- CDSCO MD-42 Registration Certificate is the mandatory license for anyone selling, stocking, or distributing medical devices in India under the Medical Device Rules, 2017.
- It replaces the need for a generic drug license for device-only sellers and requires appointing competent technical staff and maintaining proper storage conditions.
- The license is valid in perpetuity, provided you pay a retention fee every 5 years, ensuring long-term business continuity.
Introduction

India's medical device distribution landscape has changed significantly over the last few years, and not everyone operating in this space has kept up with those changes. The Medical Device Rules, 2017 brought a clear, enforceable regulatory framework for anyone involved in selling, stocking, or distributing medical devices — and by 2025-2026, that framework is being actively enforced at the state level across the country.
The Central Drugs Standard Control Organization (CDSCO) and State Licensing Authorities are no longer treating unlicensed distribution as a minor paperwork gap. Drug Inspectors are conducting market surveillance. Corporate hospital chains have tightened their vendor compliance requirements. Government procurement portals like GeM now flag unlicensed device vendors automatically. The informal window that many distributors relied on — operate first, sort compliance later — has quietly shut.
If you sell, stock, distribute, or exhibit any notified medical device in India without a valid MD-42 Registration Certificate, your exposure is real. Stock seizure, invoice rejection by institutional buyers, and in serious cases, criminal prosecution under the Drugs and Cosmetics Act, 1940 — these are not theoretical risks anymore. They are documented outcomes of enforcement actions that have taken place across Maharashtra, Delhi, Karnataka, and other major states in the last two years.
What Is a CDSCO MD-42 Registration Certificate?
The CDSCO MD-42 Registration Certificate is the official license issued by the State Licensing Authority (SLA) that legally authorizes an entity to sell, stock, exhibit, offer for sale, or distribute medical devices anywhere in India. It is not a CDSCO central license—it is state-specific, which means if you operate warehouses in Maharashtra and Delhi, you need separate MD-42 licenses for each premises.
The origin of this requirement dates to the Medical Device Rules, 2017, which were notified under the Drugs and Cosmetics Act, 1940. Before MDR 2017, medical devices existed in a blurry regulatory zone, often handled under the same framework as pharmaceutical drugs. The 2017 rules created a distinct, devices-specific regulatory identity.
Understanding Form MD-41 vs. Form MD-42: Don't Confuse the Two
This is one of the most common points of confusion among first-time applicants. Here is the exact distinction:
| Form | What It Is | Who Uses It | Stage |
| Form MD-41 | Application Form | Applicant (Business Entity) | Submitted to apply for the license |
| Form MD-42 | Registration Certificate (License) | Issued by Licensing Authority | Received after approval |
Think of MD-41 as the application you file and MD-42 as the license you receive. You apply in MD-41 to obtain MD-42.
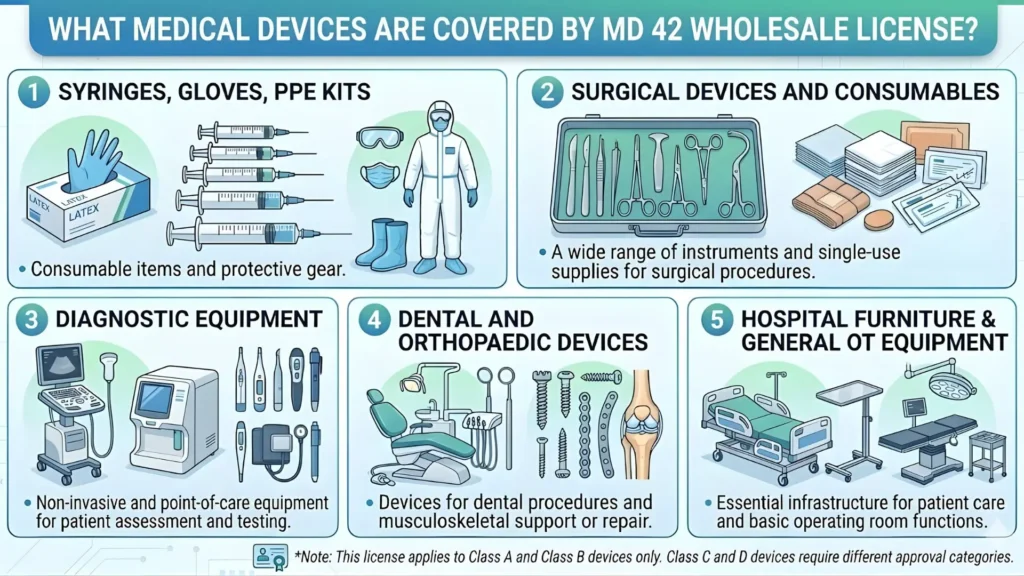
What Activities Does This License Actually Cover?
The MD-42 license covers the entire downstream supply chain for medical devices. Specifically, it covers:
- Selling medical devices from a physical premise
- Stocking or warehousing devices before distribution
- Exhibiting devices at trade counters or showrooms
- Distributing devices to hospitals, clinics, or other retailers
- E-commerce listing and sale of notified medical devices
It does not cover manufacturing (that requires different CDSCO approvals) or importing (which requires Form MD-14). It is squarely focused on the domestic distribution chain.
Difference Between Form MD-41 (Application) and MD-42 (Grant)
It is crucial to distinguish between the two forms to avoid confusion during your application:
- Form MD-41: This is the application form you fill out and submit to the licensing authority.
- Form MD-42: This is the actual license (certificate) you receive after approval.
- Think of MD-41 as your exam paper and MD-42 as your degree.
Who Needs an MD-42 License?
Does this apply to me? The short answer is yes—if you are anywhere in the supply chain between the manufacturer and the end user and you handle a notified medical device. Let's break it down by business type.
1. Medical Device Distributors and Wholesalers (B2B)
This is the primary target group. If you buy directly from manufacturers (Indian or foreign) and sell to hospitals, diagnostic labs, nursing homes, or smaller retailers, this license is non-negotiable. The license must be held for each warehouse or godown from which you operate.
2. Retailers and Dedicated Medical Device Shops
A shop that exclusively sells orthopaedic supports, hearing aids, glucose monitors, or other devices—without dispensing pharmaceutical drugs—needs the MD-42 license. Pharmacies that hold a Form 20/21 Drug License are technically covered for devices listed under Schedule K of the Drugs Act, but a growing number of state drug controllers are requiring the dedicated MD-42 for device-only operations.
3. Importers Acting as Domestic Distributors
You may already hold an MD-14 Import License. But the moment those imported goods land in your Indian warehouse and you start selling them domestically, that warehouse activity requires an MD-42. The MD-14 only covers the import clearance at the port of entry—it does not authorize domestic distribution.
4. E-Commerce Sellers of Medical Devices
Amazon, Flipkart, and other major platforms now mandatorily require you to upload your MD-42 Registration Certificate before listing notified medical devices like glucometers, BP monitors, pulse oximeters, or nebulizers. This requirement was enforced following CDSCO's directive to e-commerce platforms to ensure vendor compliance.
5. Pharmacies Adding a Medical Device Section
If a traditional pharmacy decides to set up a dedicated section for devices, the safest approach—and the one most state drug controllers now expect—is to obtain the MD-42 separately for that section or premises, rather than relying solely on the existing drug license.

Is the MD-42 License Mandatory?
Yes. It is mandatory under Rule 68 of the Medical Device Rules, 2017. The rule explicitly states that no person shall sell, stock, exhibit, or offer for sale or distribute any medical device without a valid registration certificate in Form MD-42. This is not an advisory—it is a legal prohibition.
Consequences of Operating Without an MD-42 License in 2026
State Drug Controllers have intensified enforcement operations in 2025-2026, particularly targeting the organized retail and e-commerce sectors. Here is what you risk:
| Consequence | Regulatory Basis | Severity |
| Seizure of entire device inventory | Section 18A, Drugs & Cosmetics Act | Immediate, irreversible loss |
| Criminal prosecution of the owner/director | Section 27, Drugs & Cosmetics Act | Fines + imprisonment up to 3 years |
| Business closure order by Drug Inspector | MDR 2017, Rule 68 | Operational shutdown |
| Blacklisting from hospital vendor panels | Hospital procurement policy | Long-term revenue loss |
| E-commerce listing takedown | Platform policy + CDSCO directive | Online sales blocked |
In 2025, multiple drug enforcement raids across Maharashtra, Delhi, and Karnataka resulted in crores of rupees worth of medical device inventory being seized from unlicensed distributors. Compliance is not optional.
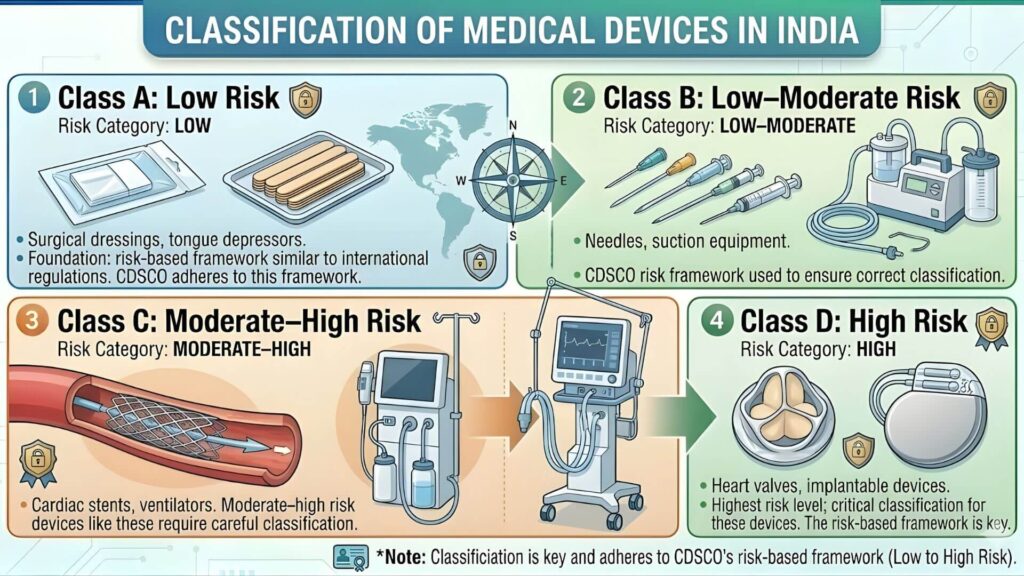
Understanding CDSCO Risk Classification
CDSCO classifies all notified medical devices into four risk categories. Your MD-42 license covers all four, but knowing where your products sit helps you understand the scrutiny level during inspection and the documentation your suppliers will carry.
| Risk Class | Risk Level | Examples | Regulatory Scrutiny |
|---|---|---|---|
| Class A | Low Risk | Bandages, examination gloves, tongue depressors | Lowest — self-declaration based |
| Class B | Low-Moderate Risk | Syringes, sutures, basic diagnostic equipment | Moderate — conformity assessment required |
| Class C | Moderate-High Risk | Ventilators, orthopaedic implants, IVD kits for HIV | High — third party audit required |
| Class D | Highest Risk | Cardiac stents, heart valves, active implantable devices | Strictest — CDSCO central review required |
Complete Product List Covered Under MD-42 Wholesale License
| Sr. No. | Product Name | Device Category | CDSCO Risk Class | Cold Chain Required | Key Regulatory Note |
|---|---|---|---|---|---|
| 1 | Cardiac Stents (Drug-Eluting) | Cardiovascular | Class D | No | Price-capped under NPPA; supplier must hold valid import/manufacturing license |
| 2 | Cardiac Stents (Bare Metal) | Cardiovascular | Class D | No | CDSCO central registration mandatory on supplier side |
| 3 | Heart Valves (Mechanical/Biological) | Cardiovascular | Class D | No | Biological valves may require cold chain — verify manufacturer label |
| 4 | Pacemakers | Cardiovascular | Class D | No | Serial number traceability mandatory at distributor level |
| 5 | Implantable Cardioverter Defibrillators (ICD) | Cardiovascular | Class D | No | Full batch traceability records required |
| 6 | Blood Pressure Monitors (Digital) | Cardiovascular/Diagnostic | Class B | No | One of the most commonly sold home-use devices under MD-42 |
| 7 | ECG Machines | Cardiovascular/Diagnostic | Class B | No | Widely distributed to clinics and diagnostic centres |
| 8 | Vascular Grafts | Cardiovascular | Class C | No | Sterility records must be maintained by distributor |
| 9 | Glucometers | Diagnostic | Class B | No | Mandatory MD-42 for e-commerce listing (Amazon/Flipkart) |
| 10 | Blood Glucose Test Strips | Diagnostic/IVD | Class B | No | Shelf life monitoring is critical — document expiry dates |
| 11 | Pulse Oximeters | Diagnostic | Class B | No | High-demand post-COVID; heavily regulated for quality |
| 12 | Digital Thermometers | Diagnostic | Class A | No | Low risk but still notified — MD-42 applies |
| 13 | MRI Machines | Imaging | Class C | No | High-value equipment — invoice and installation traceability required |
| 14 | CT Scan Equipment | Imaging | Class C | No | Radiation-emitting device; additional compliance on end-user side |
| 15 | X-Ray Machines | Imaging | Class C | No | Atomic Energy Regulatory Board (AERB) compliance required by end-user |
| 16 | Ultrasound Machines | Imaging | Class B | No | PC-PNDT Act compliance required by end-user clinic |
| 17 | Audiometers | Diagnostic | Class B | No | Calibration certificates from supplier are standard practice |
| 18 | HIV Rapid Diagnostic Kits | IVD | Class D | Yes (2°C–8°C) | Highest risk IVD — cold chain records are non-negotiable |
| 19 | Hepatitis B & C Test Kits | IVD | Class D | Yes (2°C–8°C) | Same cold chain requirements as HIV kits |
| 20 | Blood Grouping Reagents | IVD | Class C | Yes (2°C–8°C) | Used in blood banks — strict batch documentation |
| 21 | Dengue/Malaria/Typhoid Rapid Test Kits | IVD | Class C | Yes (2°C–8°C) | Seasonal demand spikes — maintain adequate cold chain capacity |
| 22 | COVID-19 Antigen Test Kits | IVD | Class C | Yes (2°C–8°C) | Still notified and regulated despite reduced demand |
| 23 | HbA1c Testing Kits | IVD | Class C | Yes (2°C–8°C) | Commonly distributed to diabetes clinics and labs |
| 24 | Pregnancy Test Kits | IVD | Class B | No | Available over-the-counter; MD-42 required for wholesale supply |
| 25 | Urine Analysis Strips | IVD | Class B | No | Shelf life and storage temperature range to be monitored |
| 26 | Hip Replacement Implants | Orthopaedic | Class D | No | Implant-specific traceability — patient card records must be maintained |
| 27 | Knee Replacement Implants | Orthopaedic | Class D | No | Same traceability requirements as hip implants |
| 28 | Spinal Implants and Cages | Orthopaedic | Class D | No | High-value; requires surgeon-specific consignment tracking in practice |
| 29 | Bone Screws, Plates and Rods | Orthopaedic | Class C | No | Batch and lot number tracking is standard practice |
| 30 | Orthopaedic Supports and Braces | Orthopaedic | Class A | No | Low risk; widely sold in retail medical device outlets |
| 31 | External Fixators | Orthopaedic | Class C | No | Sterility records required at distributor level |
| 32 | Bone Cement | Orthopaedic | Class C | Yes (controlled temp) | Verify manufacturer label for specific storage requirements |
| 33 | Intraocular Lenses (IOLs) | Ophthalmic | Class C | No | One of the most commonly implanted devices in India |
| 34 | Ophthalmic Viscosurgical Devices | Ophthalmic | Class C | Yes (2°C–8°C) | Cold chain mandatory — DI will verify refrigerator log |
| 35 | Contact Lenses (Corrective) | Ophthalmic | Class B | No | Separate CDSCO registration required per brand/SKU at supplier level |
| 36 | Contact Lens Solutions | Ophthalmic | Class A | No | Low risk but notified — covered under MD-42 |
| 37 | Tonometers | Ophthalmic | Class B | No | Distributed to ophthalmology clinics and hospitals |
| 38 | Dental Implants and Abutments | Dental | Class D | No | Traceability to implanting surgeon required in practice |
| 39 | Orthodontic Brackets and Wires | Dental | Class B | No | Commonly distributed to dental clinics and orthodontists |
| 40 | Dental Drills and Handpieces | Dental | Class B | No | Maintenance and sterility documentation from supplier |
| 41 | Dental X-Ray Units | Dental/Imaging | Class C | No | AERB compliance required by end-user |
| 42 | Impression Materials | Dental | Class A | No | Low-risk consumable widely used in dental practice |
| 43 | Ventilators | Critical Care | Class C | No | Post-COVID demand remains strong; institutional buyers dominant |
| 44 | Anaesthesia Machines | Critical Care | Class C | No | High-value capital equipment — installation records required |
| 45 | Patient Monitoring Systems | Critical Care | Class B | No | Widely distributed to hospitals, ICUs and nursing homes |
| 46 | Infusion Pumps | Critical Care | Class B | No | Calibration and maintenance records standard practice |
| 47 | Defibrillators | Critical Care | Class C | No | Serial number traceability and service records required |
| 48 | Electrosurgical Units (ESU) | Surgical | Class B | No | Commonly supplied to operation theatres and day-care centres |
| 49 | Laparoscopic Equipment | Surgical | Class B/C | No | Classification depends on specific product type |
| 50 | Surgical Instruments (Scissors, Forceps, Clamps) | Surgical | Class A/B | No | Reusable instruments — sterilisation documentation from supplier |
| 51 | Sutures (Absorbable and Non-Absorbable) | Surgical | Class B/C | No | Sterility and expiry date monitoring critical |
| 52 | Surgical Staples and Staplers | Surgical | Class B | No | Single-use — expiry and sterility records required |
| 53 | IV Cannulas | Disposables | Class B | No | High-volume consumable — batch traceability essential |
| 54 | IV Sets (Infusion Sets) | Disposables | Class B | No | Among the most widely distributed hospital consumables |
| 55 | Syringes (Disposable) | Disposables | Class B | No | Auto-disable syringes increasingly preferred under government supply |
| 56 | Hypodermic Needles | Disposables | Class B | No | Sold alongside syringes — same batch documentation applies |
| 57 | Urinary Catheters | Disposables | Class B | No | Available in Foley and intermittent variants |
| 58 | Central Venous Catheters (CVC) | Disposables | Class C | No | Higher risk — sterility documentation critical |
| 59 | Cardiac Catheters | Disposables/Cardiovascular | Class D | No | Catheterisation lab supply — full traceability required |
| 60 | Nasogastric Tubes | Disposables | Class B | No | Widely used in ICUs and general wards |
| 61 | Blood Bags | Disposables | Class C | No | Distributed primarily to blood banks and hospitals |
| 62 | Blood Transfusion Sets | Disposables | Class B | No | Batch and lot documentation required |
| 63 | Wound Dressings (Advanced) | Wound Care | Class B/C | No | Hydrocolloid and foam dressings are increasingly notified |
| 64 | Standard Bandages and Gauze | Wound Care | Class A | No | Lowest risk category — basic documentation sufficient |
| 65 | Surgical Gloves (Sterile) | Disposables | Class B | No | Sterility and expiry monitoring required |
| 66 | Examination Gloves (Non-Sterile) | Disposables | Class A | No | High-volume, low-risk — widely distributed |
| 67 | Dialysis Machines (Haemodialysis) | Nephrology | Class C | No | Distributed to dialysis centres and nephrology departments |
| 68 | Dialysis Filters and Cartridges | Nephrology | Class C | No | Single-use — batch traceability essential |
| 69 | AV Fistula Needles | Nephrology | Class B | No | High-volume consumable for dialysis patients |
| 70 | Peritoneal Dialysis Kits | Nephrology | Class C | No | Cold chain requirements vary — verify manufacturer label |
| 71 | Nebulizers | Respiratory | Class B | No | Strong retail and e-commerce demand — MD-42 mandatory for online sale |
| 72 | Oxygen Concentrators | Respiratory | Class C | No | Post-COVID regulation tightened significantly |
| 73 | CPAP and BiPAP Machines | Respiratory | Class B | No | Growing demand from sleep disorder and respiratory clinics |
| 74 | Oxygen Masks and Nasal Cannulas | Respiratory | Class A | No | Low-risk consumable — widely distributed |
| 75 | Spirometers | Respiratory | Class B | No | Distributed to pulmonology clinics and diagnostic centres |
| 76 | TENS Machines | Physiotherapy | Class B | No | Widely sold through physiotherapy equipment retailers |
| 77 | Ultrasound Therapy Units | Physiotherapy | Class B | No | Different from diagnostic ultrasound — used in rehabilitation |
| 78 | Hearing Aids | Rehabilitation | Class B | No | Regulated under MDR 2017 — MD-42 required for retail sale |
| 79 | Wheelchairs (Medical Grade) | Rehabilitation | Class A | No | Medical-grade variants are notified — standard wheelchairs may vary |
| 80 | Surgical Drapes and Gowns | Surgical | Class A | No | Single-use — expiry and sterility documentation from supplier |
Important Note for Distributors
This list reflects products notified under the Medical Device Rules, 2017 as of 2026. CDSCO periodically expands the notified device list through gazette notifications. Always verify your specific product's current notification status on the official CDSCO website before finalising your application, and ensure your suppliers hold valid manufacturing or import licenses for every product line you intend to distribute.
Eligibility Criteria for CDSCO MD-42 License: Who Can Apply?
Any registered legal business entity in India can apply for the MD-42 license. This includes sole proprietorships, partnerships, LLPs, and private limited companies. However, the entity must satisfy two core eligibility requirements: qualified technical supervision and adequate premises.
Technical Staff Requirement: Registered Pharmacist or Competent Person
This is the most important eligibility criterion. The business must have a qualified person supervising its device operations. You have two options:
| Category | Qualification Required | Experience Required | Documentation Needed |
| Registered Pharmacist | D.Pharm or B.Pharm with State Registration | None mandated (registration itself is proof) | Registration Certificate from State Pharmacy Council |
| Competent Person | B.Sc. / B.Pharm / MBBS / BDS from a recognized university | Minimum 1 year documented experience in medical device dealing | Degree Certificate + Experience Letter from a licensed entity |
One critical operational point: this qualified person must be physically present at the licensed premises during business hours. If a Drug Inspector arrives and the named technical staff member is absent, it can result in rejection or license suspension.
Business Entity Requirements
- The applicant firm must be a legally registered entity (GST registration is typically required)
- The firm must own or hold a valid commercial lease for the premises where the license is sought
- The premise must be a permanent, brick-and-mortar structure—not a temporary setup or residential address
Premises and Infrastructure Requirements for MD-42 Approval
The Drug Inspector's physical visit is the make-or-break moment of your MD-42 application. Here is exactly what they look for. Preparing your premises before the inspection is the single most effective way to avoid delays.
Storage Infrastructure: What the Inspector Will Check
- Wooden or metal storage racks: Goods must be stored on racks, never directly on the floor
- Pallets: Required for carton-level stock to prevent moisture damage
- Separate quarantine area: A physically distinct section for expired, damaged, or returned goods
- Labelling: 'Saleable Stock' and 'Quarantine/Expired Stock' sections must be clearly marked
- Adequate aisle space: The inspector must be able to walk through and inspect stock without obstruction
Cold Chain Infrastructure (Condition-Based Requirement)
Cold chain requirements are not universal—they depend on the specific devices you handle. However, if any of the following apply to your product portfolio, cold chain infrastructure is mandatory:
- In-vitro Diagnostic (IVD) reagents and kits
- Certain orthopaedic bone cements requiring controlled temperature
- Ophthalmic viscosurgical devices
- Any device where the manufacturer's label specifies a storage temperature range
If cold chain is required for your products, you must have a dedicated refrigerator (not shared with food or other items) with a digital temperature data logger that records temperature continuously. The inspector will ask to see the temperature log.
Hygiene and Facility Standards
- The premise must be free of dust, pests, and rodents (a pest control certificate helps)
- There must be no standing water, dampness, or mold—especially important for device integrity
- The premise should not be shared with a business that could contaminate the devices (e.g., a chemical store next door with shared storage)
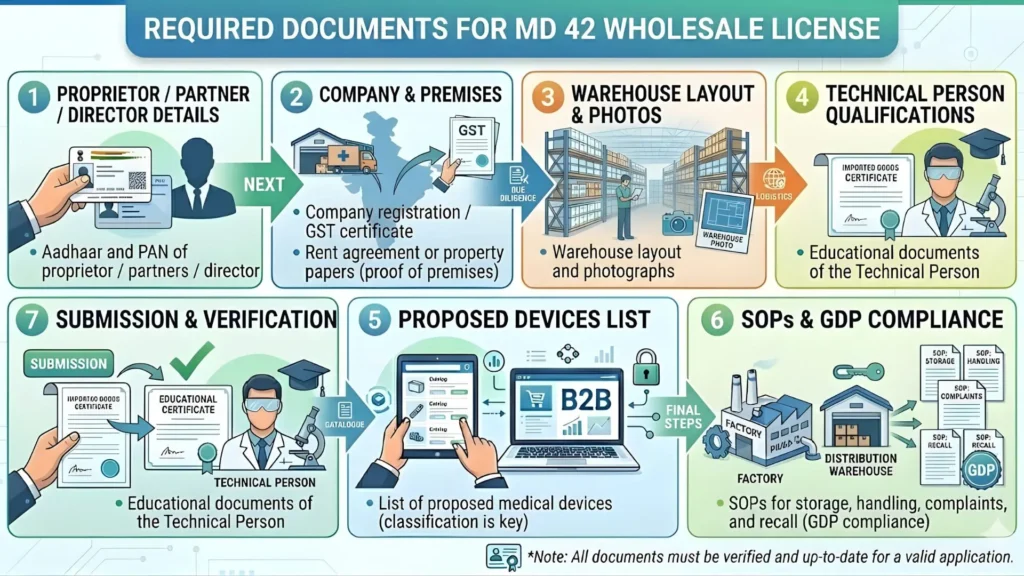
Documents Required for MD-42 Registration Certificate (2026 Checklist)
Document preparation is where most applications run into trouble. A mismatch between the name on the rent agreement and the GST certificate, or a missing NOC, can delay your application by weeks. Use this checklist methodically.
Category 1: Business Constitution Documents
- PAN Card of the firm and the proprietor/directors
- GST Registration Certificate
- Certificate of Incorporation (for companies) OR Partnership Deed (for firms) OR UDYAM Certificate (for proprietorships)
- Memorandum and Articles of Association (for private limited companies)
Category 2: Premises Proof
- Registered Rent Agreement (must specify 'commercial' or 'mixed land use'—residential agreements are rejected)
- Latest Electricity Bill of the premises
- No Objection Certificate (NOC) from the landlord, specifically consenting to use the premises for medical device storage/sale
- Site Layout Plan drawn to scale—must clearly show entrance, storage racks, cold storage (if applicable), and quarantine area
Category 3: Technical Staff Documents
- Educational Degree Certificate (B.Sc. / B.Pharm / MBBS)
- State Pharmacy Council Registration Certificate (for pharmacists only)
- Experience Letter on the previous employer's letterhead (for Competent Persons)—must state the period, nature of work, and that the firm holds a valid license
- Appointment Letter from the current firm to the Competent Person
- Acceptance Letter from the Competent Person (accepting the appointment)
- Identity Proof of the Competent Person (Aadhaar/PAN)
Category 4: Operational Documents (Prepared Before Inspection)
- Undertaking/Declaration by the applicant regarding compliance with MDR 2017
- List of medical devices proposed to be sold (product-wise or category-wise)
- Affidavit of no previous license rejection or suspension (required by some states)
Step-by-Step Process to Obtain CDSCO MD-42 Registration Certificate
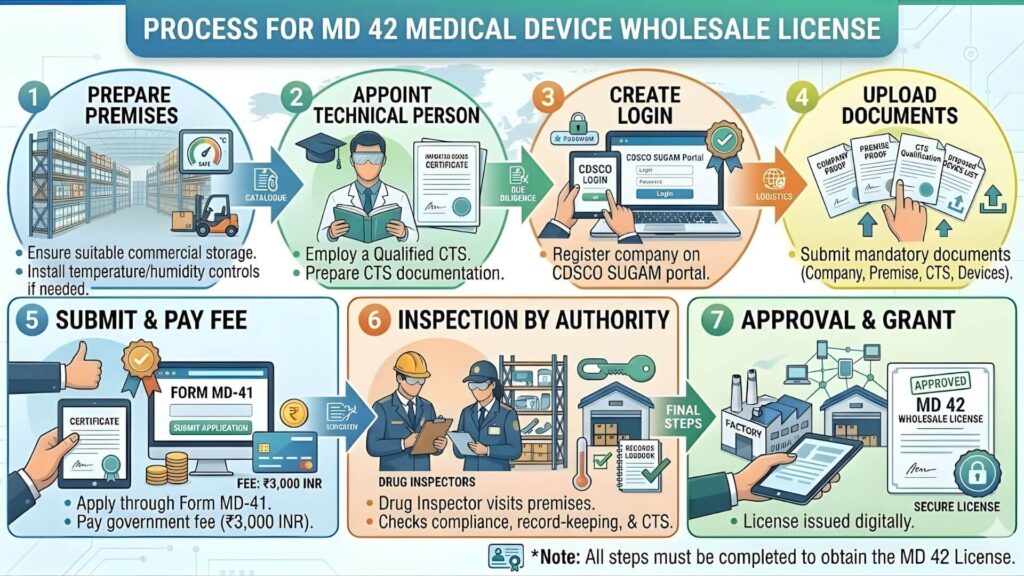
The MD-42 process is managed by the State Licensing Authority (SLA), not directly by CDSCO. Each state has its own portal and slightly different operational procedures, but the core steps are consistent across India.
Step 1: Pre-Application Audit of Your Premises (Do This First)
Before touching the portal, physically audit your premises against the checklist above. Most rejections happen because applicants skip this step. Walk through your storage area and ask: Are goods off the floor? Is there a quarantine area? Is the technical staff actually available during business hours? Fix gaps before you apply.
Step 2: Portal Registration and Application Filing
The application is filed online through the state-specific portal. States vary in their platforms:
- Delhi, Haryana, Punjab, Himachal Pradesh, Rajasthan: XLN Portal (xlnc.cdsco.gov.in or state-specific URL)
- Gujarat: eAushadhi Portal
- Maharashtra: SUGAM Portal or Aaple Sarkar
- Some states: NSWS (National Single Window System)
- Karnataka, Tamil Nadu, Andhra Pradesh: State Drug Control Department portals
Create a firm account on the relevant portal, select the application type (Form MD-41 for new license), fill in all details, and upload scanned documents. Ensure all document scans are clear and under the file size limit (usually 2 MB per document).
Step 3: Fee Payment
After form submission, pay the government fee of Rs. 3,000 online through the portal's payment gateway. Keep the payment receipt—it is required during the inspection and for all future correspondence.
Step 4: Drug Inspector's Physical Inspection
After document review by the Licensing Authority (which typically takes 7-15 days), a Drug Inspector (DI) is assigned to visit your premises. The inspection is not pre-announced with a specific time in most states, though you will receive a date window. During the visit, the DI will:
- Physically verify the premises address matches the application
- Inspect the storage infrastructure (racks, pallets, quarantine area)
- Interview the Competent Person and verify their physical presence
- Check the site layout against the submitted plan
- Verify cold chain equipment (if applicable)
- Review any existing purchase or distribution records
Step 5: Query Resolution (If Raised)
After the inspection, the DI submits a report to the Licensing Authority. If there are observations or deficiencies, the applicant receives a query letter. Common queries include requests to increase storage space, fix the quarantine area, or provide additional documentation. You must respond to queries within the stipulated time (usually 30 days) or the application lapses.
Step 6: Grant of MD-42 Certificate
Once the Licensing Authority is satisfied with the inspection report and query responses, the MD-42 Registration Certificate is issued digitally on the portal. You can download the digitally signed certificate. Store multiple copies—both digital and physical.
CDSCO MD-42 License Fees and Official Timeline (2026)
The fee structure is governed by the Second Schedule of the Medical Device Rules, 2017. These are statutory government fees and cannot be waived or negotiated.
| Activity | Form/Reference | Govt. Fee (INR) | Official Timeline | Validity |
| Application for New Grant | Form MD-41 | Rs. 3,000 | 10 days after DI satisfaction | Perpetual (Lifetime) |
| License Retention Fee | N/A (Portal payment) | Rs. 3,000 | Pay before 5-year mark | Keeps license active for next 5 years |
| Duplicate Certificate | Application to SLA | Rs. 500 | 7-10 working days | N/A |
| Amendment (change of address, staff) | Amendment form | Rs. 1,000 - Rs. 2,000 (state-dependent) | 15-30 days | Updated license issued |
The official timeline of 10 days refers to the period after the Drug Inspector has submitted a satisfactory inspection report. The actual end-to-end timeline—from application submission to receiving the certificate—is typically 30 to 45 days in most states. States with higher application volumes (Maharashtra, Delhi) may take 45-60 days. Factor this into your business planning.
MD-42 License Validity, Retention Fee, and What Happens If You Miss It
The MD-42 is one of the few regulatory licenses in India that does not have a fixed expiry date. It is valid in perpetuity—meaning it remains valid for the lifetime of the business, as long as the 5-year retention fee is paid. This is a significant advantage over pharmaceutical drug licenses, which require active renewal.
The 5-Year Retention Fee: How It Works
- The fee is Rs. 3,000, identical to the grant fee
- Payment must be made before the completion of each 5-year block from the date of original grant
- Payment is made through the same state portal used for the original application
- After payment, a retention acknowledgment is generated—keep this document
Consequences of Missing the Retention Fee
Missing the retention fee does not result in immediate criminal action, but it does result in automatic suspension of the license. An automatically suspended license means:
- You are technically operating without a valid license—all the legal consequences of unlicensed operation apply
- Hospitals and government procurement agencies will reject your invoices
- Reinstating a suspended license requires application to the SLA, additional fees, and potentially another inspection
Set a calendar reminder 6 months before your 5-year retention date. Do not wait until the last week—portal technical issues or bank holiday backlogs can cause delays.
Top Reasons MD-42 Applications Get Rejected (And How to Avoid Them)
Based on practical experience with hundreds of applications across states, here are the most frequently encountered rejection and query reasons:
Reason 1: Mismatch in Documents
The single most common reason for queries is a name or address mismatch between documents. For example, the firm name on the rent agreement uses 'Pvt. Ltd.' while the GST uses 'Private Limited.' These must be perfectly consistent. Review every document for name, address, and entity type consistency before submission.
Reason 2: Inadequate or Poorly Described Premises
Applying for a wholesale license with a 100 sq. ft. storage room raises immediate red flags. While no minimum square footage is legally prescribed, the DI exercises judgment. Ensure your space is genuinely adequate for the volume of stock you intend to hold, and ensure the site layout plan clearly shows this.
Reason 3: Technical Staff Credibility Issues
If the experience letter for a Competent Person appears inconsistent with their qualification timeline (e.g., the experience period overlaps with their college years, or the previous employer cannot be verified), the application is flagged. Use authentic experience letters from verifiable, licensed entities.
Reason 4: Technical Staff Not Present at Inspection
The DI may arrive without advance notice within the window given. If your Competent Person is unavailable on inspection day, the DI notes it, and the application is queried or rejected outright. The staff must be available throughout the inspection window.
Reason 5: No Dedicated Quarantine Area
The DI specifically looks for a physically separated area (even if it is just a clearly labeled section of a rack) for expired or damaged goods. Mixed storage is a compliance violation and will result in a query.
Reason 6: Using a Residential Address
Some applicants in smaller towns attempt to use residential addresses to save on commercial rent. Drug Inspectors are trained to identify residential setups. Without a clear commercial premise—separate entrance, racks, no domestic goods visible—the application will be rejected.
Business Benefits of Holding an MD-42 Registration Certificate
Regulatory compliance is often discussed purely in terms of avoiding penalties. But for medical device businesses in India's 2026 market, the MD-42 certificate is a genuine competitive advantage.
Access to Government Tenders and GeM Portal
Any medical device supply to government hospitals, defence establishments, AIIMS, ESI hospitals, or state health departments requires the MD-42 certificate as a mandatory document. On the Government e-Marketplace (GeM), the certificate must be uploaded against your seller profile for device categories. Without it, your bid will be technically disqualified regardless of price.
Onboarding with Major Hospital Chains
Corporate hospital chains—Apollo, Max, Fortis, Medanta, Narayana Health—all have standardized vendor onboarding processes that include regulatory document verification. The MD-42 is a non-negotiable requirement. Getting listed as a vendor with even one major chain can transform your business volume.
Authorized Distributor Appointments by Global Brands
Multinational medical device companies like Medtronic, BD, Siemens Healthineers, Philips Healthcare, and Abbott now include the MD-42 license as a prerequisite in their distributor appointment contracts. If you aspire to represent global brands in your region, this license is your entry ticket.
Protection from Regulatory Harassment
A valid MD-42 significantly reduces your vulnerability during Drug Inspector inspections. When the DI sees that your documentation is in order, the inspection becomes a routine formality rather than a business threat.
E-Commerce Platform Listing
As mentioned earlier, Amazon India, Flipkart, and 1mg now require MD-42 certificates for medical device listings. This opens a significant additional revenue channel that is completely inaccessible without the license.
MD-42 vs. Other Medical Device Licenses
| License | Form | Purpose | Issuing Authority | Who Needs It |
| Sale/Distribution License | MD-42 | Sell, stock, distribute devices domestically | State Licensing Authority (SLA) | Distributors, retailers, wholesalers |
| Import License | MD-14 | Import devices from foreign countries | CDSCO (Central) | Importers / authorized Indian representatives |
| Loan License (Stocking) | MD-42 (variant) | Stock devices on behalf of another licensed entity | State Licensing Authority (SLA) | Third-party logistics / C&F agents |
| Manufacturing License | MD-5 / MD-9 | Manufacture devices in India | CDSCO (Central/State) | Device manufacturers |
| Clinical Investigation | Form MD-22 | Conduct clinical studies on devices | CDSCO (Central) | Hospitals / research institutions |
Why Choose Silvereye Certifications for Your MD-42 Application?
The CDSCO MD-42 process sounds straightforward on paper, but the state-specific portal nuances, the variable Drug Inspector expectations, and the document precision required make errors surprisingly easy and costly. A single rejected application can delay your business by 60-90 days.
Silvereye Certifications specializes exclusively in medical device regulatory compliance. Here is what we bring to your MD-42 application:
- Deep knowledge of state-specific portal requirements across all major Indian states
- Pre-inspection audit of your premises with a detailed compliance checklist—before the DI arrives
- Complete document preparation, verification, and quality check to eliminate mismatches
- Real-time query tracking and response management so your application never lapses
- Post-grant support including retention fee reminders, amendment filings, and regulatory updates
Silvereye Certifications has helped hundreds of medical device businesses across India secure their MD-42 licenses. Our practical, audit-first approach is why clients experience a significantly higher first-attempt success rate than the national average.
Conclusion: Your MD-42 License Is a Business Asset, Not a Burden
India's medical device market is growing rapidly—projected to reach $50 billion by 2030. Every major institutional buyer, hospital chain, and global manufacturer is tightening their vendor compliance requirements. In this environment, your CDSCO MD-42 Registration Certificate is not just a piece of paper—it is your license to participate in India's healthcare growth story.
The process has a learning curve, but it is entirely manageable with the right preparation. Focus on your premises, your technical staff, and your documents. Address any gaps before you apply. And if you want to navigate the process without the risk of delays or rejections, partner with specialists who do this every day.
Get your MD-42 sorted now—before a missed opportunity makes you wish you had.
Frequently Asked Questions: CDSCO MD-42 License
Is a Registered Pharmacist mandatory to apply for MD-42?
No. A Registered Pharmacist is automatically eligible, but you can also appoint a Competent Person—a graduate in Science or Pharmacy with one year of documented experience in medical device handling. Both are equally valid under the Medical Device Rules, 2017.
What is the difference between Form 20B/21B and MD-42?
Form 20B/21B is the wholesale drug license issued under the Drugs and Cosmetics Rules, 1945—it governs pharmaceutical drugs. Form MD-42 is the device-specific license under the Medical Device Rules, 2017. If you deal exclusively in medical devices, you need MD-42. If you deal in both drugs and devices, you need both licenses.
Can I operate from home or a residential address?
Generally, no. Drug Inspectors require a dedicated commercial premise with proper storage infrastructure. A residential address without a separate commercial entrance and device-specific storage will be rejected.
How long does the entire process take from application to receiving the certificate?
Officially, the law specifies that the license shall be issued within 10 days after the DI is satisfied. In practice, the complete process—document review, DI inspection, query resolution—takes 30 to 45 days in most states. High-volume states like Maharashtra and Delhi may take 45 to 60 days.
If I have an MD-14 Import License, do I still need MD-42?
Yes. The MD-14 Import License covers only the act of importing goods into India (customs clearance at the port of entry). The moment those goods are stored in your domestic warehouse and you begin distributing them to buyers within India, that warehouse activity requires a valid MD-42 Registration Certificate.
Does the MD-42 license expire?
The license itself is valid in perpetuity—it has no fixed expiry date. However, you must pay a retention fee of Rs. 3,000 every 5 years through the portal. Missing this payment results in automatic suspension of the license.
Is cold chain infrastructure mandatory for everyone?
No. Cold chain (refrigerated storage with a temperature data logger) is required only for devices that the manufacturer specifies must be stored at controlled temperatures. Common examples include IVD reagents, certain diagnostic kits, and specific ophthalmic devices. If none of your products have such requirements, you do not need cold chain infrastructure.
Do I need a separate MD-42 for each state or each premises?
Yes, to both. The MD-42 is premises-specific and state-specific. If you operate a warehouse in Gujarat and a branch in Rajasthan, you need two separate MD-42 licenses—one from the Gujarat SLA and one from the Rajasthan SLA.
Can I sell medical devices online with this license?
Yes. E-commerce platforms like Amazon and Flipkart require you to upload your MD-42 certificate to list medical devices. The license covers the sale irrespective of channel—in-store or online—as long as the physical storage and dispatch happens from a licensed premises.
What are the government fees, and are there any other costs?
The statutory government fee is Rs. 3,000 for a new grant, payable through the state portal. This does not include professional consultancy fees for documentation support, pre-inspection audit, and query resolution—which are separate costs that vary depending on the service provider and state.







