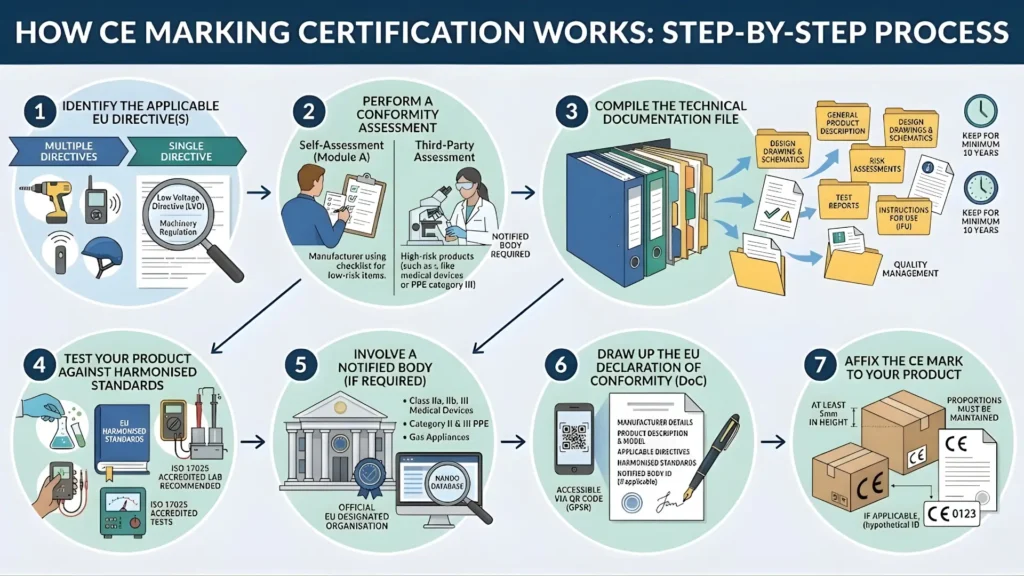
CE Marking Process for Medical Devices: Step-by-Step Overview
For quick reference, here is the end-to-end CE marking process summarised by stage. Each stage feeds into the next — shortcuts in early stages consistently create expensive problems later.
| Stage | Action | Output / Milestone |
| 1 | Confirm device qualifies as medical device under EU MDR definition | Device scope confirmed; regulatory strategy initiated |
| 2 | Classify device per EU MDR Annex VIII rules | Classification rationale documented; conformity route determined |
| 3 | Identify applicable GSPRs and harmonised standards | GSPR checklist and standards matrix completed |
| 4 | Implement ISO 13485-aligned QMS; appoint PRRC | QMS operational; internal audit completed; PRRC appointed |
| 5 | Appoint EU Authorised Representative (non-EEA manufacturers) | Signed EU AR mandate; EUDAMED registration of EU AR |
| 6 | Register as economic operator in EUDAMED; obtain SRN | SRN issued; economic operator entry live in EUDAMED |
| 7 | Prepare Technical Documentation (Annex II & III) | Complete technical file: CER, risk file, IFU, design dossier, PMS plan |
| 8 | Engage Notified Body and undergo QMS + Technical Documentation audit (Class IIa+) | Notified Body audit passed; CE Certificate of Conformity issued |
| 9 | Sign EU Declaration of Conformity | Legally binding DoC issued and filed |
| 10 | Affix CE mark; register device and UDI in EUDAMED | Device on market; EUDAMED entry live; UDI marked on label |
| 11 | Maintain post-market obligations: PMS, PSUR, PMCF, vigilance | Ongoing compliance maintained; certificate validity preserved |

How Much Does CE Marking Cost for Medical Devices?
Cost is one of the first questions every manufacturer asks. The honest answer is that it varies significantly based on device class, documentation readiness, Notified Body fees, and whether clinical investigations are required. Here is a realistic budget framework:
| Cost Component | Class I (Self-Certified) | Class IIa | Class IIb / Class III |
| QMS Implementation (ISO 13485) | £15,000–£40,000 | £20,000–£60,000 | £40,000–£120,000 |
| Technical Documentation Preparation | £10,000–£30,000 | £30,000–£80,000 | £60,000–£200,000+ |
| Clinical Evaluation Report (CER) | £5,000–£15,000 | £20,000–£60,000 | £50,000–£200,000+ |
| Clinical Investigation (if required) | Not typically required | Rarely required | £200,000–£2,000,000+ |
| Notified Body Fees | Not applicable | £30,000–£80,000 | £60,000–£200,000+ |
| EUDAMED Registration and UDI | £1,000–£3,000 | £2,000–£5,000 | £3,000–£8,000 |
| Regulatory Consultancy | £5,000–£20,000 | £15,000–£50,000 | £30,000–£100,000+ |
| Approximate Total Range | £36,000–£108,000 | £117,000–£335,000 | £243,000–£828,000+ |
These are indicative ranges only. High-risk Class III devices requiring clinical investigations can exceed £3–5 million in total regulatory investment for novel implantables. Starting with complete, well-prepared documentation is consistently the most cost-effective approach — Notified Body queries, resubmissions, and timeline overruns add up quickly.
Conclusion: CE Marking for Medical Devices in 2026
CE marking for medical devices is one of the most rigorous and consequential regulatory processes in global healthcare. In 2026, the framework under EU MDR 2017/745 is fully operational, EUDAMED is actively enforced, critical transition deadlines have passed, and new obligations under the EU AI Act are coming into force. The environment rewards manufacturers who prepared thoroughly and penalizes those who delayed.
The benefits of achieving CE marking are real and significant — legal market access across 30+ countries, global regulatory recognition, buyer confidence, competitive protection, and an internal quality framework that makes better products. But those benefits only materialize when the process is approached with genuine commitment to documentation quality, clinical evidence rigour, and post-market compliance.
Whether you are starting a first CE marking application, managing an MDR transition for legacy devices, renewing an existing certificate, or navigating the intersection of MDR and the EU AI Act for SaMD, the fundamentals remain the same: classify correctly, document thoroughly, choose your Notified Body carefully, and build post-market systems that actually work.
Frequently Asked Questions
What is CE marking for medical devices in simple terms?
CE marking is the mandatory regulatory approval required to legally sell a medical device in the EU and EEA. It confirms that the manufacturer has assessed the device and demonstrated it meets all EU safety, performance, and labelling requirements under EU MDR 2017/745. Without it, the device cannot be marketed in Europe.
Is CE marking the same as FDA approval?
No. CE marking covers the EU/EEA market under EU MDR 2017/745. FDA 510(k) clearance or PMA approval covers the US market under FDA regulations. They are separate systems with different requirements. However, documentation prepared for EU MDR commonly overlaps with FDA submissions, reducing duplication for manufacturers targeting both markets
How long does CE marking take for a medical device?
Class I self-certified devices: 3–6 months. Class IIa: 12–18 months. Class IIb: 18–24 months. Class III: 24–36 months or more. In 2026, Notified Body waiting lists of 9–15 months are adding to all Class II and III timelines. Starting early and submitting complete documentation makes the biggest difference.
Does a manufacturer outside Europe need CE marking?
Yes. CE marking is required regardless of where the manufacturer is based. A company in India, the US, China, or anywhere else must achieve CE marking to sell in the EU. Non-EEA manufacturers must also appoint an EU Authorized Representative who acts as the official EU contact and shares legal liability for compliance.
How long is a CE certificate valid?
CE certificates issued by Notified Bodies are typically valid for 3–5 years, subject to annual surveillance audits. If surveillance audits are not passed or ongoing obligations (PSUR, PMS, vigilance) are not met, certificates can be suspended early. Class I self-certified devices have no fixed expiry but must be kept current as standards and regulations evolve.
How do I renew a CE certificate?
Start the renewal process 12–18 months before expiry. The Notified Body will conduct a full re-audit of your QMS and updated technical documentation. Update your CER with the latest post-market data, refresh the risk management file, and submit a complete renewal dossier. If the certificate expires before renewal is completed, the device must be withdrawn from the EU market.
What happens if I sell a device without CE marking in the EU?
Selling a device without CE marking in the EU is illegal under EU MDR. Consequences include: market withdrawal orders by national competent authorities, import bans, financial penalties, reputational damage, and in cases involving patient harm, potential criminal liability for the legal manufacturer and EU Authorised Representative. EUDAMED now enables authorities to cross-reference registered and unregistered devices more efficiently.
Does CE marking cover the UK market after Brexit?
Partially, and temporarily. EU MDR-compliant CE-marked devices can be placed on the Great Britain (GB) market until June 30, 2030 under transitional arrangements. Northern Ireland follows EU MDR under the Windsor Framework and requires CE marking permanently. Manufacturers selling in both GB and NI need a dual compliance strategy. A new UK regulatory framework aligning closer to MDR is expected to apply from mid-2026.
Do software and AI-powered medical devices need CE marking?
Yes. Software that performs a medical purpose for individual patients qualifies as Software as a Medical Device (SaMD) under EU MDR and requires CE marking. AI-powered SaMD must also comply with the EU AI Act — AI literacy obligations apply from August 2026, and full high-risk AI compliance for CE-marked devices under Notified Body assessment is required by August 2027.
What is EUDAMED and why does it matter for CE marking?
EUDAMED is the EU's central database for medical devices, now fully operational and enforced in 2026. All manufacturers, importers, and authorised representatives must register as economic operators. All devices must be registered with UDIs. Certificates, clinical investigations, and vigilance reports are also tracked. Non-registration triggers regulatory action. EUDAMED registration is a mandatory prerequisite before a device can be placed on the EU market.





































