- CE marking certification is a legal requirement to sell products across 30 European Economic Area countries.
- Covers 26+ EU directives — from electronics, machinery, and toys to medical devices and PPE.
- 2026 brings major enforcement changes under GPSR, ESPR, and the new Machinery Regulation EU 2023/1230.
What Is CE Marking Certification?
CE marking certification is the mandatory conformity marking for products sold within the European Economic Area (EEA). The initials 'CE' stand for Conformité Européenne — French for 'European Conformity.' Affixing the CE mark to a product is the manufacturer's declaration that the product meets all applicable EU health, safety, and environmental requirements before it is placed on the market.


The CE mark is not a quality label, a trademark, or a stamp of approval from any government authority. It is a legal compliance marker — one that gives customs officials, market surveillance authorities, retailers, and consumers the assurance that the product has been assessed against the relevant EU directives and found to be compliant.
In 2026, this matters even more. The EU has rolled out the General Product Safety Regulation (GPSR), which became fully enforceable on 13 December 2024 and is now actively enforced across all member states. The Ecodesign for Sustainable Products Regulation (ESPR) is adding new sustainability-linked conformity obligations for several product categories starting in 2025–2026.
| Key Insight for 2026 CE marking certification is not just a box-ticking exercise. It is a dynamic, ongoing compliance obligation. Regulations evolve, harmonized standards are revised, and your product portfolio will change over time — your CE certification programme must keep pace with all of these. |
Who Needs CE Marking Certification?
You need CE marking certification if your product falls under one or more EU directives or regulations. This covers an enormous range of industries and product types:
- Electrical and electronic equipment (Low Voltage Directive — LVD)
- Machinery and industrial equipment (Machinery Directive 2006/42/EC, being updated to Machinery Regulation EU 2023/1230)
- Personal protective equipment (PPE Regulation)
- Medical devices (MDR 2017/745 and IVDR 2017/746)
- Radio and telecommunications equipment (RED Directive)
- Construction products (CPR)
- Toys and children's products (Toys Safety Directive)
- Pressure vessels and equipment
- Lifts and elevators
- Measuring instruments
Benefits of CE Marking Certification
CE marking certification is often discussed purely in terms of legal obligation — but the business benefits go far beyond simply staying on the right side of EU law. Here is a full breakdown of why CE marking delivers genuine competitive and commercial value.
- Access to 450 Million+ Consumers - The EEA includes 30 countries. CE marking is your single licence to sell in all of them without separate national approvals. No other regional conformity programme offers this scale.
- Competitive Advantage in Tenders - Public procurement in EU member states frequently mandates CE marking as a baseline requirement. Without it, you are disqualified from government and institutional contracts.
- ESG and Sustainability Alignment - Under the new ESPR (2024/1781), CE-compliant products increasingly need to demonstrate sustainability. This aligns with ESG investor expectations and corporate sustainability reporting obligations.
- E-commerce and Online Marketplace Access - Amazon EU, Zalando, Cdiscount, and other major EU platforms now verify CE compliance for regulated product categories. Non-compliant products are delisted under the GPSR.
- Single Market Entry — One Mark - Unlike the USA (UL/FCC), Canada (CSA), or Japan (PSE), the EU single market recognises one mark across all 30 countries. CE marking eliminates country-by-country approval costs.
- Increased Buyer and Retailer Confidence - Major EU retailers, distributors, and B2B buyers will not stock or procure products that lack CE marking. Certification immediately expands your distribution and procurement opportunities.
- Global Market Credibility - CE marking is internationally recognised as a rigorous safety benchmark. Many markets outside the EU — including Gulf Cooperation Council (GCC) countries — use CE compliance as a pre-qualification criterion.
- Reduced Trade Friction - CE-marked products pass through EU customs more smoothly, face fewer spot checks, and are less likely to be subject to corrective market surveillance actions.
- Brand Reputation and Consumer Trust - For end consumers, the CE mark is a recognisable symbol of safety compliance. In competitive categories like consumer electronics, toys, and baby products, it is a purchase-influencing factor.
CE Marking Eligibility Criteria — Who Qualifies and Who Must Apply?
A common misconception is that CE marking eligibility is something you apply for. In reality, CE marking is a legal obligation — not an approval to be granted. The correct question is not 'Am I eligible for CE marking?' but 'Is my product required to carry the CE mark, and am I the responsible party?'
Products That Are Covered
CE marking applies to any product that falls within the scope of one or more EU New Approach directives or regulations. The key regulated product categories are:
| Product Category | Primary Directive / Regulation | Risk Level |
| Electrical equipment (50–1000V AC) | Low Voltage Directive 2014/35/EU | Medium |
| Wireless / radio devices | Radio Equipment Directive 2014/53/EU | Medium–High |
| Machinery and industrial tools | Machinery Regulation EU 2023/1230 | High |
| Medical devices (Class I–III) | MDR 2017/745 / IVDR 2017/746 | Very High |
| Personal protective equipment | PPE Regulation (EU) 2016/425 | High |
| Children's toys | Toys Safety Directive 2009/48/EC | High |
| Construction products | Construction Products Regulation 305/2011 | Medium |
| Gas appliances | Gas Appliances Regulation (EU) 2016/426 | High |
| Pressure equipment | Pressure Equipment Directive 2014/68/EU | High |
| Lifts and elevators | Lifts Directive 2014/33/EU | High |
| Measuring instruments | Measuring Instruments Directive 2014/32/EU | Medium |
| Explosives for civil use | Explosives Directive 2014/28/EU | Very High |
| All general consumer products | GPSR (EU) 2023/988 — applicable from Dec 2024 | Variable |
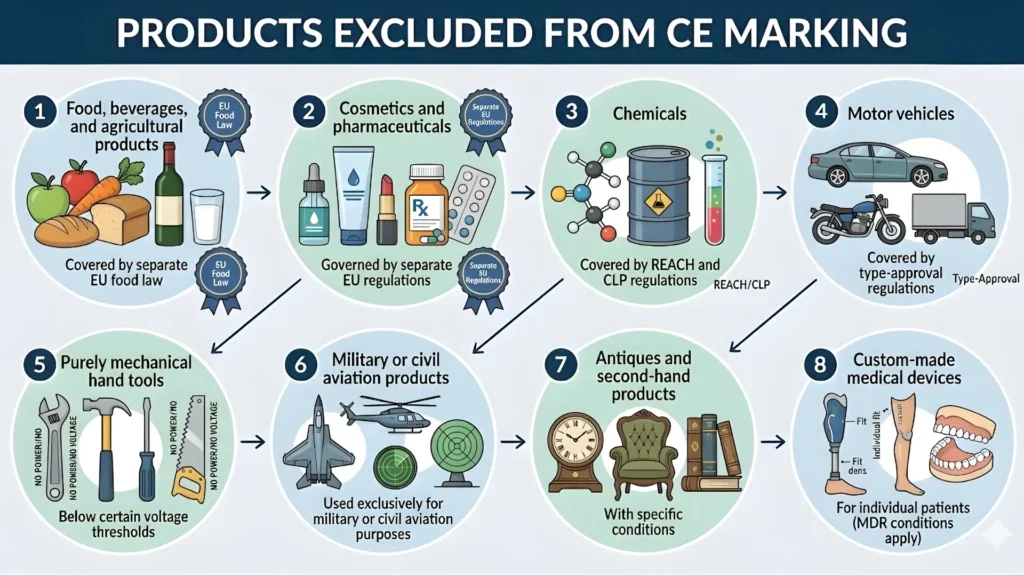
Products That Are Excluded from CE Marking
Not all products require CE marking. The following categories are typically excluded:
- Food, beverages, and agricultural products (covered by separate EU food law)
- Cosmetics and pharmaceuticals (governed by separate EU regulations)
- Chemicals (covered by REACH and CLP regulations)
- Motor vehicles (covered by type-approval regulations)
- Purely mechanical hand tools below certain voltage thresholds
- Products used exclusively for military or civil aviation purposes
- Antiques and second-hand products (with conditions)
- Custom-made medical devices for individual patients (with conditions under MDR)

Who Is the Responsible Economic Operator?
CE marking responsibility depends on your role in the supply chain. The EU distinguishes between several economic operators, each with defined obligations under GPSR and applicable directives:
| Economic Operator | CE Marking Responsibility | Key Obligation |
| Manufacturer | Full responsibility | Compile technical file, sign DoC, affix CE mark |
| EU Authorised Representative | Acts on behalf of non-EU manufacturer | Hold technical file, cooperate with authorities |
| Importer (EU-based) | Shared responsibility | Verify manufacturer's compliance before import |
| Distributor (EU-based) | Duty of verification | Check CE mark and DoC are present and correct |
| Fulfilment Service Provider | New under GPSR 2023/988 | Traceability and storage obligations |
| Online Marketplace | New under GPSR 2023/988 | Remove non-compliant listings, cooperate with authorities |
CE Marking Requirements: What You Must Have in Place
CE marking requirements are set by the applicable EU directive(s) for your product. However, there is a common framework of requirements that applies across virtually all CE marking programmes. Understanding these core requirements will help you structure your compliance approach efficiently.
1. Product Conformity with Essential Requirements
Every EU directive defines a set of Essential Requirements — the mandatory health, safety, environmental, or performance characteristics your product must meet.
| Directive | Essential Requirements (Examples) |
| LVD 2014/35/EU | Protection against electric shock, insulation, overheating, short circuit |
| RED 2014/53/EU | Radio spectrum efficiency, EMC performance, cybersecurity (from Aug 2025) |
| Machinery Reg. 2023/1230 | Mechanical integrity, control system safety, emergency stop, ergonomics |
| Toys 2009/48/EC | Mechanical safety, flammability, chemical safety, electrical safety |
| MDR 2017/745 | Clinical performance, biocompatibility, sterility, labelling accuracy |
| PPE 2016/425 | Protection effectiveness, comfort, ergonomics, durability |
2. Conformity Assessment Procedure
You must select and carry out the correct conformity assessment module — the formal procedure for evaluating your product's compliance. Modules range from internal self-assessment to full third-party certification by a notified body.
| Module | Description | Notified Body Required? | Typical Products |
| Module A | Internal production control — self-assessment | No | Lower-risk general products, Class I medical devices |
| Module B+C | EU type-examination + production conformity | Yes | Category II PPE, certain gas appliances |
| Module D | Quality assurance of production process | Yes | High-risk machinery, pressure equipment |
| Module E | Quality assurance of product testing | Yes | Series-produced high-risk products |
| Module F | Product verification by individual test | Yes | One-off or small-batch high-risk products |
| Module G | Unit-by-unit verification | Yes | Bespoke high-risk equipment |
| Module H | Full quality assurance | Yes | Class III medical devices, complex machinery |
3. Technical Documentation File
You must compile and maintain a technical documentation file (also called a Technical Construction File or TCF) that provides full evidence of your product's compliance. This file is your legal proof — and you must produce it within 10 days if requested by a market surveillance authority.
A complete technical file must include:
- Full product description, model designations, and intended use
- Design drawings, component schematics, and circuit diagrams
- List of applicable EU harmonized standards and normative references
- Risk assessment documentation (methodology, identified hazards, mitigation measures)
- Test reports from internal or accredited third-party laboratories
- Calculations, engineering analyses, and material specifications
- Instructions for use (IFU) in all official EU languages of the target markets
- Safety warnings, labelling artwork, and packaging information
- Quality management system documentation (where applicable)
- Notified body certificate(s) and assessment reports (where applicable)
4. EU Declaration of Conformity (DoC)
The EU Declaration of Conformity is the manufacturer's formal, signed declaration that the product meets all applicable EU requirements. It is a legally binding document. Under GPSR 2023/988, consumers and authorities must be able to access the DoC easily — ideally via a QR code or URL link on the product or packaging.
The DoC must include: manufacturer's identity, product description, applicable directives, harmonized standards used, notified body details (if applicable), date, place, and authorized signature.
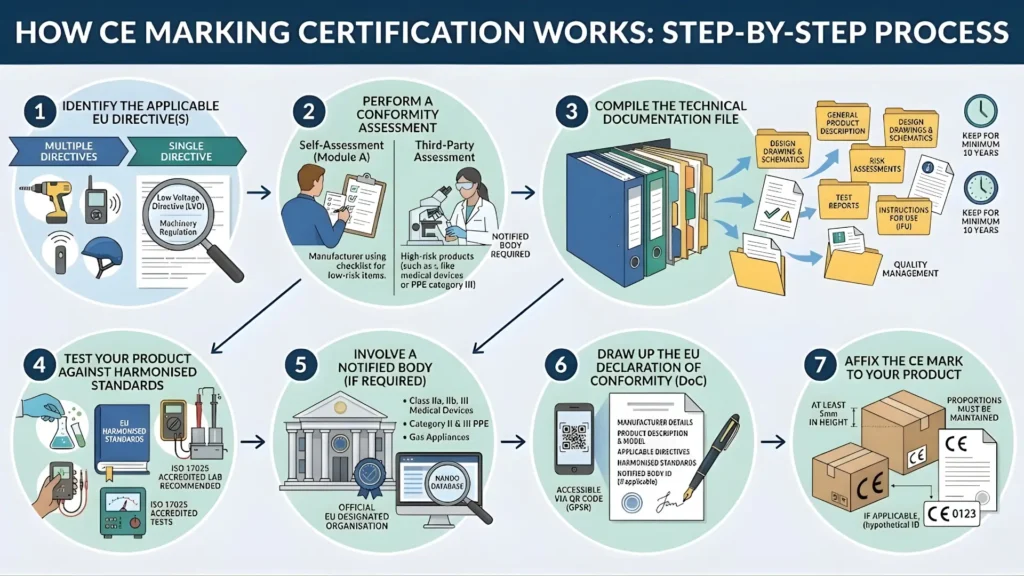
How CE Marking Certification Works: Step-by-Step
Step 1 — Identify the Applicable EU Directive(s)
The very first step is to identify which EU directive or regulation applies to your product. Some products fall under a single directive; others fall under multiple directives simultaneously. For example, an electric power tool must comply with both the Low Voltage Directive and the Machinery Directive.
Key EU Directives and Regulations
| Directive / Regulation | Product Category | Key Requirement |
| Low Voltage Directive (LVD) 2014/35/EU | Electrical equipment (50–1000V AC) | Safety testing, technical file |
| Machinery Regulation EU 2023/1230 | Machines, tools, industrial equipment | Risk assessment, technical file |
| Radio Equipment Directive (RED) 2014/53/EU | Wireless devices, radio equipment | Radio spectrum, EMC testing |
| Medical Device Regulation (MDR) 2017/745 | Medical devices Class I–III | Clinical evaluation, notified body |
| PPE Regulation (EU) 2016/425 | Helmets, gloves, protective clothing | Category-based testing |
| Toys Safety Directive 2009/48/EC | Children's toys | Chemical, mechanical, electrical tests |
| GPSR (EU) 2023/988 | All consumer products | Safety risk assessment, traceability |
Step 2 — Perform a Conformity Assessment
After identifying the relevant directive(s), you must conduct a conformity assessment — evaluating whether your product meets all the requirements of the applicable directive.
- Self-assessment (Module A) — You conduct the assessment internally. Applies to lower-risk products.
- Third-party assessment — Required for higher-risk products such as Class II/III medical devices, category III PPE, and certain gas appliances.
Step 3 — Compile the Technical Documentation File
Every manufacturer applying for CE marking certification must compile a technical file — your legal evidence that the product meets all required standards. A complete technical file typically includes:
- General product description and intended use
- Design drawings, schematics, and component specifications
- List of applicable EU harmonised standards
- Results of risk assessments
- Test reports (internal or from accredited labs)
- Calculations and engineering analyses
- Instructions for use (IFU) and safety labelling
- Quality management documentation (if applicable)
You are legally required to keep the technical file for a minimum of 10 years after the product has been placed on the market.
Step 4 — Test Your Product Against Harmonized Standards
The EU publishes lists of harmonized standards in the Official Journal of the European Union. Products that comply with these standards are presumed to conform with the essential requirements of the applicable directive. You can test your product in-house (with calibrated equipment) or through an ISO 17025-accredited test laboratory — the latter is strongly recommended for regulated products.
Step 5 — Involve a Notified Body (If Required)
A notified body is an organisation officially designated by an EU member state to carry out conformity assessment procedures. You must involve a notified body for high-risk products:
- Class IIa, IIb, and III medical devices
- Category II and III personal protective equipment
- Gas appliances under Regulation (EU) 2016/426
- Certain pressure equipment
- Lifts under the Lifts Directive
The European Commission maintains the NANDO database (New Approach Notified and Designated Organisations) at ec.europa.eu/growth/tools-databases/nando. Always verify your chosen notified body is listed there.
| 💡 Tip: Always confirm the notified body is designated for the specific directive AND product scope you need — not just for the directive in general. |
Step 6 — Draw Up the EU Declaration of Conformity (DoC)
The EU Declaration of Conformity (DoC) is a legally binding document in which the manufacturer formally declares that the product meets all applicable EU requirements. It must include:
- Manufacturer's name and address
- EU authorised representative's name and address (if applicable)
- Product description, model, or type number
- Specific directives and regulations being declared
- List of harmonised standards or other technical specifications applied
- Name and address of the notified body plus certificate reference numbers (if applicable)
- Date and place of declaration, and signature of the responsible person
Under the new GPSR (2023/988), the DoC must be easily accessible — ideally through a QR code or direct URL printed on the product packaging.
Step 7 — Affix the CE Mark to Your Product
Once all steps are complete and you are satisfied that your product complies, you can affix the CE mark. Key rules:
- The CE mark must be at least 5mm in height (proportions must be maintained if scaled)
- Placed on the product itself, its packaging, or accompanying documents — wherever most practical
- Must not be obscured, partly hidden, or hard to identify
- If a notified body was involved in production control, their four-digit ID number must appear after the CE mark (e.g., CE 0123)

CE Marking Certification Costs: What Should You Budget in 2026?
One of the most common questions businesses ask is: 'How much does CE marking certification cost?' The answer depends on your product type, risk category, and whether a notified body is required.
| Cost Component | Estimated Range (EUR) |
| Initial regulatory gap analysis | €500 – €2,500 |
| Technical documentation preparation | €1,000 – €8,000 |
| Product testing (accredited lab) | €500 – €15,000+ |
| Notified body assessment (if required) | €3,000 – €30,000+ |
| CE marking consultant (optional) | €1,500 – €10,000 |
| EU authorised representative (per year) | €300 – €2,000 |
| Total — low-risk product, self-declaration | €1,500 – €5,000 |
| Total — high-risk product, notified body | €10,000 – €60,000+ |
CE Marking Validity, Maintenance and Renewal
One of the most misunderstood aspects of CE marking certification is the question of validity and renewal. Unlike some certifications — ISO 9001, for example — CE marking does not have a fixed expiry date that triggers a formal renewal process. However, that does not mean your CE certification is permanent or maintenance-free.
How Long Is CE Marking Valid?
CE marking is valid for as long as:
- The product remains unchanged from the version that was assessed and documented
- The applicable EU directive(s) and harmonized standards remain current and unchanged
- No new applicable directive or regulation has entered into force that covers your product
- Your notified body certificate (if applicable) has not expired or been withdrawn
- Your EU Authorized Representative remains appointed and legally compliant (for non-EU manufacturers)
In practice, this means CE marking can remain valid for many years for a stable, unchanged product in a stable regulatory environment. However, the rapid pace of EU regulatory change in 2025–2026 means that almost all businesses will need to review and update their CE certifications more frequently than in previous years.
When Must You Re-Evaluate Your CE Certification?
You must review and potentially update your CE marking certification whenever any of the following trigger events occur:
| Trigger Event | Action Required | Timeline |
| Product design change (hardware, software, materials) | Re-assess affected areas of compliance; update technical file and DoC if needed | Before modified product is placed on market |
| New or revised applicable directive enters into force | Full re-assessment against new directive requirements; new DoC required | By the directive's date of application |
| Harmonised standard revised or withdrawn | Re-test against updated standard; update technical file references | Before previous standard's withdrawal deadline |
| Notified body certificate expiry | Re-apply to notified body for renewed assessment/certificate | Typically 3–5 years (check your certificate) |
| Market surveillance authority requests compliance action | Immediate review, corrective action, and updated documentation | As directed by authorities — often very tight deadlines |
| GPSR 2023/988 obligation changes | Review traceability, DoC accessibility, and economic operator obligations | Ongoing — active enforcement since Dec 2024 |
| ESPR delegated act for your product category | Assess new ecodesign requirements; update technical file and test programme | Per the timeline in the specific delegated act |
| EU AR relationship ends | Appoint new EU AR before previous one's term expires; update DoC | Before expiry — no gap in EU AR coverage permitted |
Technical File Retention Requirements
Regardless of whether your CE marking remains valid or is updated, you are legally required to retain your technical documentation for defined periods:
| Directive / Regulation | Technical File Retention Period |
| Most CE directives (general) | 10 years from the date the product was last placed on the market |
| Medical Devices (MDR 2017/745) | Minimum 10 years; 15 years for implantable devices |
| Construction Products (CPR) | 10 years from the date the product was last placed on the market |
| Machinery Regulation 2023/1230 | 10 years from the date the product was last placed on the market |
Notified Body Certificate Renewal
If a notified body issued a certificate for your product (EU type-examination certificate, quality system approval, etc.), that certificate will have a defined period of validity — typically 3 to 5 years. Before it expires, you must:
- Contact your notified body well in advance of the expiry date (typically 6–12 months before)
- Submit updated technical documentation reflecting any product or standard changes since the original assessment
- Undergo any required re-testing, re-assessment, or audit as determined by the notified body
- Obtain and file the renewed certificate before the original expires — a lapse in certificate validity means your product must be withdrawn from the market
| 2026 Renewal Priority Alert If your CE marking certification was set up before 2023, review it urgently against the following changes: (1) GPSR 2023/988 — new DoC accessibility requirements and traceability obligations are now legally enforceable. (2) RED Cybersecurity Delegated Regulation — applicable August 2025 for all internet-connected radio equipment. (3) ESPR Delegated Acts — first product categories entering ecodesign compliance obligations in 2025–2026. (4) Machinery Regulation 2023/1230 — applicable January 2027; preparation and technical file updates should begin now. |
| Read More :- Medical Device Registration |
2026 CE Marking Regulation Updates: What Has Changed?
If your last review of CE marking requirements was more than 12 months ago, you need to update your knowledge. Several significant regulatory changes are now in force.
1. General Product Safety Regulation (GPSR) — EU 2023/988
The GPSR replaced the old General Product Safety Directive and became applicable on 13 December 2024. Key changes:
- Online marketplaces are now co-responsible for product safety — directly impacting Amazon EU, eBay, and similar platforms
- Economic operators (manufacturers, importers, distributors, fulfilment providers) all have clearly defined duties
- Traceability requirements are stronger — products must carry identification information traceable throughout the supply chain
- Digital DoC access — a QR code or URL linking to the Declaration of Conformity is expected for consumer products
2. New Machinery Regulation — EU 2023/1230
The Machinery Directive 2006/42/EC is being replaced by the Machinery Regulation (EU) 2023/1230, which becomes fully applicable on 14 January 2027. Start preparing now.
- New provisions for software-driven and AI-integrated machinery
- Stronger cybersecurity and digital safety requirements
- Expanded essential health and safety requirements (EHSRs)
- New risk assessment requirements for collaborative robots (cobots)
3. Ecodesign for Sustainable Products Regulation (ESPR) — EU 2024/1781
The ESPR extends ecodesign obligations dramatically. From 2025 onwards, CE marking certification will increasingly need to demonstrate compliance with:
- Repairability and durability requirements
- Material efficiency and recyclability standards
- Digital product passport obligations (phased in from 2026–2030)
4. Updated RED Cybersecurity Requirements
The Radio Equipment Directive has new cybersecurity requirements under Commission Delegated Regulation (EU) 2022/30, fully applicable since August 2025. All internet-connected radio equipment — smart devices, wearables, IoT products — must now comply with cybersecurity, privacy protection, and fraud prevention requirements.
CE Marking vs. UKCA Marking: What You Need in 2026
Since Brexit, manufacturers selling in both Europe and the United Kingdom must understand the distinction between CE marking (for the EU/EEA market) and UKCA marking (for Great Britain — England, Scotland, and Wales).
| Aspect | CE Marking | UKCA Marking |
| Coverage | EU 27 member states + Iceland, Norway, Liechtenstein (EEA) | Great Britain only (England, Scotland, Wales) |
| Northern Ireland | Accepted under Windsor Framework | UKNI marking required for some products |
| Governing body | European Commission / EU member states | Office for Product Safety and Standards (OPSS), UK Government |
| Applicable from | Varies by directive — most products since 1993–2003 | Mandatory from 1 January 2025 (extensions applied repeatedly) |
| Current UK position | CE marking accepted in GB until 31 Dec 2027 (most products) | UKCA marking required from 2028 for most regulated products |
| Technical requirements | EU harmonised standards and directives | UK-designated standards (mirrors EU standards in most areas) |
| Notified body | EU Notified Body (listed in NANDO) | UK Approved Body (listed on UKAS / OPSS database) |
| DoC requirement | EU Declaration of Conformity | UK Declaration of Conformity (same structure, different references) |
Common CE Marking Certification Mistakes to Avoid
Many businesses — especially SMEs and exporters from outside the EU — make avoidable mistakes that can invalidate certification or lead to market withdrawal.
- Applying the CE mark without completing the full conformity assessment — the CE mark is not a self-service label; affixing it without required documentation is illegal
- Ignoring multiple applicable directives — missing even one can make your entire CE certification invalid
- Using outdated harmonised standards — always check the Official Journal of the EU for the most current list
- Poor or incomplete technical documentation — market surveillance authorities regularly request technical files
- No EU authorised representative — non-EU manufacturers are legally required to appoint an EU AR in most regulated product categories
- Confusing CE mark with a quality mark — CE marking is a regulatory compliance mark, not a quality endorsement
- Failing to update certification after product changes — even minor modifications may require re-evaluation and updated DoC
CE Marking for Non-EU Manufacturers: The Authorized Representative Requirement
If your manufacturing operations are based outside the EU — whether in India, China, the USA, South Korea, or anywhere else — you must appoint an EU-based Authorised Representative (EU AR) before placing products on the EU market under most CE marking directives and the GPSR.
The EU AR is legally responsible for your product's compliance in the EU, must be named in your Declaration of Conformity, maintains your technical documentation in the EU, and cooperates with market surveillance authorities on your behalf.
Many specialist CE marking firms in Germany, the Netherlands, and Ireland offer EU AR services for a recurring annual fee, typically ranging from €300 to €2,000 per product line.
Conclusion: Is CE Marking Certification Worth It?
The answer is an unqualified yes — if you want access to the world's largest single market and want to protect your business from legal risk.
CE marking certification is an investment, not just a cost. It opens doors to 30 European countries, signals credibility to buyers and distributors, and protects your brand from the catastrophic reputational damage of a market surveillance failure or recall.
In 2026, with the GPSR now in active enforcement, the Machinery Regulation transition underway, and ESPR adding sustainability obligations, CE compliance is becoming both more complex and more closely monitored than ever before. The businesses that get ahead of these changes — by understanding the CE marking certification process thoroughly and working with the right partners — will gain a genuine competitive edge in the European market.
Whether you are a startup preparing your first product launch in Europe, an established manufacturer updating your compliance programme, or a non-EU exporter navigating EU regulations for the first time, the time to start your CE marking certification journey is right now.
Frequently Asked Questions
What is CE marking certification?
CE marking certification is the process by which a manufacturer confirms that a product meets all applicable EU health, safety, and environmental requirements. The CE mark is then affixed to the product as legal proof of compliance, allowing it to be sold across the 30 EEA countries without further national approvals.
Is CE marking mandatory for all products sold in the EU?
No — CE marking is mandatory only for products that fall within the scope of one or more EU CE directives or regulations. Products not covered by any EU directive (such as most foods, cosmetics, and chemicals) are governed by separate EU legislation and do not carry the CE mark.
How long does CE marking certification take?
For lower-risk products using self-declaration (Module A), the process typically takes 4–12 weeks. For high-risk products requiring notified body involvement — such as Class II/III medical devices or category III PPE — expect 6 to 18 months or longer, depending on the notified body's workload and your technical documentation readiness.
How much does CE marking certification cost?
Costs range from approximately €1,500–€6,000 for simple, low-risk products to €15,000–€80,000+ for high-risk products requiring full notified body assessment. Key cost drivers are product complexity, the number of applicable directives, testing requirements, and whether a notified body and consultant are needed.
Can a non-EU manufacturer apply for CE marking?
Yes. Manufacturers anywhere in the world can apply for CE marking — the process and requirements are the same. However, non-EU manufacturers must appoint an EU Authorised Representative (EU AR) who is based in the EU/EEA and is legally named in the Declaration of Conformity. The EU AR acts as the point of contact for EU authorities.
What is the difference between CE marking and ISO certification?
CE marking is a mandatory legal requirement for EU market access. ISO certification (e.g., ISO 9001, ISO 13485) is a voluntary international quality or management system standard. ISO certification does not replace CE marking, but a certified quality management system can support and simplify the CE marking conformity assessment process.
Does CE marking have an expiry date?
CE marking does not have a fixed expiry date. However, your certification must be reviewed and potentially updated whenever: the product is modified; the applicable directive or harmonised standards are revised; your notified body certificate expires; or a new regulation applies to your product. In practice, 2026 triggers major reviews for most businesses due to GPSR, ESPR, and the new Machinery Regulation.
What is the CE marking Declaration of Conformity?
The EU Declaration of Conformity (DoC) is the manufacturer's signed legal declaration that the product meets all applicable EU requirements. It must list all applicable directives, harmonised standards used, notified body details (if applicable), and be signed by an authorised person. Under GPSR 2023/988, the DoC must now be digitally accessible — via QR code or URL — for consumer products.
What happens if I sell non-CE-marked products in the EU?
Consequences range from product seizure at customs to formal market withdrawal orders, public recalls, fines, and in serious cases, criminal prosecution under national law in EU member states. Non-compliant products are also entered into the EU Safety Gate (RAPEX) rapid alert database, causing reputational damage across all 30 EEA countries simultaneously.
Q10. Do I need CE marking to sell on Amazon EU or other EU online marketplaces?
Yes. Under GPSR (EU) 2023/988 — fully in force since December 2024 — EU online marketplaces including Amazon EU, eBay EU, Zalando, and others are legally required to ensure that regulated products listed on their platforms carry valid CE marking, have accessible Declarations of Conformity, and meet traceability requirements. Non-compliant listings are subject to removal and marketplace-initiated reporting to authorities.





































